INTRODUCTION
Percutaneous endoscopic gastrostomy (PEG) is the gold standard approach for long-term enteral nutrition. Prolonged dysphagia is the main indication for PEG, which may be the consequence of neurologic or obstructive disease, the later most commonly associated with head or neck cancer 1,2.
Refeeding syndrome (RS) is a life-threatening condition defined as the potentially fatal shifts in fluids and electrolytes, resulting from hormonal and metabolic changes that may occur in malnourished patients receiving nutritional therapy 3. Its main clinical features include fluid-balance abnormalities, altered glucose metabolism, hypophosphatemia, hypomagnesemia, hypokalemia and vitamin deficiency. These features may induce heart failure, cardiac dysrhythmias, neuromuscular symptoms and hematologic dysfunction leading to death in a few weeks 4.
When dysphagic patients are referred for PEG, they often present with reduced food intake. Ingestion below 50% of daily needs is observed in most cases all over the weeks and established severe malnutrition is very common in this setting 5. According to the criteria of the National Institute for Health and Clinical Excellence (NICE), reduced body mass index (BMI), unintentional weight loss, little or no nutritional intake and low levels of phosphorus, magnesium and potassium are the main risk factors to develop RS 6 (Table 1).
Phosphorus is the most common intracellular anion, being essential for all cellular processes and the integrity of cell membranes. It is required for energy production in the form of adenosine triphosphate (ATP), regulates the oxygen affinity for hemoglobin and plays an important role in the renal acid-base buffer system 6. Actually, hypophosphatemia is the most typical hallmark of RS and is generally considered as severe when serum concentration is below 1.5 mg/dl 7,8. It may be present in 0.43% of hospitalized patients to 25% in specific subgroups such as cancer patients under nutritional support. Although not pathognomonic, hypophosphatemia is highly suggestive of RS 5.
Previous studies have already identified several predictors for adverse events after PEG as advanced age, male gender, low BMI and low serum albumin 9. A recent study published by our group assessed the prevalence of serum electrolyte abnormalities prior PEG and found that hyponatremia was associated with significantly shorter survival 10. Since this study, our team requires normal standard electrolytes (which in our hospital setting includes sodium, potassium and chloride) in order to perform endoscopic gastrostomy. Although teams taking care of PEG-fed patients should consider these aspects and systematically monitor serum electrolytes, except for our previous study, the real impact of reduced serum electrolytes as a risk for RS was never addressed in the literature 5.
The present study aims to: a) evaluate the changes in serum phosphorus and magnesium when patients underwent PEG; b) determine the mortality rate of PEG-fed patients after one week and one month of enteral nutrition; and c) determine if hypophosphatemia or hypomagnesemia, as risk markers of RS, are associated with early mortality in PEG-fed patients.
MATERIAL AND METHODS
STUDY DESIGN
A single center, observational and retrospective study was performed in a large hospital setting. This study was conducted in accordance with the principles of the Declaration of Helsinki.
PATIENTS
Patients aged 18 years old or older, referred to the Artificial Feeding Team (GENE) of the Gastroenterology Department of Hospital Garcia de Orta, submitted to PEG from 2003 to 2016 due to prolonged dysphagia and who have died using PEG were initially eligible. A retrospective analysis of a prospectively collected database was performed and the following data was collected for each patient: age, gender, clinical indication for PEG, BMI, mid-upper arm circumference (MUAC), serum albumin and transferrin, serum levels of the ions under study (phosphorus and magnesium) and dates of gastrostomy and death. Early mortality was defined as death occurring during the first month of PEG-feeding. Survival was recorded in months after PEG until death or in weeks for patients who died during the first month. All information present in the study database was obtained using the clinical files of the Artificial Feeding Team. Patients who did not present all the data needed for the study purpose were excluded.
All patients or their representatives have signed the informed consent for PEG. Patients underwent PEG after a 12-hour of fasting. Antithrombotic therapy was managed according to the most recent guidelines 11. Defects in coagulation were corrected prior to the procedure. Any patient received intravenous intensive fluid therapy in the 48-72 hours before the procedure. All patients had to be stable before PEG. Unstable patients were refused or postponed.
Two gastroenterologists performed all procedures with patients under deep sedation with propofol, midazolam, fentanyl, and/or droperidol. During the procedure, oxygen saturation, heart rate and electrocardiographic signs were monitored. The "pull" method was used in most cases. The "push" method with gastropexy was applied routinely in head and neck cancer patients after one previous case of ostomy metastasis diagnosed in our department. This approach avoids the passage of the tube through the mouth, pharynx and esophagus, allowing a safe procedure without risk of seeding cancer cells in the gastrostomy tract 12,13.
Nutritional Risk Screening 2002 14
NRS 2002 was performed in every patient as part of a systematic hospital routine.
ANTHROPOMETRIC EVALUATION
Anthropometric evaluation was performed before the gastrostomy procedure, according to the ISAK manual of the International Society for the Advancement of Kinanthropometry 15. The average of three consecutive measurements was recorded on the patients' file.
BMI was obtained in most patients using the equation weight/height2. If patients were unable to easily stand up for weight and height evaluation, BMI was estimated using the MUAC and the regression equations described by Powell-Tuck and Hennessy, which were previously used and proved to provide a reliable BMI estimation in PEG-fed patients 16. Each patient was classified according to age: having low weight if BMI < 18.5 kg/m2 for patients under 65 years or BMI < 22 kg/m2 for patients 65 years old or older 17.
2MUAC was measured in centimeters, using a flexible measuring tape wrapped around the mid upper arm, halfway between the olecranon and the acromion process.
LABORATORY EVALUATION
A blood sample was obtained just before the PEG procedure. Serum albumin, transferrin and electrolytes were measured as part of patient global nutritional evaluation. Values of albumin < 3.5 g/dl and transferrin < 200 mg/dl were considered as low values, suggestive of malnutrition and/or poor prognosis. Normal cut-off values for serum electrolytes were considered, according to the institutional protocol of our hospital laboratory: phosphorus (2.5-4.8 mg/dl) and magnesium (1.5-2.1 mg/dl).
STATISTICAL ANALYSIS
The statistical analysis was performed using the Statistical Package for Social Sciences (SPSS(r) Inc., Chicago, IL), version 21, and Microsoft Office Excel Professional 2013(r).
Continuous variables were expressed as mean ± standard deviation and categorical variables as percentage. The non-parametric Spearman's correlation test and the Mann-Whitney test were applied to compare variables without normal distribution and the parametric Student's t-test was used where variables followed a normal distribution. The Chi-square test (or alternatively, the Fisher's exact test when individuals in each category were less than five) was also used to compare nominal variables. All tests were performed at a 5% level of statistical significance.
RESULTS
PATIENTS
From all the adult patients that underwent PEG from 2003 to 2016, 197 presented clinical files with all the required data, and were included in the study: 137 men and 60 women, aged between 26 and 100 years (mean 68.6 ± 14.7 years) that died under PEG-feeding. One-hundred and twenty patients (62.5%) were below 65 years old. The main characteristics of the population are described in Table 2. The most frequent clinical indications for PEG were neurologic diseases (60.9%), including post-stroke persistent dysphagia, amyotrophic lateral sclerosis, dementia, among others disorders, and head or neck cancer (34.5%). A minority of patients underwent PEG due to other causes (4.6%).
NUTRITIONAL ASSESSMENT: NRS 2002, ANTHROPOMETRY AND SERUM PROTEINS
NRS 2002 was ≥ 3 points in all patients, signaling high nutritional risk.
BMI before PEG ranged from 13.2 to 37 kg/m2 (mean 21.2 ± 4.5 kg/m2). Eighty-eight patients (44.7%) presented low BMI, which was adjusted to age group.
Serum albumin and transferrin at the time of PEG were low in 99 (50.3%) and 141 (71.6%) patients, respectively. Mean albumin was 3.45 ± 0.6 g/dl and mean transferrin was 180.3 ± 45 mg/dl. Low serum albumin was associated with low serum transferrin (p < 0.01). Although albumin and transferrin are dependent of several factors, low serum proteins levels, together with the reduced ingestion, NRS 2002 and BMI suggest high prevalence of malnutrition from baseline in most patients eligible for gastrostomy feeding.
ELECTROLYTE STATUS
Serum phosphorus and magnesium were evaluated in all patients. At the time of gastrostomy, serum phosphorus was low in 13 patients (6.6%) and serum magnesium was low nine patients (4.6%). Only one patient (0.5%) presented both low serum phosphorus and magnesium. The number of patients who presented low serum phosphorus and/or magnesium was similar regardless of the clinical indication for PEG (p > 0.05). Unlike serum magnesium (p > 0.05), low serum phosphorus concentration was associated with low patient BMI (p = 0.02) and low serum albumin (p = 0.02) and transferrin (p = 0.02).
SURVIVAL
There were no major post-PEG adverse events and no death was attributable to a complication of the technique. All deaths were caused by the evolution of the underlying disorders. Survival after PEG ranged from less than one month to a maximum of 68 months (mean 13.7 ± 15.4 months). Overall survival tended to be lower in patients with low BMI and low serum proteins. Nevertheless, neither BMI, nor albumin or transferrin were significantly lower in patients who died in the first week or during the first month of PEG-feeding (p > 0.05). Early mortality was 17.8%, namely nine patients died during the first week (4.6%) and 25 died in the first month (13.2%) of PEG-feeding.
Potential impact of hypophosphatemia or hypomagnesemia in the outcome
Serum phosphorus concentration was positively correlated with overall survival (R = 0.24, p < 0.01). No statistically significant correlation was achieved for magnesium (p > 0.05).
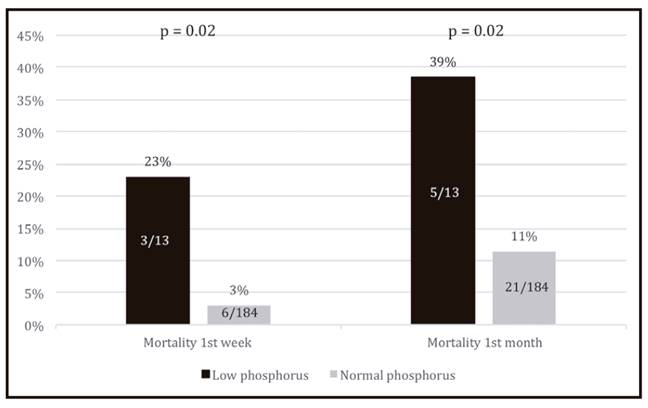
When compared with patients with normal serum phosphorus (three patients dying out of 13, 23%), hypophosphatemic patients present a significantly higher mortality (six patients dying out of 184, 3%) in the first week of PEG-feeding (p = 0.02). The impact of low phosphorus concentration also influences short-term survival beyond one week post-gastrostomy since an association between low serum phosphorus and higher mortality during the first month of PEG-feeding was also found (p = 0.02) (Fig. 1).
Considering the indication for PEG, no differences in survival relative to the frequency of low phosphorus concentration were found, in particular when the two main groups, head or neck cancer and neurologic disease, were compared.
Figure 2 represents Kaplan-Meier survival analysis according to the serum phosphorus levels.
DISCUSSION
Protein-energy malnutrition is a high prevalent condition in the gastroenterology clinical practice, especially when food intake is severely reduced for long periods 2,5,18. The risk of RS appears to increase with the duration of fasting and severity of malnutrition. Specific subgroups have been considered at increased risk such as patients with anorexia nervosa, chronic alcoholism, diabetes mellitus and cancer patients under radiation or chemotherapy 17. Actually, the incidence of RS is unknown, largely because of unclear definitions and diversity of its clinical features. There is no unequivocal consensus on how many of the typical abnormalities are required to diagnose RS. Hypophosphatemia is usually considered as a proxy marker but epidemiologic data differs according to the population studied 18,19.
Patients referred for endoscopic gastrostomy are usually at high risk of RS according to the NICE criteria. Our data showed that more than 50% of these patients presented low BMI and low serum proteins at the time of PEG, both associated with poor outcome. Food ingestion is considerably reduced. Malnutrition and systemic inflammation are highly prevalent in this setting.
RS is mainly characterized by serum electrolyte disturbance after starting nutritional therapy. It is caused by a serum insulin peak in previously fasted individuals leading to sudden intracellular shift of the serum ions to fulfil the depleted stores 20,21. In patients who already present low serum electrolytes, namely phosphorus and magnesium, the risk of RS increases and clinical manifestations may become more evident and potentially severe 5. To the best of our knowledge, no previous studies have assessed the prevalence of RS in patients undergoing PEG neither its potential impact in survival.
During the last years, serum phosphorus and magnesium have been determined before PEG in a large number of our patients. Our data showed that hypophosphatemia and hypomagnesemia were present in 4-7% of patients. A previous study of our Artificial Feeding Team found higher prevalence of electrolyte disturbances in this population; nevertheless, mean survival was markedly lower, which can be attributed to more severe disease and poor baseline health condition due to sample selection bias 9. Since this study, standard electrolytes are a prerequisite before PEG, including potassium, an electrolyte associated to the RS. So, in the present study we evaluate phosphorus and magnesium, the other electrolytes related to the RS. In our experience, serum phosphorus was associated with BMI and serum proteins. Low phosphorus was more prevalent in the more severely malnourished patients undergoing PEG, signaling an association between hypophosphatemia and worse clinical and nutritional status.
RS usually develops soon after restarting nutrition and may increase short-term mortality 5. Our results revealed that almost 5% and 13% of PEG-fed patients died in the first week or in the first month after the procedure, respectively. Mortality in the first week may be attributed to RS or alternatively, be a consequence of the high degree of malnutrition, inflammation or individual comorbidities. Early deaths may also be associated with procedural complications, but there were no records of major post-PEG adverse events in our sample. Low BMI and serum proteins are generally considered as markers of poor prognosis, but in our sample, there was no significant association with early mortality. On the other hand, a significant association between hypophosphatemia and mortality in the first week after PEG was detected. Since hypophosphatemia may be an important risk factor for RS and mortality in the first week potential attributed to RS, the excess of hypophosphatemic patients dying during the first week may signify RS deaths. Mortality until the end of the first month was also higher in patients with hypophosphatemia before PEG, suggesting that the risk of death associated with RS may persist after the first PEG-feeding days. The same effect on patient short-term outcome was not observed for magnesium.
Although RS might be highly prevalent in patients undergoing PEG considering the high number of patients with established underfeeding and malnutrition before starting enteral nutrition, this study shows that early mortality which could be attributed to hypophosphatemia and hypomagnesemia, frequently used markers of RS, is low. In our hospital, most endoscopic gastrostomies are performed in an outpatient setting and patients are discharged a few hours after the procedure. Patients and their caregivers are instructed to provide reduced enteral food quantities before the first outpatient appointment, usually scheduled to the fourth day after the procedure. Nevertheless, they do not always comply with the recommendations of starting PEG-feeding with hypocaloric regimens, by fear of exacerbating malnutrition. Even so, RS may be less common than initially expected. One possible explanation could be the loss of absorptive capacity due to small bowel mucosa atrophy induced by severe malnutrition, which could prevent complete nutrient digestion/absorption in the early post-PEG period. This hypothesis may explain less RS cases than initially expected and justifies further studies to evaluate the intestinal mucosa in these patients.
The present study displays some limitations. First, it is a retrospective study carried out in a single hospital and collected data was dependent on patient clinical files, which are not always complete. However, data was collected prospectively and our double registration in paper and informatics support helps to avoid missing information. Second, the exact pathophysiological mechanisms of hypophosphatemia and hypomagnesemia were not assessed and comorbidities that could indirectly affect electrolyte balance were not explored. Finally, our data do not allow to be sure that early mortality was definitely caused by RS rather than other causes. Nevertheless, the increased early mortality associated with low serum phosphorus is noteworthy.
This study provides teams taking care of PEG patients with a simple biochemical marker of poor outcome and reduced short-term survival. Efforts to prevent RS must always be performed in these patients and hydroelectrolytic balance routinely achieved before starting artificial nutrition. Even in outpatients, when electrolyte disturbances are present in the day of the gastrostomy procedure, our team is now correcting the identified disorders before discharge, and nutritional support is introduced much slower and more closely monitored.
CONCLUSION
Theoretically, most patients undergoing PEG have a high-risk of RS according to the NICE criteria. Established malnutrition and low serum electrolytes are known risk factors to develop this life-threatening condition. The prevalence of hypophosphatemia and hypomagnesemia at the moment of the gastrostomy procedure may reach 6%. Hypophosphatemia is significantly correlated with poor nutritional status. Mortality during the first week of PEG-feeding is low and seems not to be associated with low BMI and serum proteins. An excess of early mortality may be attributed to hypophosphatemia and RS. Hypophosphatemia is associated with mortality during the first week after PEG and this effect tends to be extended to the first PEG-feeding month. Hypophosphatemia should be viewed as a risk marker of RS-related death in PEG-fed patients.