Contribution to scientific literature
It is known that off-label antineoplastic drugs represent 6.7-33.2% of all prescriptions in patients with cancer in Western countries and are mainly used in patients with advanced cancer and there is some concern about their actual effectiveness and safety. In our context, their use is less frequent than the one described in literature; they were mostly used in metastatic tumours, with relatively little effectiveness and the patient’s performance status must be taken into account to select the patients to be treated in order to optimise their effectiveness and safety.
Introduction
Antineoplastic drugs, like all medicines, require authorization from the pertinent regulatory agency to be commercialised. Such authorization allows oncologists to prescribe them once their efficacy and safety have been assessed favourably. It is possible to use a drug beyond therapeutic indications or conditions of use approved in the summary of product characteristics. If there are preliminary data on the efficacy of an antineoplastic agent but the drug is still under clinical research, the patient may access it through participation in a clinical trial, by means of compassionate use or expanded access1. On other occasions, there is no therapeutic alternative for a specific patient and the oncologist suggests the use of medicines in conditions different from those authorised in the data sheet2. Finally, antineoplastic drugs authorised in a nearby country or which will be commercialised soon in Spain, may also be prescribed as foreign medicines3. These three cases were regulated by Royal Decree 1,015/2,0094, but there is a fourth case: the medicine is authorised in Spain, but the Pharmacy and Therapeutics Committee of the pertinent centre has not authorised its use within the local environment.
Little is known about the health outcomes of the use of oncological treatments in special situations. Their exceptional nature and the absence of authorised therapeutic alternatives increase the uncertainty of whether their use is actually beneficial for patients. Since the palliative systemic treatment authorised within the context of metastatic disease in patients with adult solid tumours is frequently ineffective, the decision to use off-label antineoplastic agents is always controversial and difficult to make. Most studies on the off-label use of drugs only analyse frequency of use and drug type or they show different positions from the different groups involved, among which there are usually conflicts (consultants, doctors, pharmacists, pharmaceutical industry, and patients)5-10.
The purpose of this work is to retrospectively analyze the health outcomes in a group of oncological patients treated with off-label antineoplastic drugs at a single centre, describing the type of special use situation, response rate, survival, and toxicity.
Method
It is a descriptive, observational, retrospective, single-centre study performed with the cooperation of the Department of Medical Oncology and the Pharmacy Unit, of all consecutive cases in which antineoplastic drugs were administered in special use situations in the Department of Medical Oncology at a university hospital in the last ten years.
The subjects of this study were patients with any type of tumour and in any stage to whom antineoplastic medicines were administered in special use conditions in the last ten years. Special situations included the compassionate use of investigational drugs, the use of medicines in conditions different from those authorised in the summary of product characteristics (off-label due to prescriptions, route of administration, dosage, or administration frequency different from those approved), the use of foreign medicines, which medicines will be commercialised in Spain but are already authorised in a nearby country and, finally, the use of medicines approved in Spain but not included in the hospital’s pharmacotherapeutic guide.
Patients could be men and women. They had to be of legal age and must have been evaluated by the Pharmacy Committee with a positive assessment of the application for antineoplastic medicines in a special use situation and treated in the Department of Medical Oncology of the centre between 1 January 2,005 and 31 December 2,014.
Patients from the Pharmacy Unit’s database were identified. Data related to patients and the treatment administered were collected retrospectively from the Medical Oncology case histories (up to year 2,010 they were stated on paper and from year 2.010 they were digital). Access to digital histories was immediate and paper histories were requested from the centre’s documentation service. A database was created with SPSS version 15, where the variables to be analysed were transcribed.
Independent variables related to patients were age, sex, performance status measured by the ECOG scale11, comorbidity measured by the Charlson index12, type of cancer according to the third edition of the International Classification of Diseases for Oncology13, and tumour stage pursuant to the AJCC (American Joint Committee on Cancer) classification14. Independent variables related to treatment were treatment type (chemotherapy, hormone therapy, biological therapy, and immunotherapy), drug name, treatment purpose (adjuvant, neoadjuvant, palliative), treatment line, number of cycles (when treatment was oral and daily, a month of treatment was deemed a cycle), dose reduction, dose delay, type of special situation, and prescribing oncologist.
Dependent variables analysed were survival, response to treatment, and toxic effects. Overall survival (OS) was defined as the time period running from commencement of treatment in a special situation to death due to any cause or last contact with patient. Disease-free survival (DFS) was defined as the time period running from commencement of treatment to relapse (adjuvant context of early-onset disease). Progression-free survival (PFS) was calculated measuring the time between commencement of treatment and progression of the tumoural disease (palliative context of metastatic disease). Duration of response was the time period from the moment response to treatment is demonstrated to progression. They were measured in months. To know patients’ vital status, the National Death Index, Ministry of Health and Social Affairs, was consulted. The response to treatment according to changes in size was classified in complete, partial, stable, or progression following the RECIST 1.1 requirements15. Toxicity was classified into categories according to pathophysiology and anatomy, and severity, following the Common Terminology Criteria for Adverse Events V3.0 (CTCAE)16. Only serious toxicity was collected (grades 3 and 4).
A descriptive analysis of variables was performed. For the comparison of qualitative variables, the chi-square test was applied, as corrected by Fisher, and for quantitative variables, the t-test was used. For the calculation of survival, the Kaplan Meyer method17 was applied and the Log-Rank test18 was employed to compare survival curves. A multivariate analysis was further performed through the Cox method (proportional hazard model) for survival. The best predictive model was reached for the inclusion or exclusion of variables of interest. For the statistical data analysis, SPSS version 15 was used. In the statistical analysis, p<0.05 was considered to indicate statistical significance.
The study was approved by the Ethics Committee of the University Hospital Puerta del Mar of Cádiz and has been performed in accordance with the ethical standards as laid down in the 1,964 Declaration of Helsinki and its later amendments or comparable ethical standards. For this type of study formal consent is not required.
Registered on the website of the Spanish Agency for Medicines and Health Products with JBC-ABI-2016-01 code.
Results
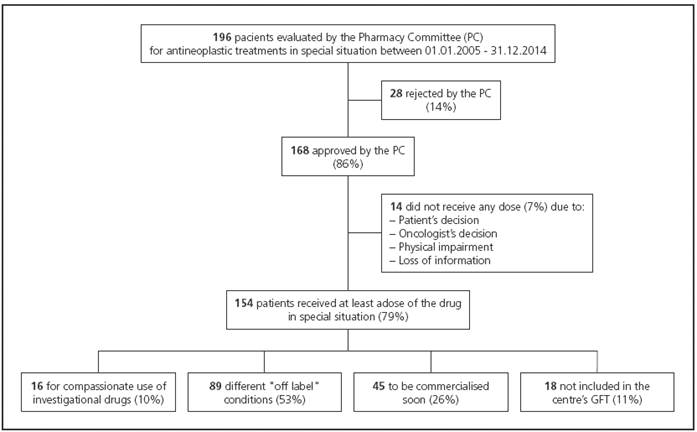
196 case histories of patients evaluated by the Pharmacy Committee after application for Medical Oncology service were studied. Out of the 196 applications, 168 (86%) were approved and 154 patients (92%) received at least a dose of medicine in special situations. Figure 1 describes the study flow chart. The median follow-up was 11 months (range 0-116 months).
The features of the 168 patients having an application approved by the Pharmacy Committee are shown in Table 1. Features of treatments are stated in Table 2.
Table 1 Features of 168 patients with an application approved by the Pharmacy Committee

*Measured by the ECOG scale (Eastern Cooperative Oncology Group)(11)
** Measured by the Charlson scale12
Only 8 patients received off-label medicines drugs as adjuvant treatment. They were 8 women treated with trastuzumab in 2,005. Two of them recurred, one of them after 55 months and the other one, after 71 months. One of them died after 77 months of cancer.
In 160 patients with stage IV or recurrent cancer, a subjective response (patient’s improved symptoms) was observed in 52 patients (32.5%). As regards the objective response rate, there were 3 complete responses (1.9%) and 14 partial responses (8.8%), with an overall response rate of 10.7%. Other 25 patients (15.6%) showed a stable disease and in 62 cases (38.8%) the response was deemed progression. Response was not assessable in 56 patients (35.1%). In responding patients the median duration of response was 2.5 months (range 1-17 months).
The median progression-free survival in 92 assessable patients was 5 months (range: 1 - 92) and the median overall survival in 138 assessable patients was 11 months (4-92). Figure 2 represents the progression-free survival and overall survival curves.
In the univariate analysis, the performance status variable measured by the ECOG scale influenced on overall survival, patients with higher functional impairment (ECOG 2 and 3) having worse survival in comparison to patients with better performance status (ECOG 0 and 1) (hazard ratio -HR- 0.41; confidence interval -CI- 95% 0.25-0.68. P= 0.001). Patients who received off-label drug in the first or second line also enjoyed higher survival than patients who received them in subsequent lines (HR 0.64; CI 95% 0.49-0.93. P= 0.02). Patients who received less cycles of off-label drugs (1 to 2 cycles) had shorter survival than those who received more cycles (3 or more); HR was 3.67 (CI 95% 2.51-5.37. P=0.0001). Finally, patients with partial response (HR 0.23; CI 95% 0.11-0.45.
P= 0.0001), stabilization (HR 0.17; CI 95% 0.09-0.32. P= 0.0001), or progression (HR 0.44; CI 95% 0.28-0.69. P= 0.0001) enjoyed better survival than patients not assessable for response (Table 3). In the multivariate model, age being an adjustment variable, the performance status (HR 0.36; CI 95% 0.17-0.77. P= 0.009) and the number of cycles (HR 3.66; CI 95% 2.08-6.44. P= 0.0001) influenced independently on overall survival.
Grades 3 and 4 toxic effects in 139 patients assessable for safety are stated in Table 4. The most frequent grade 3 and 4 toxicity were asthenia (19%), neutropenia (10.7%), and nausea and vomiting (8.9%).
Discussion
The most-represented tumours in our study were central nervous system, followed by colorectal, head and neck, and breast, more frequently treated with biological therapy followed by chemotherapy. Most of them (53%) received treatment in off-label (different condition) special situations between the first and third lines of treatment (67.8%), receiving a median of 4 cycles, and dose delays, reduction, and treatment interruptions being frequent; 8% did not receive any dose. They were mostly used for palliative purposes in metastatic or recurrent tumours, with little effectiveness (it being still less if the patient’s functional status was impaired) and with toxic effects within the frequency and intensity to be expected in the daily medical care context of cancer treatment. In fact, there is final evidence that although palliative chemotherapy is used to improve quality of life in patients with advanced cancer, its use does not improve quality of life near death in patients with regular or poor functional status and it worsens quality of life in patients with a good functional status19.
Use of antineoplastic drugs in special use situations is widespread although there is variability in frequency of indication. Our experience is far from others in the Western world where off-label prescriptions represent 6.7-33.2% of all prescriptions2,10,20. In our case, they involve 2.5% of all antineoplastic treatment indications only (around 800 patients per year are treated with intravenous or oral antineoplastics at our centre, out of which only a median of 20 patients per year are in special situations).
Our study coincides with others in that most off-label prescriptions in patients with cancer were indicated for palliative purposes, in some cases related to clinical benefit and in others, not2. There is some discussion on which parameters-to-be-analysed must be employed to suggest a drug for regulatory approval within the context of metastatic cancer treatment. Overall survival is the optimal goal, the response rate and progression-free survival being admitted, with some controversy, as surrogate markers of overall survival21. Our study, with an objective response rate of 10.7%, is far from 30% deemed to be the limit to consider an antineoplastic agent sufficient activity(22). The progression-free survival rate of 5 mon-ths and overall survival rate of 11 months in our study do not actually provide us with positive or negative information on effectiveness, since it is an heterogeneous group of patients as regards neoplasms and treatment, but they do provide us with an idea about its overall outcome when a patient with advanced cancer is proposed an off-label treatment. At least we may assert that most indicated treatments are not being administered in the last three months of life, life expectancy being considered, as a general rule, not to indicate active treatments and to limit the therapeutic effort. Actually, there are only two main reasons to administer an active treatment in most patients with advanced cancer: to help them live more and/or live better 23. Our study may not assure the former since it is not a comparative study and in relation to the latter, authors have serious doubts, since the subjective benefit observed in one-third of them may be seriously compromised by toxic effects.
Awareness of medicines safety is higher and higher and there is evidence that off-label prescriptions are frequently inappropriate and expose patients to a higher risk of adverse effects24,25. The safety problem in our study has been limited. Oncologists are trained to handle drugs with not contemptible toxicities and side effects reported in our study are within the frequency and intensity to be expected in the daily health care context of cancer treatment. The most frequently detected adverse effect in the study was asthenia. Almost 20% of patients suffered from it. It is a toxic effect that significantly impacts on patients’ quality of life and limits the possible palliative benefit of off-label antineoplastic drugs, given that it is difficult to control 26. Other frequent side effects, the neutropenic fever and digestive toxicity in the form of nauseas and vomiting, affected around 10% of cases and also had a significant impact on patients’ quality of life.
There is little information on factors related to the doctor prescribing off-label medicines. In a study, it was demonstrated that age and attitude to medicine based on scientific evidence, not sex, influenced on the number of off-label prescribed drugs 27. Our study differentiated outcomes pursuant to the prescribing oncologist, but we may not draw conclusions since each oncologist was specialised in a specific tumour pathology and the differences detected are mainly due to these different neoplasms treated.
Possible explanations for the use of off-label drugs in the clinical practice are the different types of tumours, the difficulty to carry out or access clinical trials, the frequent use of drugs in different conditions or users’ demanding attitude. The oncologist is frequently responsible for finishing the active treatment of a patient with advanced cancer who has received multiple lines of treatment unsuccessfully. Limitation of the therapeutic effort is performed by making shared and informed decisions considering the patients’ and relatives’ values. The question is: Why does the oncologist continue suggesting treatments, frequently ineffective or slightly effective, instead of limiting the therapeutic effort? It is a difficult question, but it may be related to the demanding attitude of patients and relatives who, immersed in a medicalised society that offers, in an unreal way, solutions to everything and gives an optimistic vision of the disease, demand a solution where there is not one. Furthermore, it is also necessary to recognise that limitations on communication skills of sanitary professionals may, in part, facilitate the option of a new treatment when the ideal would be to suggest the limitation of the therapeutic effort28.
The use of off-label antineoplastic drugs does not necessarily mean the absence of scientific evidence on it1. Moreover, a medicine frequently has very promising predictions of benefiting patients or it has already demonstrated its beneficial effect in clinical trials, but regulatory authorities have not yet approved it. In such situation, the oncologist has the moral duty to suggest it as a therapeutic alternative, since patients cannot wait for months or years. The opposed situation does also occur. That is, a medicine has demonstrated being beneficial in clinical trials but such benefit is lost when it is applied to the general population. This occurs because clinical trials test medicines in an ideal situation, with very selected participants, with no comorbidities and with optimal functional statuses. The real world is different and it conditions the loss of medicines’ effectiveness29. For instance, in our study no patient with melanoma responded to the drug ipilimumab, when in clinical trials it demonstrated significant response rates with an impact on such patients’ survival. Something similar has been described by other authors30.
Although much more experience is required to guide the use of off-label antineoplastic drugs, our study does know the health outcomes in these patients in the real world. Such information may be useful to the oncologist in order to decide, together with the patient, the off-label prescription. The authors of this study think that if said information is useful, much more useful is to know that the performance status measured by the ECOG scale predicts the benefit for the patient.
A limitation of the study is related to the analysis of health outcomes in so heterogeneous patients given that they had multiple types of tumours and they were treated with multiple and different types of antineoplastics. That is true, but what is also true is that they all had been treated with a off-label drug and, despite the aforementioned factors, it is very valuable to know such health outcomes. Like all retrospective studies, the classification bias because the study’s variables may not be measured correctly due to missing information or because information not well collected in the patients’ case histories is lost, is also evident.
To conclude, it is known that off-label antineoplastic drugs represent 6.7-33.2% of all prescriptions in patients with cancer in Western countries and are mainly used in patients with advanced cancer and there is some concern about their actual effectiveness and safety. In our context, their use is less frequent than the one described in literature; they were mostly used in metastatic tumours, with relatively little effectiveness and the patient’s performance status must be taken into account to select the patients to be treated in order to optimise their effectiveness and safety.