INTRODUCTION
There is emerging evidence that male obesity negatively impacts fertility 1) (2. In this respect, in a multivariate analysis conducted by Bener et al. 3, patients having a body mass index (BMI) > 30 were found to have a three-fold increased risk of infertility. In a more detailed way, Hammiche et al. 2 found sperm concentration and total motile sperm count in men of subfertile couples were detrimentally affected by a high central adiposity. Lastly, a systematic review of the literature also concluded overweight and obesity were associated with an increased prevalence of azoospermia or oligozoospermia 4.
Several hypotheses have been proposed to explain the pathological mechanisms underlying this association. Recent studies have found that excess adipose tissue has a negative impact on reproductive hormone levels such as testosterone 5. Furthermore, given that human spermatogenesis is highly sensitive to heat 6, raised gonadal heat resulting from increased scrotal adiposity might be associated with reduced sperm function and subfertility. Lastly, a relationship between obesity and increased sperm oxidative stress was also reported by Fariello et al. 7 .
Fortunately, a recent animal study has found that simple diet and exercise interventions can be used to reverse the damaging effects of obesity on sperm function 8.
However, to date, no intervention studies have been focused on reversing these deleterious effects by regular exercise, despite recent observational studies showed that physically active subjects seemed to have a more anabolic hormonal environment and a healthier semen production 9. The lack of information in the literature may be explained, at least in part, given that previous studies suggested a negative impact of some types of exercise on semen quality 10) (11. Fortunately, a recent study concluded that physical activity was not deleterious to testicular function in young healthy adults 12.
For the reasons already mentioned, it was hypothesized that aerobic training could improve semen quality. Accordingly, the main objective was to determine the effect of a short-term aerobic training program on semen quality and reproductive hormone profile in sedentary obese adults.
MATERIALS AND METHODS
PARTICIPANTS
Ninety adults with obesity volunteered for this longitudinal study from the community. Characteristics of participants at baseline are summarized in table I. All subjects met the following inclusion criteria: a) young adults (25-40 years); b) obese, defined as a BMI ≥ 30 kg/m2 as was suggested for reproductive endocrinology research 13; and c) medical approval after completing a pre-participation physical examination.
Table I Characteristics of sedentary obese adults enrolled in the intervention (n = 45) and control (n = 45) groups
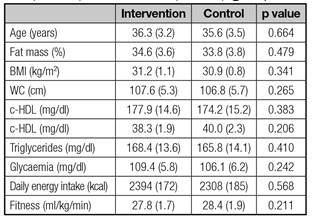
Results expressed as mean (SD). BMI: Body mass index; WC: Waist circumference; c-HDL: High density lipoprotein cholesterol; SBP: Systolic blood pressure; DBP: Diastolic blood pressure.
On the other hand, exclusion criteria were: a) participation in a training program in the six months prior to their participation in the trial; b) testicular varicocele and/or genital infection, leukocytospermia, chronic illness and serious systemic diseases; c) previous surgery (e.g., vasectomy reversal, varicocele removal, etc.); d) receiving medication and/or antioxidant consumption that may interfere with the redox homeostasis; and e) toxic habits (smoking and/or alcohol).
Participants were randomly allocated to the intervention (n = 45) or control group (n = 45) using a concealed method.
This research has been conducted in full accordance with ethical principles, including the World Medical Association Declaration of Helsinki (version 2002). Participants gave their written informed consent prior to study participation. Furthermore, this protocol was approved by an Institutional Ethics Committee.
INTERVENTION PROGRAM
Participants in the intervention group performed a 16-week aerobic training program in a treadmill, three sessions per week, consisting of a warm-up (10-15 minutes), 35-50 minutes treadmill exercise (increasing five minutes each four weeks) at a work intensity of 50-65% of peak heart rate (increasing a 5% each four weeks) measured during a maximal treadmill test, and cooling-down (5-10 minutes). In order to ensure that the training workload was appropriate, all participants from the intervention group wore a wireless wearable heart rate monitor (Sport Tester PE3000, Polar Electro, Kempele, Finland).
SEMEN ANALYSIS
Semen was collected after three days of ejaculation abstinence (verified by self-report on the day of sample collection) by manual masturbation into a sterile container on site and examined within 30 minutes of ejaculation. It should be pointed out that within this time it has been shown that sperm motility is stable 14. Semen quality assessment included semen volume (ml), sperm concentration (%), sperm motility (%) and normal morphologic features (%). All parameters were examined out according to the World Health Organization (WHO) guidelines 15 by trained laboratory technicians. A 1-ml sample was diluted (1:20) with formaldehyde to examine the sperm concentration by hemocytometer (Hauser Scientific Inc., Horsham, PA). The progressive sperm motility percentage was measured using the known volume of the specimen, which was placed onto a clean glass slide and covered by a coverslip, and examined under positive phase-contrast microscopy at a magnification of ×400 with the help of an ocular grid. For morphological classification, 100 spermatozoa were counted using a high-quality phase-contrast microscope (magnification ×600). Thin, well-spread smears were air-dried, fixed, and stained according to the Papanicolaou method. The classification, including head shape/size defects, neck and midpiece defects, tail defects, and cytoplasmic droplets, was based on the manual published by the WHO.
SERUM REPRODUCTIVE HORMONES
Blood samples (5 ml) were collected from the antecubital vein after a 12-h fast (08.00-09.00 a.m.). The whole blood was centrifuged at 3,000 rpm for ten minutes. The plasma was separated and stored at -80 °C until further analysis. Serum levels of follicle-stimulating hormone (FSH), luteinizing hormone (LH), estradiol and testosterone were determined by ELISA (Diagnostics Systems Laboratories Inc., Texas, USA).
Body composition and physical fitness
Regarding anthropometric assessment, fat free mass percentage was assessed by bioelectrical impedance analysis (MC-180, Tanita Ltd., UK). Participants were requested to not perform moderate or vigorous exercise for 24 hours prior to testing as well as to abstain from eating or drinking for two hours before testing. Moreover, they were asked to urinate immediately prior to data collection. The following equation was used to calculate the body mass index (BMI = weight [kg]/height [m]2) being expressed as kg/m2. Height was determined with an accuracy of 0.1 cm by precision stadiometer. Body weight was assessed with an accuracy of 0.1 kg using an electronic balance. Lastly, waist circumference (WC) was measured as halfway between the costal edge and the crista.
In order to determine physical fitness, all participants (n = 90) performed a continuous maximal incremental test, using the standard Bruce treadmill protocol, until exhaustion. Gas exchange data were collected throughout the test using a breath by breath metabolic system. The electrocardiograms (ECGs) were continuously recorded using a 12 lead stress analysis system. In this respect, the criteria used to determine the maximal oxygen consumption (VO2 max) was the maximal O2 value at plateau despite increasing workload (< 2 ml/kg/min increase in VO2 between progressive stages). Furthermore, it should be pointed out that all participants, including the control group, underwent a pre-training period to be familiarized with the correct use of the treadmill.
NUTRITIONAL INTAKE RECORD
To control the potential confounding effect of diet, participants included in intervention and control groups recorded their nutritional intake for seven days. Participants were instructed about completion of the nutritional protocol.
STATISTICAL ANALYSIS
The results of biochemical and anthropometric assessments were expressed as mean (SD). Given the limited sample size, the Shapiro-Wilk test was used to assess whether data were normally distributed. To compare the mean values, repeated one-way analysis of variance (ANOVA), with post hoc Bonferroni correction to account for multiple tests, were used. Regarding semen quality and reproductive hormone assessments, results were expressed as median (5-95th percentiles). The Mann-Whitney U test was used to compare differences between the intervention and control groups. Spearman's coefficient was used to identify correlations among tested parameters. The significance of the changes observed was ascertained to be p < 0.05.
RESULTS
Physical fitness, expressed as VO2 max, was significantly improved (27.8 ± 1.7 vs 30.6 ± 1.4 ml/kg/min; p = 0.0371) in the intervention group. Body composition was improved as fat mass percentage was significantly reduced (34.6 ± 3.2 vs 32.1 ± 3.0%; p = 0.0322). Similarly, WC was significantly decreased after being exercised (107.6 ± 5.3 vs 104.1 ± 4.7; p = 0.0438).
When compared to pre-test, sperm concentration (p = 0.032), progressive sperm motility (p = 0.021) and normal morphology percentages (p = 0.026) were significantly improved after the completion of the training program. On the other hand, no significant changes were observed in semen volume in the intervention group.
Regarding reproductive hormone profile, the ratio testosterone/estradiol was significantly improved (p = 0.002). In addition, it was found testosterone level was significantly increased (p = 0.037) after being exercised. These results are summarized in table II. Furthermore, negative significant correlations were found between WC and both sperm concentration (r = -0.32; p = 0.028) and progressive sperm motility (r = -0.29; p = 0.036) in the intervention group. Similar results were found between WC and testosterone levels (r = -0.38; p = 0.017) after being exercised.
Table II Semen quality and serum reproductive hormone profile in sedentary obese adults enrolled in the intervention (n = 45) group

Results expressed as median (5-95th percentiles). Volume: semen volume expressed as ml. Sperm concentration expressed as 106/ml. Progressive sperm motility and normal morphology expressed as percentages (%). FSH and LH expressed mIU/ml. Testosterone expressed as ng/ml. Estradiol expressed as pg/ml.
Lastly, no significant differences were found between the intervention and control groups when assessing energy intake (2,394 ± 172 vs 2,306 ± 188 kcal; p = 0.316) and mean daily vitamin intake (9.2 ± 1.8 vs 9.6 ± 2.1 mg/d vitamin E, p = 0.512; 79.7 ± 18.2 vs 81.4 ± 20.6 mg/d vitamin C, p = 0.431).
No significant changes in any of the tested parameters were found in the control group (Table III). Finally, it should be also pointed out that neither sports-related injuries nor withdrawals from the program were reported during the entire study period.
Table III Seminal outcomes and serum reproductive hormone profile in sedentary obese adults enrolled in the control group (n = 45)
Results expressed as median (5-95th percentiles). Volume: semen volume expressed as ml. Sperm concentration expressed as 106/ml. Progressive sperm motility and normal morphology expressed as percentages (%). FSH and LH expressed mIU/ml. Testosterone expressed as ng/ml. Estradiol expressed as pg/ml.
DISCUSSION
The most striking finding was that aerobic training improved semen quality in sedentary obese adults. Similarly, Gaskins et al. 16 demonstrated that higher physical activity and less TV watching were significantly associated with higher total sperm count and sperm concentration in young healthy men. However, it should be pointed out when designing a training protocol that, depending on sport modality and/or intensity, physical activity may impact in a positive or negative way on semen quality. In this respect, bicycling ≥ 5 h/week was associated with lower sperm concentration and motility 17. Likewise, long-term strenuous treadmill exercises (80% VO2 max) have a deleterious effect on reproduction 11. Lastly, physical exercise at high altitude was associated with a testicular dysfunction leading to a reduced sperm concentration in non-obese mountaineers 10.
Despite neither antioxidants nor markers of oxidative damage in seminal plasma were assessed in the present study, it may be hypothesized that the reduction of oxidative damage induced by aerobic training may explain, at least in part, the improvement of semen quality, mainly if we take into consideration a similar intervention program based on aerobic training increased antioxidant system and reduced oxidative damage in adults with type 2 diabetes mellitus 18. Similarly, Tartibian and Maleki 19 found that spermatozoa from recreationally active men may be less susceptible to oxidative stress-induced DNA damage and hence infertility.
Previous studies have concluded that the reduction in semen quality found in men with BMI > 25 kg/m2 could be explained, at least in part, by an increased serum estradiol level 20. The current study also showed reproductive hormone profile was improved after the completion of the training program. The significant reduction of fat mass after the completion of the training program may play a key role supporting this finding 21. Consistent with these results, aerobic training at moderate intensity for 24-weeks significantly increased testosterone level in abdominally obese adults 22. It should be emphasized that our intervention program lasted just 16 weeks so that it may be considered to be more feasible and practical for participants. Lastly, it should be noted that participants remained overweight or obese after the completion of the training program. This could explain why we did not observe a larger improvement in the reproductive hormonal profile, as was previously reported by Hakonsen et al. 21.
Another challenge of the present study was to identify significant associations between semen characteristics and anthropometric parameters in order to provide an easier, quicker, cheaper assessment of the seminal outcomes. In this line, negative significant correlations were found between WC and both sperm count and progressive motility, thus confirming that abdominal fat impairs semen quality 2) (23. Conversely, it should be pointed out that Eskandar et al. 24 did not find any significant correlation between BMI and semen quality parameters in men seeking fertility treatment. However, they did not assess any indices of central fat distribution, such as WC and/or hip circumference, to confirm their results. Similarly, a systematic review with meta-analysis revealed little evidence for a relationship with semen parameters and increased BMI 25. The latter authors also concluded the main limitation of this review was that data from most studies could not be aggregated for meta-analysis 25.
This study had some limitations and strengths. The major weakness was the relatively short duration of the exercise intervention, so that there was no follow-up to determine whether these positive effects induced by aerobic training were maintained. In addition, the short sample size may also limit the generalization of the results.
Strengths of the present study included the excellent adherence, rate suggesting the training program was effective and easy to follow up. Secondly, the majority of studies previously reported were originated from fertility clinics, where patient cohorts are frequently biased toward, sub-fertile men, which may also confound findings. Thirdly, some studies rely on self-reporting of parameters such as lifestyle factors and BMI, which can lead to under reporting 8. Lastly, given that spermatogenesis takes approximately 64 days 26, the follow-up period in the present study (112 days) should be able to detect any change induced by the intervention program.
Finally, it was concluded that: a) aerobic training improved semen quality in obese sedentary adults; and b) this finding could be explained, at least in part, by an improvement of the reproductive hormone profile after the completion of the intervention program.
There is a clear need for long-term, well-conducted studies to determine whether correction of semen quality induced by exercise improves the outcomes of live birth and pregnancy rate in obese adults.














