Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Nefrología (Madrid)
versión On-line ISSN 1989-2284versión impresa ISSN 0211-6995
Nefrología (Madr.) vol.34 no.5 Cantabria 2014
https://dx.doi.org/10.3265/Nefrologia.pre2014.Jul.12527
ORIGINALS
Effect of calcium acetate/magnesium carbonate in the treatment of hyperphosphataemia in dialysis patients in real clinical practice. One year follow up
Efectos del acetato cálcico/carbonato magnésico en el tratamiento de la hiperfosfatemia en pacientes en diálisis en la práctica clínica real. Seguimiento durante un año
Ángel L.M. de Francisco, Lara Belmar, Celestino Piñera, María Kislikova, Miguel Seras, Mara Serrano, Zoila Albines, Cristina Sango and Manuel Arias
Servicio de Nefrología. Hospital Universitario Marqués de Valdecilla. Santander, Cantabria
ABSTRACT
Background: This observational study was conducted to investigate the use and effectiveness of calcium acetate/magnesium carbonate (CaMg) in the treatment of hyperphosphataemia in dialysis patients in real-world clinical practice.
Methods: 120 adult CKD patients on dialysis who received CaMg alone or in combination with other phosphate binders were followed-up for 3-12 months. Serum phosphorus, calcium, magnesium, parathyroid hormone and albumin concentration was measured at baseline and after 3, 6 and 12 months respectively. In addition, CaMg dosage, use of concurrent phosphate binders, vitamin D and cinacalcet was documented. Patients were evaluated in 2 subgroups - CaMg alone (n=79) vs. CaMg + concurrent phosphate binder (n=41).
Results: In both subgroups serum phosphorus levels decreased significantly from baseline at 3, 6 and 12 months of CaMg treatment. The percentage achievement of recommended serum phosphorus targets improved after CaMg initiation. At month 6, a total of 78% were within the Kidney Disease Outcomes Quality Initiative (K/DOQI) target range. Total corrected serum calcium increased during CaMg treatment, but mildly exceeded the upper limit of normal in three patients only. Asymptomatic significant increases in magnesium (p<0.001) were observed in the monotherapy group at 3, 6 and 12 months. A total of 80 patients (67%) experienced episodes of mild hypermagnesaemia (>2.6mg/mL, 1.05mmol/L).
Conclusions: This analysis of current clinical practice shows that - consistent with findings from a randomised controlled trial - CaMg treatment leads to marked improvement in serum phosphorus levels, helping patients in trying to achieve K/DOQI and KDIGO (Kidney Disease Improving Global Outcome) targets.
Key words: Calcium acetate/magnesium carbonate, Hyperphosphataemia, Phosphate binder, Dialysis.
RESUMEN
Antecedentes: Este estudio observacional se llevó a cabo para investigar el uso y la efectividad, en la práctica clínica real, del acetato cálcico/carbonato magnésico (CaMg) en el tratamiento de la hiperfosfatemia en pacientes en diálisis.
Métodos: Se realizó un seguimiento durante 3-12 meses en 120 pacientes adultos con enfermedad crónica renal en tratamiento con diálisis que recibían monotratamiento con CaMg o en combinación con otros quelantes del fósforo. Se midieron en suero los valores de fósforo, calcio, magnesio, hormona paratiroidea y concentración de albúmina a nivel basal y tras 3, 6 y 12 meses, respectivamente. Además, se documentó la dosis de CaMg, el uso de quelantes de fósforo concomitantes, la vitamina D y el cinacalcet. Los pacientes se dividieron en 2 subgrupos: aquellos a los que solo se les administraba CaMg (n=79) frente a los que recibían CaMg y un quelante de fósforo concomitante (n=41).
Resultados: En ambos subgrupos, los niveles de fósforo sérico disminuyeron de forma significativa, con respecto a los basales, a los 3, 6 y 12 meses de tratamiento con CaMg. El porcentaje de logro de los niveles recomendados de fósforo sérico mejoró tras iniciar el tratamiento con CaMg. El mes 6, un total del 78% se encontraba dentro de las recomendaciones objetivo de Calidad de los Resultados de la Insuficiencia Renal (K/DOQI). El calcio sérico total corregido aumentó durante el tratamiento con CaMg, pero superaba levemente los límites superiores normales solo en tres pacientes. Asimismo, se observaron incrementos significativos del magnesio asintomáticos (P<0,001) en el grupo de monoterapia a los 3, 6 y 12 meses. Un total de 80 pacientes (67%) sufrieron episodios de hipermagnesemia leve (>2,6 mg/mL, 1,05 mmol/L).
Conclusiones: El presente análisis de la práctica clínica habitual, en consonancia con los datos obtenidos de un ensayo aleatorizado controlado, demuestra que el tratamiento con CaMg mejora de forma considerable los niveles de fósforo sérico y ayuda a los pacientes a conseguir los objetivos K/DOQI y KDIGO (mejora de los resultados globales en la enfermedad renal).
Palabras clave: Acetato cálcico/carbonato magnésico, Hiperfosfatemia, Quelante de fósforo, Diálisis.
Introduction
A number of US1 and European2 observational studies reported an increased mortality risk with hyperphosphataemia in dialysis patients. Dietary restriction of phosphate and dialysis are usually not sufficient to control hyperphosphataemia. Consequently, oral phosphate binders are administered in the majority of dialysis patients.
The ideal phosphate binder would avidly bind dietary phosphate, have minimal systemic absorption, few side effects, a low pill burden and be inexpensive. Calculations based on the doses usually applied indicated that the cost of treatment with sevelamer- or lanthanum-based agents is substantially higher than the cost of calcium- or magnesium-based binders. Based on total sales figures, annual costs of sevelamer or lanthanum exceeded those of calcium- or magnesium-based binders by four to eight times or more, depending on the dose.3
Calcium acetate/magnesium carbonate (CaMg) is a combination phosphate binder. Both components, administered separately or together, are already well-established phosphate-lowering agents.4-14 As high doses of phosphate binders are often required to achieve sufficient phosphate reduction, the risk of hypercalcaemia must be considered when using a pure calcium salt as a phosphate binder. In calcium-magnesium combined preparations the proportion of calcium is reduced compared to drugs containing calcium salts only, thus limiting the risk of hypercalcaemia and of a continuously positive calcium balance.15 A controlled randomised trial investigated the effect of CaMg on serum phosphorus levels compared with sevelamer hydrochloride (HCl). CaMg was non-inferior to the comparator in controlling serum phosphorus levels at Week 25. There was no change in ionised calcium; there was minimal increase in total serum calcium and a small increase in serum magnesium. CaMg had a good tolerability profile and thus may represent an effective treatment of hyperphosphataemia.16
However, controlled clinical trials do not reflect the real world of clinical practice. The use of less stringent inclusion/exclusion criteria in observational studies allows a closer reflection of the overall patient population than in randomised clinical trials.17 There is currently little information available on CaMg in real-life clinical practice. The aim of this study was to investigate the use and effectiveness of CaMg in real-world clinical practice in patients on dialysis with hyperphosphataemia.
Subjects and methods
Study population
Patients aged ≥18 years with chronic kidney disease (CKD) stage 5 on haemodialysis (HD) or peritoneal dialysis (PD) who had been prescribed CaMg to control serum phosphorus participated in this study. The study was conducted in conformity with International Conference on Harmonisation - Good Clinical Practice (ICH-GCP) and the Declaration of Helsinki. All patients provided their written informed consent prior to entering the study.
Study design and procedure
This open-label, prospective non-interventional observational study enrolled patients from a single centre (Servicio de Nefrología, Hospital Universitario Marqués de Valdecilla, Universidad de Cantabria, Santander, Spain) between July 2010 and November 2013. The treating physician exclusively determined initiation, dose and duration of CaMg treatment based on recommendations outlined in department protocols. Accordingly, patients on phosphate binder treatment with inadequate phosphorus control or high pill burden or treatment-naïve patients with P >5.5mg/dL (1.8mmol/L), Ca <10.4mg/dL (2.6mmol/L), Mg <3.4mg/dL (1.4mmol/L), parathyroid hormone (PTH) >150pg/mL (16.5pmol/L) and Kt/V ≥1.3 received CaMg (calcium acetate 435mg containing 110mg elemental calcium combined with magnesium carbonate 235mg containing 60mg elemental magnesium [OsvaRen®]) at doses determined by the treating physician. Patients received an initial dose prescribed by physicians related to serum phosphorus levels but in general 3-4 pills/day. Dose increase or decrease was based on serum phosphorus levels. Co-medication was maintained according to patient requirements. No clinic visits were required other than those regularly scheduled. No laboratory or diagnostic tests were performed other than those associated with usual patient care. Routine biochemical parameters were measured using standard methods. Measurement of intact PTH was performed using the Scantibodies Total Intact PTH (Abraham Way, Santee, CA 92071).
Besides patient demographics PTH, serum phosphorus, calcium, magnesium and albumin concentration was obtained at baseline and after 3, 6 and 12 months of CaMg treatment respectively. CaMg dosage, concurrent phosphate binder usage, vitamin D and cinacalcet usage were documented throughout the study. Tolerability was assessed by means of adverse event profile and safety laboratory parameters at each study visit.
The primary endpoint of this study was efficacy of CaMg in real clinical practice, measured as change in serum phosphorus over one year.
Statistical analysis
All data analyses were carried out according to a pre-established analysis plan. The collected data were analysed with epidemiological methods, using the SPSS for Windows programme package (Version 15.0, Chicago, IL). For continuous variables, statistic parameters including arithmetic mean, standard deviation and range were calculated. Frequency distributions for discrete variables were provided as percentage in relation to the total sample. Evaluation of parameters measuring the clinical course was performed by intraindividual difference analysis using the Student's t-test. Differences were calculated per patient and subsequently averaged. For normality and homogeneity of data, Kolgomorov-Smirnow and Levene tests were used. For comparison between groups, the T-Test and Mann-Whitney U test was applied. Missing data were not imputed. All tests were two-sided, and significance was declared at the 0.05 level. Corrected calcium (mg/dL) was calculated as total calcium (mg/dL) + 0.8 [4.0 - albumin (g/dL)]. For analysis, patients were divided post-hoc into 2 subgroups. Group A comprised patients receiving CaMg alone, group B comprised patients receiving CaMg in addition to one or more other phosphate binders.
Results
Patients and baseline characteristics
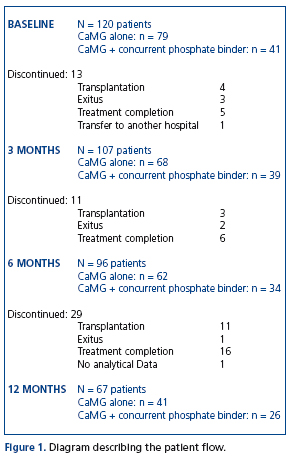
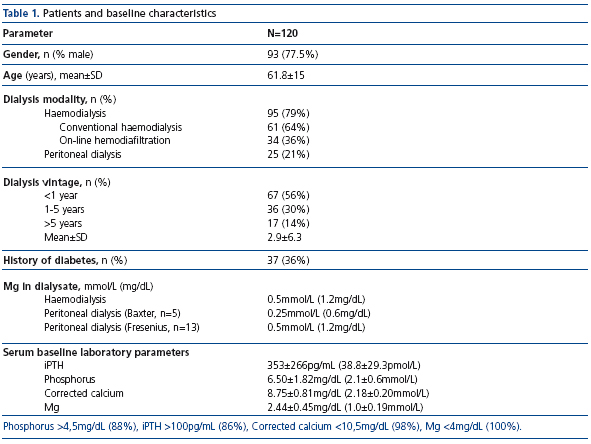
In total, 120 patients participated in the study. At database lock, one-year data were available from 67 patients (Figure 1). Patient demographics, magnesium concentration in dialysis fluids, and baseline laboratory values at initiation of CaMg therapy are shown in Table 1. Ninety-five patients received haemodialysis (61 conventional haemodialysis and 34 on-line hemodiafiltration), 25 patients were on peritoneal dialysis. Magnesium concentration in the dialysis fluid was 0.5mmol/L (1.2mg/dL) with the exception of 5 PD patients for whom a dialysis solution containing 0.25mmol/L (0.6mg/dL) (Baxter®) was used. 79 out of 120 patients received CaMg alone and 41 patients CaMg plus concurrent phosphate binder. In addition to phosphate binding agents, patients received calcitriol, paricalcitol and calcimimetics (Table 2). At baseline, calcium carbonate and lanthanum carbonate were administered most frequently (23% and 28%, respectively).
Phosphate binder dosage
Once CaMg was introduced, phosphate binders remained either unchanged, or were reduced or stopped during the observation period. No dose increases were observed during the course of the study, nor were new phosphate binding agents initiated. Before introducing CaMg, there was no washout period considered in clinical practice. At the end of month 12, mean (±SD) pill number of CaMg was 4.75±1.50. In the group CaMg alone (n:41), at the end of the month 12, mean (±SD) pill number of CaMg was 4.48±1.66.
Evolution of serum phosphorus levels over time
Significant reductions in serum phosphorus were achieved in both treatment groups (Figure 2). No differences were observed between the groups as to the degree of phosphate decrease. Serum phosphorus levels were similar between the groups at all time-points. At baseline, the majority of patients had serum phosphorus levels above the Kidney Disease Improving Global Outcome (KDIGO) and Kidney Disease Outcomes Quality Initiative (K/DOQI) recommended target of 4.5mg/dL (1.4mmol/L) and 5.5mg/dL (1.8mmol/L) respectively. During the course of the study the number of patients with phosphorus levels within the target range increased in both groups (Figure 3). The highest number of patients within the K/DOQI target range was achieved at 6 months with 78% of patients combined from both groups and at one year with 75%
Evolution of calcium over time
After initiation of CaMg, corrected calcium increased in both groups similarly (Figure 4). In the group receiving CaMg alone, increases were significant compared to baseline at all time-points, whereby in the group receiving CaMg in combination with other phosphate binders, the differences versus baseline reached statistical significance at month 6. Only three out of 120 patients had transient Ca levels mildly above the upper limit of normal (>10.4mg/dL, >2.6mmol/L).
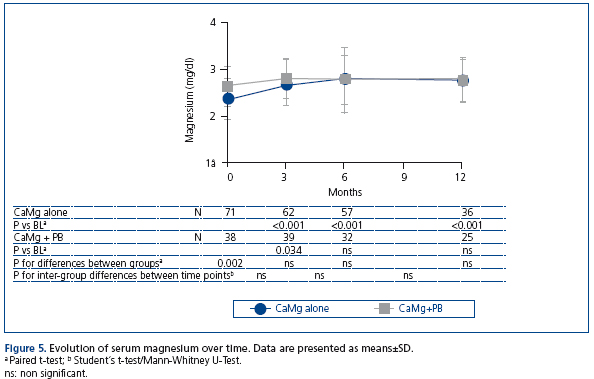
Evolution of magnesium over time
In patients receiving CaMg alone, magnesium levels increased significantly from 2.44±0.45mg/dL (1.00±0.19mmol/L) at baseline to 2.75±0.44mg/dL (1.13±0.18mmol/L) at the end of one year treatment (p<0.001). No significant increases were observed in the group receiving combination treatment (Figure 5). In this group, baseline levels were significantly higher compared to the monotherapeutic group (2.61±0.44mg/dL vs. 2.35±0.43mg/dL / 1.07±0.18mmol/L vs. 0.97±0.18mmol/L). Magnesium levels were similar in both groups during months 3 to 12. A total of 80 patients (67%) experienced episodes of mild hypermagnesaemia (>2.6mg/mL, 1.05mmol/L) during CaMg treatment. No cases of moderate hypermagnesaemia (4.8-8.6mg/dL, 2.0-3.6mmol/L) were observed.
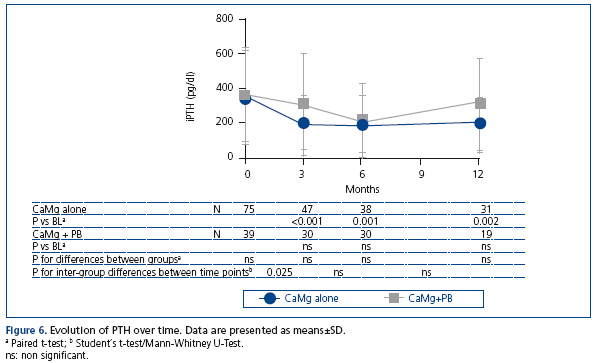
Evolution of PTH over time
PTH decreased in both groups over time (Figure 6), whereby PTH levels were generally lower at all time-points in the group receiving monotherapy. Here, decreases from baseline were significant at all time-points. In the group receiving CaMg in addition to other phosphate binders, the decrease was less pronounced, reaching significance only at month 6.
Evolution of albumin over time
Albumin levels increased slightly throughout the study (Figure 7). In monotherapy group the increase from baseline was statistically significant only in month 6. From month 3 on, albumin levels were significantly higher in the group receiving CaMg in addition to another phosphate binder.
Safety
In total, 53 patients (44.2%) discontinued CaMg due to transplantation (34%), exitus (11.3%), Transfer to another hospital (1.9%), treatment completion (50.9%) and no analytical data (1.9%). No treatment-related serious adverse events were reported during the course of the study.
Discussion
The present observational study explored the effect of CaMg on serum phosphorus levels over one year in dialysis patients under real practice conditions. CaMg treatment resulted in statistically significant reductions in serum phosphorus without affecting the control of related laboratory parameters such as magnesium, calcium, PTH and albumin. This effectiveness of CaMg in clinical practice is consistent with results observed in a randomised controlled clinical trial.16
The publication of K/DOQI clinical guidelines for Bone Metabolism and Disease in 200318 promoted significant progress in the issues related to mineral metabolism in CKD. Motivated by the growing evidence regarding the association of mineral metabolism disturbances and cardiovascular disease in CKD, the KDIGO workgroup recommended new targets for mineral metabolism related biochemical parameters.19 However, several observational studies have concluded that compliance with mineral metabolism targets in dialysis patients was poor. Al Aly et al. reported that CKD-MBD biochemical markers in the majority of dialysis patients fell outside K/DOQI targets, with only 40% having serum phosphorus within the recommended ranges.20 We have also previously shown in 1,312 patients undergoing haemodialysis at six Spanish centers that the prevalence of patients outside of K/DOQI targets for phosphorus was 46%.21 Serum phosphate levels are not controlled in >50% of HD patients, mainly because the effectiveness of any phosphate binder therapy is compromised by poor adherence due to high pill burden. At the end of one year of CaMg treatment, the mean number of tablets required to achieve phosphorus control was 4.75±1.50, which is lower than the number of CaMg tablets administered in the CALMAG study (7.3±3.03 tablets per day).16 Higher numbers of tablets have been reported in other studies using calcium salts.5,6,8 This reduction of pill burden might translate into better compliance which, in turn, leads to improved outcomes. Accordingly, in the present study, 78% of patients achieved the K/DOQI target range for serum phosphorus at the end of 6 months treatment with CaMg.
Phosphate binders have been used for many years, but the ideal binder in terms of efficacy, patient adherence, safety and costs has yet to be found. Phosphate binders containing aluminium or calcium have well-known drawbacks, such as aluminium toxicity or increased calcium load causing hypercalcaemia which has been associated with vascular calcification. Newer agents such as sevelamer- and lanthanum-based binders avoid calcium burden but are very expensive. Magnesium-containing phosphate binders offer a chance to circumvent some of the problems associated with other agents. In spite of good experience with the use of magnesium-based phosphate binders and excellent results,9-14 the impact of magnesium in CKD patients still remains unclear in clinical routine practice. The CALMAG study compared safety parameters of CaMg to sevelamer hydrochloride.16 Both regimens were equally well tolerated with a similar number of adverse events.
In our study we observed mild increases in serum magnesium from baseline at the end of one year treatment, but no symptomatic hypermagnesaemia was observed. Although a small increase in serum magnesium occurred in the CaMg group in the CALMAG study, the mean concentration was consistently well below potential symptomatic levels (<4.8mg/dL / 2.0mmol/L). In fact, this minor increase in serum magnesium may have beneficial effects. Various epidemiological studies demonstrated associations between low serum magnesium levels and an increased risk for metabolic syndrome, type 2 diabetes mellitus (T2DM), hypertension and atherosclerosis.22 However, the use of magnesium as a therapeutic agent is only indicated for pre-eclampsia and specific forms of arrhythmias. In a recent study by Khan et al. evaluating 3,530 participants, multivariable-adjusted models indicated that individuals in the lowest quartile of serum magnesium were ~50% more likely to develop atrial fibrillation.23 In this context, it is important to consider data from a recent meta-analysis reporting an 11.6% prevalence of atrial fibrillation in dialysis patients. The overall incidence of 2.7/100 patient-years was higher than in the general population and has been associated with an increased risk of stroke and mortality.24 Moreover, magnesium supplementation and mild hypermagnesaemia might have beneficial effects in CKD patients with regards to calcification and mortality.25 There is concern that increased serum magnesium levels may affect bone in dialysis patients dependent as well as independent of its PTH-lowering effect.26 In this context, we need more information about the bone effects of magnesium but preliminary data obtained from the CALMAG study shows that CaMg and sevelamer-HCl lower serum levels of iFGF-23 to a similar extent. Changes in bone parameters were dependent on characteristics of the respective phosphate binder; in contrast to sevelamer-HCl, CaMg had no impact on bone turnover markers. In fact the bone turnover parameters alkaline phosphatase, bone AP, procollagen type 1 amino-terminal propeptide 1 osteoprotegerin, beta-crosslaps and tartrate-resistant acid phosphatase 5b increased significantly in the sevelamer-HCl group and they remained almost unchanged in the CaMg group, after the initial phase of P lowering.27 Further studies would be needed that could give us information about the long-term effects of elevated levels of serum magnesium in patients on dialysis.
In our study, a dialysis magnesium concentration of 1.2mg/dL (0.5mmol/L) was used in more than 95% of patients. In dialysis patients, magnesium concentrations are dependent mainly on dialysate magnesium. Magnesium crosses the dialysis and peritoneal membrane readily. Investigation of various magnesium dialysate concentrations in patients undergoing haemodialysis or peritoneal dialysis showed that concentrations of 1.8mg/dL (0.75mmol/L) may cause mild hypermagnesaemia, whereas a concentration of 0.6mg/dL (0.25mmol/L) often leads to hypomagnesaemia. Results for the magnesium dialysate concentration of 1.2mg/dL (0.5mmol/L) are less consistent but magnesium levels were within the normal range in the majority of cases.28 The inconsistent results of several studies suggest that other factors, such as nutrition and magnesium supplements (e.g. antacids), may also play an important role in determining serum magnesium levels in dialysis patients.
Finally, magnesium containing phosphate binders are relatively inexpensive compared to sevelamer- or lanthanum-based agents. Calculated costs for the treatment with CaMg were about 80% lower compared to sevelamer hydrochloride.16,29 Nephrologists raised some concerns about the costs of introducing magnesium monitoring. That, however, would be offset by the lower cost of magnesium-containing phosphate binders over sevelamer or lanthanum. Furthermore, most modern multi-channel biochemistry analysers include the measurement of magnesium, if required, without major cost implications.30
A recent metanalisis included all randomised and non-randomised trials that compared outcomes between patients with chronic kidney disease taking calcium-based phosphate binders (with those taking non-calcium-based binders. The final interpretation is that non-calcium-based phosphate binders are associated with a decreased risk of all-cause mortality compared with calcium-based phosphate binders in patients with chronic kidney disease.31 In our opinion further studies are needed comparing Ca-Mg binders (with low amount of calcium and magnesium effect) with non calcium binders.
Potential limitations of this study are possible bias introduced by the real world of clinical practice. Physicians prescribe drugs based on their own decisions and the study does not include a washout period before starting CaMg treatment as in controlled clinical trials. The results from this study would have been strengthened if an active comparator or placebo arm had been included. But the present study was designed to assess whether results obtained from clinical trials of CaMg could be replicated in the clinical practice setting. Additional limitations include that dietary data were not collected in this study, primarily for the purpose of ensuring consistency of dietary phosphate intake. We did not precisely record tolerability, but efficacy and tolerability of CaMg have been well documented in previous studies.16
In order to determine the effect of co-medication on phosphorus control by CaMg, conduction of a multivariable analysis was intended. However, this approach was not feasible due to the non-interventional nature of this study. Subgroups with distinct allocation of calcium-containing and calcium-free phosphate binders were too small to yield significant results. Controlled, randomised clinical trials are required to explore the impact of co-medication on phosphorus control.
Conclusions
Magnesium-containing phosphate binders have certain intrinsic advantages, since their use avoids aluminium toxicity and allows calcium intake to be reduced or eliminated. The combination of calcium acetate and magnesium carbonate presents a phosphate binder, which is effective and well tolerated in clinical practice, and saves about 80% of treatment costs. Magnesium levels were only slightly elevated and there was minimal increase in total serum calcium. This analysis of current clinical practice shows that -consistent with findings from a randomised controlled trial- CaMg treatment provides marked improvement in serum levels of phosphorus helping patients in trying to achieve K/DOQI and KDIGO targets.
Conflict of interest
Authors declare potential Conflicts of Interest.
ALM de F speaker honoraria Amgen, Fresenius, Genzyme and Shire.
Referencias Bibliográficas
1. Block GA, Hulbert-Shearon TE, Levin NW, Port FK. Association of serum phosphorus and calcium x phosphate product with mortality risk in chronic hemodialysis patients: a national study. Am J Kidney Dis 1998;31:607-17. [ Links ]
2. Floege J, Kim J, Ireland E, Chazot C, Drueke T, de Francisco A, et al. Serum iPTH, calcium and phosphate, and the risk of mortality in a European haemodialysis population. Nephrol Dial Transplant 2011;26:1948-55. [ Links ]
3. Tonelli M, Pannu N, Manns B. Oral phosphate binders in patients with kidney failure. N Engl J Med 2010;362(14):1312-24. [ Links ]
4. Friedman EA. Calcium-based phosphate binders are appropriate in chronic renal failure. Clin J Am Soc Nephrol 2006;1:704-9. [ Links ]
5. Qunibi WY, Hootkins RE, McDowell LL, Meyer MS, Simon M, Garza RO, et al. Treatment of hyperphosphatemia in hemodialysis patients: The Calcium Acetate Renagel Evaluation (CARE Study). Kidney Int 2004;65:1914-26. [ Links ]
6. Qunibi WY, Moustafa M, Muenz LR, He DY, Kessler PD, Diaz-Buxo JA, et al. A 1-year randomized trial of calcium acetate versus sevelamer on progression of coronary artery calcification in hemodialysis patients with comparable lipid control: the Calcium Acetate Renagel Evaluation-2 (CARE-2) study. Am J Kidney Dis 2008;51:952-65. [ Links ]
7. Ring T, Nielsen C, Andersen SP, Behrens JK, Sodemann B, Kornerup HJ. Calcium acetate versus calcium carbonate as phosphorus binders in patients on chronic haemodialysis: a controlled study. Nephrol Dial Transplant 1993;8:341-6. [ Links ]
8. Schaefer K, Scheer J, Asmus G, Umlauf E, Hagemann J, von Herrath D. The treatment of uraemic hyperphosphataemia with calcium acetate and calcium carbonate: a comparative study. Nephrol Dial Transplant 1991;6:170-5. [ Links ]
9. Delmez JA, Tindira CA, Windus DW, Norwood KY, Giles KS, Nighswander TL, et al. Calcium acetate as a phosphorus binder in hemodialysis patients. J Am Soc Nephrol 1992;3:96-102. [ Links ]
10. Delmez JA, Kelber J, Norword KY, Giles KS, Slatopolsky E. Magnesium carbonate as a phosphorus binder: a prospective, controlled, crossover study. Kidney Int 1996;49:163-7. [ Links ]
11. Spiegel DM, Farmer B, Smits G, Chonchol M. Magnesium carbonate is an effective phosphate binder for chronic hemodialysis patients: a pilot study. J Ren Nutr 2007;17:416-22. [ Links ]
12. Spiegel DM. The role of magnesium binders in chronic kidney disease. Semin Dial 2007;20:333-6. [ Links ]
13. Spiegel DM, Farmer B. Long-term effects of magnesium carbonate on coronary artery calcification and bone mineral density in hemodialysis patients: A pilot study. Hemodial Int 2009;13:453-59. [ Links ]
14. Tzanakis IP, Papadaki AN, Wei M, Kagia S, Spadidakis VV, Kallivretakis NE, et al. Magnesium carbonate for phosphate control in patients on hemodialysis. A randomized controlled trial. Int Urol Nephrol 2008;40:193-201. [ Links ]
15. Deuber HJ. Long-term efficacy and safety of an oral phosphate binder containing both calcium acetate and magnesium carbonate in hemodialysis patients. Nieren-und Hochdruckkrankheiten 2004;33:403-8. [ Links ]
16. de Francisco AL, Leidig M, Covic AC, Ketteler M, Benedyk-Lorens E, Mircescu GM, et al. Evaluation of calcium acetate/magnesium carbonate as a phosphate binder compared with sevelamer hydrochloride in haemodialysis patients: a controlled randomized study (CALMAG study) assessing efficacy and tolerability. Nephrol Dial Transplant 2010;25:3707-17. [ Links ]
17. Ureña P, Jacobson SH, Zitt E, Vervloet M, Malberti F, Ashman N, et al. Cinacalcet and achievement of the NKF/K-DOQI recommended target values for bone and mineral metabolism in real-world clinical practice--the ECHO observational study. Nephrol Dial Transplant 2009;24(9):2852-9. [ Links ]
18. National Kidney Foundation. K/DOQI clinical practice guidelines for bone metabolism and disease in chronic kidney disease. Am J Kidney Dis 2003;42(Suppl 3):S1-S202. [ Links ]
19. Kidney Disease: Improving Global Outcomes (KDIGO) CKD-MBD Work Group. KDIGO clinical practice guideline for the diagnosis, evaluation, prevention, and treatment of Chronic Kidney Disease-Mineral and Bone Disorder (CKD-MBD). Kidney Int Suppl 2009;(113):S1-S130. [ Links ]
20. Al Aly Z, González EA, Martin KJ, Gellens ME. Achieving K/DOQI laboratory target values for bone and mineral metabolism: an uphill battle. Am J Nephrol 2004;24:422-6. [ Links ]
21. Lorenzo V, Martin-Malo A, Perez-Garcia R, Torregrosa JV, Vega N, de Francisco AL, et al. Prevalence, clinical correlates and therapy cost of mineral abnormalities among haemodialysis patients: a cross-sectional multicentre study. Nephrol Dial Transplant 2006;21(2):459-65. [ Links ]
22. Geiger H, Wanner C. Magnesium in disease. Clin Kidney J 2012;5(Suppl 1):i25-i38. [ Links ]
23. Khan AM, Lubitz SA, Sullivan LM, Sun JX, Levy D, Vasan RS, et al. Low serum magnesium and the development of atrial fibrillation in the community: the Framingham Heart Study. Circulation 2013;127(1):33-8. [ Links ]
24. Zimmerman D, Sood MM, Rigatto C, Holden RM, Hiremath S, Clase CM. Systematic review and meta-analysis of incidence, prevalence and outcomes of atrial fibrillation in patients on dialysis. Nephrol Dial Transplant 2012;27(10):3816-22. [ Links ]
25. Massy ZA, Drüeke TB. Magnesium and outcomes in patients with chronic kidney disease: focus on vascular calcification, atherosclerosis and survival. Clin Kidney J 2012;5(Suppl 1):i52-i61. [ Links ]
26. Navarro-Gonzalez JF, Mora-Fernandez C, Garcia-Perez J. Clinical implications of disordered magnesium homeostasis in chronic renal failure and dialysis. Semin Dial 2009;22:37-44. [ Links ]
27. Covic A, Passlick-Deetjen J, Kroczak M, Büschges-Seraphin B, Ghenu A, Ponce P, et al. A comparison of calcium acetate/magnesium carbonate and sevelamer-hydrochloride effects on fibroblast growth factor-23 and bone markers: post hoc evaluation from a controlled, randomized study. Nephrol Dial Transplant 2013;28(9):2383-92. [ Links ]
28. Cunningham J, Rodríguez M, Messa P. Magnesium in chronic kidney disease Stages 3 and 4 and in dialysis patients. Clin Kidney J 2012;5(Suppl 1):i39-i51. [ Links ]
29. Plagemann T, Prenzler A, Mittendorf T. Considerations about the effectiveness and cost effectiveness of therapies in the treatment of hyperphosphataemia. Health Econ Rev 2011;1(1):1. [ Links ]
30. De Francisco ALM, Rodriguez M. Magnesium - its role in CKD. Nefrologia 2013;33(3):389-99. [ Links ]
31. Jamal SA, Vandermeer B, Raggi P, Mendelssohn DC, Chatterley T, Dorgan M, et al. Effect of calcium-based versus non-calcium-based phosphate binders on mortality in patients with chronic kidney disease: an updated systematic review and meta-analysis. Lancet 2013;382(9900):1268-77. [ Links ]
![]() Correspondence:
Correspondence:
Ángel L.M. de Francisco
Servicio de Nefrología
Hospital Universitario Marqués de Valdecilla
Avda. Valdecilla, s/n,
39008, Santander
angelmartindefrancisco@gmail.com
Enviado a Revisar: 13 Mar. 2014
Aceptado el: 11 Jul. 2014