INTRODUCTION
Sulfated polysaccharides from marine sources are currently being investigated as therapeutics in a wide range of diseases. Several reports on marine natural products have described anticoagulant and non-anticoagulant effects of novel molecules without the undesirable side effects of heparin, which interacts with distinct plasma proteins1,2. Both positive and negative effects of heparin are related to its unique structural features such as heterogeneity of intra-chain domains and its highly negative charge and selective sulfation3. Recent studies have revealed other potential therapeutic effects of complex carbohydrates on many biological models of inflammation, cancer, metastasis, viral infection, atherosclerosis and acute tissue injury4,5.
Diverse structural features such as sugar composition, type of linkage, molecular weight (Mw), distribution and charge density allow polysaccharides from different sources to interact with a wide variety of molecules involved in biological processes3,5. For instance, the study of lacquer polysaccharides indicated that carboxyl and sulfate groups have a synergistic anticoagulant action; the presence of 6-O-SO3 in the side chains is a key feature for this activity while sulfation at 2-position has not such effect. Sulfation of alginate, a polymer of (1 → 4) linked β-D-mannuronic acid and α-L-guluronic acid that naturally contain carboxyl groups, increases its anticoagulant properties, at the same time, position of carboxyl groups may also have impact on this bioactivity2. The fucoidan obtained from Fucus vesiculosus displays procoagulant effect when it presents a minimal degree of sulfation of 0.5 and a chain of at least 70 sugar units6.
The tunic and organs of different ascidians have been studied as a source of new bioactive natural products7–8 910. Among those compounds, polysaccharides like L-galactans have shown to be the main constituent of this filter feeding marine invertebrate tunic[11,12]. These carbohydrates present different size and structure and they likely have distinct biological effects; for instance tunic of Herdmania monus is mainly composed by a 3-O-sulfated 4-linked α-L-galactan, the galactan from Styela plicata is also substituted by sugar residues at 2-position and Ciona intestinalis possess a poorly sulfated and highly branched α-L-galactan11-12 13.
Here, we aimed to evaluate anticoagulant and cytotoxic effects of a galactose-rich polysaccharide fraction obtained from the tunic of Microcosmus exasperatus. Data obtained revealed a mechanism of coagulation different from heparin-like anticoagulants at innocuous doses.
MATERIAL AND METHODS
Biological material
The ascidia Microcosmus exasperatus was collected at Guanabara Bay, Rio de Janeiro, Brazil. Tunics were separated from the internal organs and cleaned, depigmented and freeze-dried before extraction.
Extraction of the polysaccharides from the tunic
The defatted and dried tunics (64.9 g) of M. exasperatus were extracted by three successive proteolytic digestions in a 0.1 M sodium acetate buffer containing 5 mM EDTA, 5 mM cysteine and papain (10%); each enzymatic digestion was incubated for 24 h at 60 °C14. Supernatants of the three digestions were collected and polysaccharides present in this final solution were precipitated by the exhaustive addition of ethanol and kept at 4 °C for 24 h. Finally, the crude precipitate of polysaccharides was obtained by centrifugation (4500 rpm for 10 min at 15 °C), dialyzed using a 1 kDa MWCO dialysis tube and freeze-dried.
Fractionation of the crude precipitated of polysaccharides
The isolated extract (4.1 g) was dialyzed with deionized water by using a 1000 kDa molecular weight cut-off membrane; retained and eluted fractions were collected, evaporated and freeze-dried. The retained fraction was subsequently solubilized and submitted to freezing–thawing cycles until precipitation stopped; supernatants were separated from precipitates through centrifugation for 10 min at 10 °C.
The presence of negative charges in the carbohydrates, which could be assigned to sulfate groups, was tested through the complex formed between sulfated polysaccharides and dimethylmethylene blue (DMB).
Glycosyl composition analysis
1000RS fraction (2.0 mg) was hydrolyzed with 2.0 M trifluoroacetic acid (TFA) for 8 h at 100 °C. The obtained hydrolyzate was reduced (18 h) and acetylated (12 h) by using NaBH4 (pH 9.0) and acetic anhydride:pyridine (1:1, v/v) at room temperature, respectively. Alditol acetates were separated by GC–MS (Varian Saturn 2000R-3800) using a DB-225 column (30m × 0.25 mm i.d) and identified by comparing their retention time and mass fragmentation pattern with the alditol acetates of standard monosaccharides15.
Blood clotting assays
Anticoagulant activity of the fraction 1000RS at different concentrations (11.9-44.8 µg/mL) was determined by measuring the clotting time (in seconds) of unfrozen human platelet-poor plasma using an ACL ELITE PRO coagulometer at 37 °C. The activated partial thromboplastin time (aPTT), prothrombin time (PT), thrombin time (TT), anti-Xa factor and a dilute Russell’s Viper Venom time-based assay (Lupus anticoagulant-dRVVT) reagents were prepared and used according to the manufacturer’s (HemosIL®, Instrumentation Laboratory) instructions. Clot formation in PT, TT, aPTT and dRVVT assays was measured at 660 nm and calcium chloride was added to trigger coagulation reactions in aPTT assay. Unfractionated heparin (200.47 IU/mg; 6th International Standard) was used as positive control. Results were expressed as the mean of the response ± standard deviation (SD).
Chromogenic assay (anti-Xa factor) was made by incubating (30 s) 1000RS with plasma, Chromogenic substrate (anFXasub, 15 s) and Factor Xa reagent (anFxaRgt, 90 s). The concentration of residual Factor Xa was measured (60 s) by detection of paranitroaniline at 405 nm.
Cellular viability of hematopoietic cells from C57BL/6 mice (MTT assay)
Hematopoietic cells were obtained from the bone marrow of femurs and tibias of six male and female between 4 and 6 months C57BL/6 mice. After collecting bone marrow, cells were treated with ACK (Ammonium-Chloride-Potassium) solution to eliminate red cells and then incubated overnight in a flask with RPMI medium containing 10% (v/v) fetal bovine serum. Afterwards, only non-adherent cells that correspond to hematopoietic cells were collected and used subsequently. Experiments were done in 96-well plates with 5 x 105 cells/well (100 µL) and three different concentrations (0.5, 0.25 and 0.0078 mg/mL) of 1000RS and heparin were used for 24 h of culture. After this period, cells were incubated with 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazoliumbromide (MTT) aqueous solution (5 mg/mL, 20 µL/well) for 2 h, centrifuged at 200 g for 8 min and the supernatant solution was discarded. Formazan crystals were dissolved with a solution (200 µL) containing triton-X, HCl and isopropyl alcohol. Finally, the absorbance was measured at 560 nm in a plate reader. Cell viability was expressed as the percentage of viable cells normalized using control cells. Results are expressed as mean values and standard deviation (SD) of six different experiments in triplicate16.
All protocols used for the animal experiments were approved by the Animal Ethics committee of the Hospital Naval Marcílio Dias, Rio de Janeiro (Law n° 11.794 of 8 October 2008, Legislative Decree n° 6.899 of 15 July 2009).
RESULTS
1000RS represents a yield of 5.3% of the crude extract of polysaccharide obtained from M. exasperatus. It was a fraction mainly composed by galactose (>90%), while other monomers like arabinose, xylose, mannose and glucose are present in minor proportion. A solution of this fraction formed a complex with the cationic dye DMB and the intense coloration suggests that this fraction possess negatively charged polysaccharides.
To evaluate how this fraction affects the secondary hemostasis, aPTT, TT and PT assays (Table 1) were performed in vitro.
Table 1 Anticoagulant activity of the 1000RS fraction (Fig.1)
| Sample | Amount (µg/mL) | Clotting time (s) | ||
|---|---|---|---|---|
| aPTT | TT | PT | ||
| Control | 0.0 | 26.7 ± 0.0 | 15.9 ± 0.0 | 13.9 ± 0.0 |
| Heparin | 1.9 | 86.8 ± 6.4 | 169.5 ± 0.2 | 15.5 ± 0.3 |
| 2.8 | 163.7 ± 21.7 | 169.5 ± 0.3 | 16.9 ± 0.4 | |
| 3.7 | 247 ± 0.0 | 169.5 ± 0.4 | 18.4 ± 0.6 | |
| 1000RS | 11.9 | 53.4 ± 1.0 | 16.3 ± 0.1 | 13.9 ± 0.2 |
| 23.3 | 72.3 ± 1.8 | 16.1 ± 0.2 | 15.1 ± 0.5 | |
| 34.2 | 96.0 ± 4.0 | 19.4 ± 0.2 | 15.4 ± 0.1 | |
| 44.8 | 129.0 ± 9.5 | 21.9 ± 0.8 | 16.4 ± 0.2 | |
Clotting time (n=3) are expressed (in seconds) as the mean ± SD.
1000RS prolonged the aPTT in a dose dependent manner, whereas the TT and PT were poorly affected by the different concentrations of 1000RS (Fig.1).
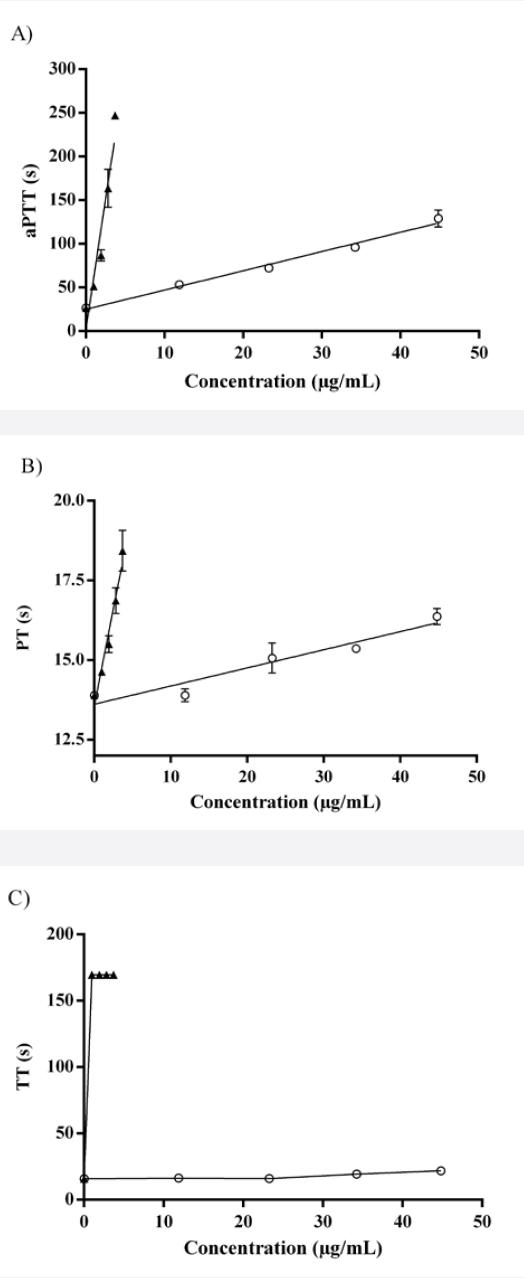
Figure 1 Anticoagulant activity of heparin (▲) and 1000RS fraction (○): A) aPTT, B) PT, C) TT. The highest reference time is 247.5 seconds for PTT, 165 for PT and 169.5 for TT. Standard curves (n=3) were analyzed by linear regression statistics and the coefficients of determination (r) were obtained for all of them; heparin curves: 0.97 (aPTT), 0.98 (PT), 0.72 (TT) and 1000RS curves: 0.99 (aPTT), 0.96 (PT), 0.89 (TT).
This fraction did not prolong the prothrombin and thrombin, suggesting that the inhibition of the coagulation may be due to an interaction with different coagulation factors such as factor X, V and II of the common pathway. For this reason the effect of 1000RS on factor X was evaluated through the anti-Xa assay. At the same time, to better understand how this fraction interferes with the thrombus formation, the lupus anticoagulant assay was also carried out (Table 2).
Table 2 Anti-Xa and Lupus anticoagulant assay of 1000RS fraction.
| Sample | Amount (µg/mL) | dRVVT (s) | Anti-Xa factor (UI/mL)a |
|---|---|---|---|
| Control | 0.0 | 30.1 ± 0.0 | 0.05 ± 0.01 |
| 1000RS | 11.9 | 26.6 ± 0.3 | 0.02 ± 0.02 |
| 23.3 | 26.1 ± 0.0 | 0.00 ± 0.01 | |
| 34.2 | 25.2 ± 0.2 | 0.00 ± 0.01 | |
| 44.8 | 27.3 ± 0.8 | 0.00 ± 0.03 |
dRVVT and anti-Xa values are represented as the mean of the values ± SD of n=3 independent experiments. aValues less than or equal to -0.2 UI/mL are undetectable, they are considered to be 0.00 UI/mL.
The anticoagulant activity of 1000RS is not due to the inhibition of the factor X, since the amount of residual Xa at different concentrations of the fraction was lower than the control (Table 2). At the same time, the activity of 1000RS as a lupus anticoagulant was evaluated using the diluted Russell’s Viper Venom test (dRVVT); this fraction did not impair the activation of factor X by an enzyme present in the viper venom.
Furthermore, as the ascidian polysaccharide fraction showed to be a potential anticoagulant fraction with probably less adverse effects than heparin or other known anticoagulants, it is important to determine if this fraction contains carbohydrates that could be cytotoxic at the anticoagulant doses. For this purpose, in vitro cell viability was evaluated with the MTT assay using hematopoietic cells obtained from C57BL/6 mice (Fig. 2)17.

Figure 2 Cellular viability of murine hematopoietic cells after treatment with the 1000RS (○) and Heparin (□) at different concentrations. Data are shown as mean of the % viability ± SD of at least n=6 independent experiments performed in triplicate.
After treatment of cells with 1000RS and heparin, the viability observed has not significantly changed and sometimes it was even higher than the control (set as 100% of viable cells). These results indicate that polysaccharides that constitutes 1000RS fraction are potential anticoagulant agents with non-cytotoxic effect at least at doses used here.
DISCUSSION
Previous reports on the polysaccharide composition of ascidian tunics had demonstrated that this tissue is mainly constituted by a high molecular weight galactan, which have different sulfation pattern according to the specie of tunicate11,18. This new fraction from M. exasperatus also showed the presence of polysaccharides that are mainly constituted by galactose residues, which may be negatively charged along the chain. Additionally, this fraction was retained by the 1000 kDa MWCO membrane, which supports the existence of high molecular weight polysaccharides in this ascidian.
Plasma proteases that are required for clot generation are common targets of several anticoagulants. Although those substances can effectively avoid thrombus formation and growth, their use represents a significant risk of excessive bleeding and hemostatic disorders19. Considering that sulfated polysaccharides have frequently been shown as anticoagulant substances, we hypothesized that 1000RS could be a potential inhibitor of the coagulation process20,21.
1000RS can significantly inhibit the intrinsic coagulation pathway, but neither the extrinsic pathway nor the thrombin-mediated fibrin formation are affected by this polysaccharide fraction22. Thus, it does not alter the coagulation process by interacting with factor VII or factor II (prothrombin) of the extrinsic and common coagulation pathways, respectively23. Additionally, the poor effect on thrombin and prothrombin time suggests that the extent of fibrinogen is normal and remains unaffected in the presence of this fraction, resulting in normal fibrin formation. Also, the lack of inhibitory activity on thrombin indicates that thrombin-induced activation of factor V, factor VIII and platelets, essential components of blood coagulation, remains unaltered24.
The aPTT prolongation by 1000RS was 22.2-fold lower than heparin; the clotting time increases, in average, 2.36 s (r= 0.9619) and 59.36 s (r= 0.9830) after the treatment at different concentrations of the polysaccharide fraction and heparin, respectively. Although heparin is more potent, this result showed that 1000RS has anticoagulant activity and probably inhibits clot formation through the interaction with one or more key intrinsic pathway clotting factors. Heparin acts by blocking activated factor X (factor Xa)23,24.
The amount of residual factor Xa after treatment with this fraction was lower than control at the different concentrations tested25, which means that it does not bind to the active site of activated Factor X (FXa) and allows the interaction between FXa and its substrate. Therefore the thrombin generation can be normally achieved26. Furthermore, an enzyme present in the viper venom did not activate factor X in the presence of 1000RS fraction; with this activated factor in the presence of factor V and phospholipid, prothrombin is converted to thrombin27–2829. Hence, it does not decrease the availability of phospholipids to support this reaction and the clot is formed at all the concentrations tested. The fact that this fraction does not act as a lupus anticoagulant, along with the negative effect on factor X and thrombin time, confirmed that it is not an inhibitor of the common coagulation pathway. This indicates that the anticoagulant effect is mainly due to the inhibition of an intrinsic coagulation factor such as prekallikrein, high molecular weight kininogen, XII, XI, IX and VIII factors23,29. Consequently, it can be assumed that 1000RS does not behave as a heparin-like anticoagulant, being a more specific inhibitor and possibly reducing some side effects related to the continuous use of anticoagulants such as heparin. Nevertheless, the fact that this inhibitor may acts directly against a specific coagulation factor could represent a bleeding risk factor.
This fraction did not display cytotoxic effect on hematopoietic cells obtained from C57BL/6 mice at anticoagulant doses. Cellular viability was higher than that obtained with the control at some of tested concentrations. It could directly correlate to the increase in number of metabolic active cells. Those murine hematopoietic stem cells continue viable after treatment at different concentrations of 1000RS and heparin.
CONCLUSION
The highest anticoagulant effect of 1000RS occurs at innocuous doses, however, the anticoagulant effect and innocuousness should be done using in vivo anticoagulant models and more accurate viability assays with normal human cell lines. This outcome is also promising when comparing 1000RS with other bioactive polysaccharides such cellulose, starch sulfuric acid esters and chitin disulfuric acid, which have been shown to be considerably cytotoxic30.
Regarding the lack of activity on factor X or thrombin, it could be assumed that 1000RS does not interact with some serine protease inhibitors (serpins) such as antithrombin III (ATIII) and heparin cofactor II (HCII); regulatory plasmatic proteins which mainly inactivate these two serine proteases in plasma31. Once again, it supports the idea that the ascidian polysaccharide fraction does not have the same mechanism of action shared by several sulfated polysaccharides that, like heparin, has an anticoagulant action based on the potentiation of these natural inhibitors. This opens the possibility of having a new anticoagulant substance that can catalyze the allosteric change of other serpins like kallistatin (PI4), protein C inhibitor (PCI), Protein Z-dependent proteinase inhibitor (ZPI), which can inhibit some homeostatic blood proteases of the intrinsic pathway like kallikrein, active protein C and FXIa, respectively. It is also possible that it can act joining the protease with its respectively serpin through a “bridge” made of its glycosidic chain32,33.














