Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Nutrición Hospitalaria
versión On-line ISSN 1699-5198versión impresa ISSN 0212-1611
Nutr. Hosp. vol.25 no.2 Madrid mar./abr. 2010
Effects of telmisartan vs olmesartan on metabolic parameters, insulin resistance and adipocytokines in hypertensive obese patients
Efectos de telmisartan vs olmesartan sobre parámetros antropométricos, resistencia a la insulina y adipocitoquinas en pacientes hipertensos obesos
D. A. de Luis, R. Conde, M. González-Sagrado, R. Aller, O. Izaola, A. Dueñas, J. L. Pérez Castrillón and E. Romero
Institute of Endocrinology and Nutrition. Medicine School and Unit of Investigation. Hospital Río Hortega. RD-056/0013 RETICEF. University of Valladolid. Valladolid. Spain.
ABSTRACT
Background: Angiotensin II regulates the production of adipokines. The objective was to study the effect of treatment with telmisartan versus olmesartan in hypertensiveobese and overweight patients.
Subjects: A sample of 65 overweight and obese patients with mild to moderate hypertension was analyzed in a prospective way with a randomized trial. Patients were randomized to telmisartan (80 mg/day) or olmesartan (40 mg/day) for 3 months. Weight, body mass index, blood pressure, basal glucose, insulin, total cholesterol, LDLcholesterol, HDL-cholesterol, triglycerides, HOMA, QUICKI, leptin and adiponectin were determined at basal time and after 3 months of treatment.
Results: Sixty five patients gave informed consent and were enrolled in the study. Patients treated with telmisartan had a significative decrease of glucose 10.53 mg/dl (CI 95%: 2.6-18.5), insulin 2.51 mUI/L (CI 95%: 2.07-7.17) and HOMA 1.08 (CI 95%: 0.39-2.55). Patients treated with olmesartan had a significative decrease of total cholesterol 20.2 mg/dl (CI 95%: 5.8-34.9) and LDL cholesterol 22.6 mg/dl (CI 95%: 9.7-35.6). Only leptin levels have a significant decrease in telmisartan group 7.39 ng/ml (CI 95%: 1.47-13.31).
Conclusion: Telmisartan improved blood pressure, glucose, insulin, HOMA and leptin in hypertensive diabetic patients. Olmesartan improved blood pressure and lipid levels.
Key words: Adiponectin. Hypertension. Insulin resistance. Leptin. Olmesartan. Telmisartan.
RESUMEN
Introducción: La angiotensina II puede regular la producción de adipocitoquinas. El objetivo de nuestro trabajo fue evalaur el efecto sobre parámetros bioquímicos del tratamiento con telmisartan versus olmesartan en pacientes obesos hipertensos.
Pacientes: Se analizó una muestra de 65 pacientes con hipertensión moderada severa y obesidad, mediante un ensayo clínico randomizado. Los pacientes fueron randomizados en dos ramas; telmisartan (80 mg/día) u olmesartan (40 mg/día) durante 3 meses. Se determinaron en el tiempo basal y tras 3 meses los siguientes parámetros; peso, índice de masa corporal, presión arterial, glucosa, insulina, colesterol total, LDL-colesterol, HDL-colesterol,triglicéridos, HOMA, QUICKI, leptina y adiponectina.
Resultados: Los pacientes que recibieron telmisartan tuvieron una disminución significativa de los niveles de glucosa 10,53 mg/dl (CI 95%: 2,6-18,5), insulina 2,51 mUI/L (CI 95%: 2,07-7,17) y HOMA 1,08 (CI 95%: 0,39-2,55). Los pacientes tratados con olmesartan presentaron una disminución significativa de colesterol 20,2 mg/dl (CI 95%: 5,8-34,9) y LDL colesterol 22,6 mg/dl (CI 95%: 9,7-35,6). Solo, los niveles de leptina disminuyeron de manera significativa con telmisartan 7,39 ng/ml (CI 95%: 1,47-13,31).
Conclusion: Telmisartan mejora los niveles de presión arterial, glucosa, insulina, HOMA y leptina en pacientes hipertensos obesos. Olmesartan mejoró los niveles de presión arterial y el perfil lipídico.
Palabras clave: Adiponectina. Hipertensión. Resistencia a la insulina. Leptina. Olmesartan. Telmisartan.
Introduction
Obesity and insulin resistance are associated with cardiovascular risk factors, including altered levels of adipocytokines.1 Epidemiologic evidence of this rising tide of obesity and associated pathologies has led, in the last years, to a dramatic increase of research on the role of adipose tissue as an active participant in controlling pathologic processes.2,3.
The current view of adipose tissue is that of an active secretory organ, sending out and responding to signals that modulate appetite, insulin sensitivity, energy expenditure, inflammation and immunity. Adipocytokines are proteins produced mainly by adipocytes.4 These molecules have been shown to be involved in the pathogenesis of the metabolic syndrome and cardiovascular disease. Adiponectin is an adipocyte-derived collagen like protein identified through an extensive search of adipose tissue. Hypoadiponectinemia increased risk of coronary artery disease together with the presence of multiple risk factors, indicating that adiponectin is a key factor of the metabolic syndrome.5 Leptin is a 16 KDa protein secreted primarily from adipocytes. Recent reports suggest that leptin contributes to atheroscleoris and cardiovascular disease in obese patients.6 Insulin resistance and hyperinsulinemia are characteristics findings of this metabolic syndrome (MetS) and are very common in patients with essential hypertension.7.
Circulating angiotensin II, the active product of the renin-angiotensin system, is a hormonal regulator of cardiovascular function and electrolyte metabolism. Angiotensin II is also produced by local reninangiotensin systems in many organs including adipose tissue.8 In addition, angiotensin II regulates the production of adipokines. Angiotensin II increases the expression and the release of pro-inflammatory cytokines,9 increases leptin ob gene expression and secretion,10 and reduces plasma levels and gene expression of adiponectin, and insulin-sensitizing, anti-inflammatory adipokine.11 In turn, blockade of the renin-angiotensin system with inhibitors of angiotensin II formation or angiotensin II AT1 receptor blockers decreases body weight, improves insulin-sensitivity and prevents development of insulin resistance.12 Telmisartan and olmesartan are two antagonists of angiotensin II receptors used as antihypertensive drugs.
To clarify the effect of angiotensin II system blockade on adipocytokines, we studied the effect of treatment with telmisartan versus olmesartan in a randomized clinical trial in hypertensive obese and overweight patients.
Subjects and methods
Subjects
A sample of 65 obese and overweight patients with mild to moderate hypertension was analyzed in a prospective way with an open-randomized trial. We used WHO/ISH13 definitions for hypertension defined as systolic and diastolic blood pressure > 140 or > 90 mmHg, respectively. These patients were studied in an Endocrinology Unit and written informed consent was obtained. The study has been approved by the local ethics committee. Exclusion criteria included a history of cardiovascular disease or stroke during the previous 36 months, total cholesterol > 300 mg/dl, triglycerides > 400 mg/dl, blood pressure > 140/90 mmHg, the use of sulfonylurea, metformine, acarbose, thiazolidinediones, insulin, glucocorticoids, antineoplasic agents, angiotensin-converting enzyme inhibitors, psychoactive medications, drinking and/or smoking habit.
Procedure and calculations
Patients were randomized to telmisartan (80 mg/day) or olmesartan (40 mg/day) for 2 months. Weight, body mass index, blood pressure, basal glucose, insulin, total cholesterol, LDL-cholesterol, HDLcholesterol, triglycerides, leptin and adiponectin levels were measured at basal time and after 3 months of treatment.
Body weight was measured to an accuracy of 0.1 kg and body mass index (BMI) was calculated as follows: BMI = body weight (kg)/(body height (m))2.
The homeostasis model assessment for insulin sensitivity (HOMA) was calculated as follows: HOMA = (glucose x insulin)/22.5.14 Quantitative Insulin-Sensitivity Check index (QUICKI), a surrogate index of insulin sensitivity, was calculated as follows: QUICKI= 1/(log (insulin)+ log (glucose)).15
Blood pressure was measured twice after a 10 minutes rest with a random zero mercury sphygmomanometer, and averaged.
Assays
Serum total cholesterol and triglyceride concentrations were determined by enzymatic colorimetric assay (Technicon Instruments, Ltd., New York, N.Y., USA), while HDL cholesterol was determined enzymatically in the supernatant after precipitation of other lipoproteins with dextran sulphate-magnesium. LDL cholesterol was calculated using Friedewald formula. Plasma glucose levels were determined by using an automated glucose oxidase method (Hitachi 917, Roche Diagnostics, Mannheim, Germany). Insulin was measured by enzymatic colorimetry (Insulin, WAKO Pure-Chemical Industries, Osaka, Japan).
Adipocytokines
Leptin was measured by ELISA (Diagnostic Systems Laboratories, Inc., Texas, USA) with a sensitivity of 0.05 ng/ml and a normal range of 10-100 ng/ml. Adiponectin was measured by ELISA (R&D systems, Inc., Mineapolis, USA) with a sensitivity of 0.246 ng/ml and a normal range of 865-21424 ng/ml. Ratio adiponectin/leptin levels were calculated.
Statistical analysis
A power calculation based on weight improvement was performed. Thirty patients in each group were necessary to detect a change of 6 ng/dl in leptin levels, with an error type I < 0.05 and a statistical power of 80%.
The results were expressed as average ± standard deviation. The distribution of variables was analyzed with Kolmogorov-Smirnov test. Quantitative variables with normal distribution were analyzed with a twotailed, paired Student's-t test and ANOVA test. Nonparametric variables were analyzed with the Friedman and Wilcoxon tests. Qualita tive variables were analyzed with the chi-square test, with Yates correction as necessary, and Fisher's test. A p-value under 0.05 was considered statistically significant.
Results
Sixty five patients gave informed consent and were enrolled in the study. Baseline characteristics of patients were presented in table I, without statistical differences.
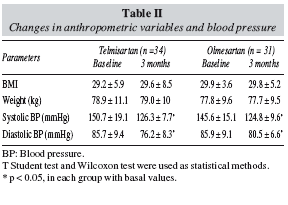
Table II shows a significantly decrease in systolic and diastolic blood pressures without changes in weight, with both treatments.
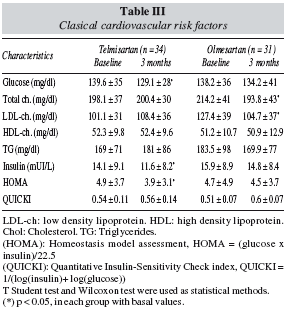
Table III shows the differences in classic cardiovascular risk factors. Patients treated with telmisartan had a significantly decrease of glucose 10.53 mg/dl (CI 95%: 2.6-18.5), insulin 2.51 mUI/L (CI 95%: 2.07-7.17) and HOMA 1.08 (CI 95%: 0.39-2.55). Patients treated with olmesartan had a significantly decrease of total cholesterol 20.2 mg/dl (CI 95%: 5.8-34.9) and LDL cholesterol 22.6 mg/dl (CI 95%: 9.7-35.6).
Table IV shows differences between basal and after treatment levels of adipocytokines. Only leptin levels have a significant decrease in telmisartan group 7.39 ng/ml (CI 95%: 1.47-13.31).
Discussion
The major finding of this study was that telmisartan 80 mg per day significantly improved insulin, HOMA, glucose and leptin levels. However, olmesartan improved lipid levels. Both drugs had the same beneficial effect on blood pressure levels.
Recently, Furuhashi et al.16 showed that blockade of the renin-angiotensin system by angiotensin-converting enzyme inhibitor (ACEI) and/or angiotensin II receptor blocker (ARB) decreased adipocyte size with improvement in insulin sensitivity. This previous data may partially explain our results with telmisartan. Other study17 suggests that other ARB (candesartan)-induced decrease in plasma insulin level might be induced an increase in plasma adiponectin in patients with renal dysfunction. In agreement with these results, the blockades of renin-angiotensin system are reported to decrease plasma insulin level and to increase plasma adiponectin level in patients without renal dysfunction, too.18 Our study did not show modification in levels of adiponectin, but telmisartan decrease leptin levels.
Recently, telmisartan displays the ability to act as partial agonist of PPARgamma,19 this stimulation induces the differentiation of pre-adipocytes to mature adipocytes, increases the subcutaneous fat and reduces the visceral fat related with insulin resistance. Several are the mechanisms through to explain the increased insulin sensitivity induced by blockade this system. They are represented by 1) vasodilatation, which increases the blood flow in skeletal muscle,20 2) inhibition of the impairment of insulin signaling induced by angiotensin II,21 decrease of tumor necrosis factor (TNF-alpha in skeletal muscle,22 increase in the ratio of insulin-sensitive type 1 fiber in muscle fiber composition. 23 In summary, the effect of ARB on adiponectin levels may be mediated by the decrease in insulin levels, which is due to the effect of ARB on enhancing insulin sensitivity24. Moreover, RAS blockade may stimulate phosphatidylinositol-3-kinase activity, which regulates insulin-stimulated adiponectin exocytosis.25
In our study, two characteristics differentiated telmisartan from olmesartan 1) telmisartan showed a reduction of blood pressure and leptin levels while olmesartan did not decrease leptin and 2) a greater impact of telmisartan on the glucose control.
Physiological increase in plasma leptin has been shown to significantly inhibit glucose-stimulated insulin secretion in vivo and to determine insulin resistance.26 Serum leptin concentrations reflects the total amount of fat present in the body, and lower plasma leptin levels have been reported consistently among weight losing patients.27 The results of our study show that significant reduction in leptin levels after telmisartan treatment occur when weight is unchanged. The decrease of leptin levels, which may reflect a drug induced improvement of leptin sensitivity, may play a role, indirectly or directly, in the induction of diabetes control. Telmisartan improved glucose tolerance and it can be due to a possible insulin-independent mechanism, possibly acting through an enhanced uptake and utilization of glucose by tissues mediated by leptin without changes in insulin sensitivity measured as HOMA (homeostasis model assessment).
As above-mentioned, there is a general consensus that angiotensin II has a trophic role in adipose tissue. However the effects of angiotensin II on adipocyte metabolism and differentiation are not conclusive, while others show that angiotensin II promotes it. Leptin gene expression is under the control of PPARgamma. PPARgama represses the expression of leptin ob gene.28 In animal models, angiotensin II AT1 receptor blockers enhanced insulin sensitivity and improved the serum lipid profile in obese.29.
In summary, the administration of telmisartan improved blood pressure, glucose, insulin, HOMA and leptin in hypertensive obese patients. Olmesartan improved blood pressure and lipid levels. These results suggest that telmisartan could be more useful in preventing atherosclerosis in these patients than olmesartan.
Referencias
1. Fantuzzi G. Adipose tissue, adipokines, and inflammation. J Allergy Clin Immunol 2005; 115: 911-9. [ Links ]
2. Meier U, Gressner AM. Endocrine regulation of energy metabolism: review of pathobiochemical and clinical chemical aspects of leptin, ghrelin, adiponectin and resistin. Clin Chemistry 2004; 50: 1511-25. [ Links ]
3. De Luis DA, M Gonzalez Sagrado, R Conde, R Aller, O Izaola. Circulating adipocytokines in obese non diabetic patients, relation with cardiovascular risk factors, anthropometry and resting energy expenditure. Ann Nutr Metab 2007; 51: 374-8. [ Links ]
4. Matsuda M, Shimomura I, Sata M. Role of adiponectin in preventing vascular stenosis. The missing link of adipo-vascular a-xis. J Biol Chem 2002; 277: 37487-91. [ Links ]
5. Kumada M, Kihara S, Sumitsuji S. Association of hypoadiponectinemia with coronary artery disease in men. Arterioscler Thromb Vasc Biol 2003; 23: 85-9. [ Links ]
6. Shimomoura I, Hammer RE, Ikemoto S. Leptin reverses insulin resitance and diabetes mellitus in mice with congenital lipodystrophy. Nature 1999; 401: 73-6. [ Links ]
7. Hirose H, Saito I, Kawabe H, Satura T. Insulin resistance and hypertension: seven year follow up study in middle-aged Japanese men (the KEIO study). Hypertens Res 2003; 26: 780-95. [ Links ]
8. Engeli S, Schling P, Gorzelniak K, Boschmann M, Janke J, Ailhaud G et al. The adipose-tissue renin-angiotensin-aldosterone system: role in the metabolic syndrome? Int J Biochem Cell Biol 2003; 35: 807-25. [ Links ]
9. Skurk T, Van Harmelen V, Haumer H. Angiotensin II stimulates the release of interleukin-6 and interleukin-8 from culturesd human adipocytes by activation of NF-kB. Arterioscler Thromb Vasc Biol 2004; 24: 1199-203. [ Links ]
10. Cassis LA, English VL, Bharadwaj K, Boustany CM. Differential effects of local versus systemic angiotensin II in the regulation of leptin release from adipocytes. Endocrinology 2004; 145: 169-74. [ Links ]
11. Hattori Y, Akimoto K, Gross SS, Hattori S, Kasai K. Angiotensin II induced oxidative stress elicits hypoadiponectinemia in rats. Diabetologia 2005; 48: 1066-74. [ Links ]
12. Jandeleit-Dahm KA, Tikellis C, Reid CM, Johston CI, Cooper ME. Why blockade of rennin-angiotensin system reduces the incidence of new-onset diabetes. J Hypertens 2005; 23: 463-73. [ Links ]
13. World Health Organization-International Society of Hypertension guidelines for the management of hypertension. J Hypertens 1999; 17: 151-83. [ Links ]
14. Mathews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher Df. Homesotasis model assessment: insulin resistance and beta cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985; 28: 412-4. [ Links ]
15. Katz A, Nambi SS, Mather K. Quantitative insulin sensitivity check index: a simple, accurate method for assessing insulin sensitivityin humans. J Clin Endocrinol Metab 2000; 85: 2402-5. [ Links ]
16. Furuhashi M, Ura N, Takizawa H, Yoshida D, Moniwa N, Murakami H et al. Blockade of the rennin-angiotensin system decrease adipocyte size with improvement in insulin sensitivity. J Hypertens 2004; 22: 1977-82. [ Links ]
17. Furuya R, Odamaki M, Kugamai H, Hishida A. Impact of angiotensin II receptor blocker on plasma levels of adiponectin and advanced oxidation protein products in peritoneal dialysis patients. Blood Purif2006; 24: 445-50. [ Links ]
18. Yenicesu M, Yilmaz MI, Caglar K, Sonmez A, Eyleten T, Acikel C, Kilic S, Bingol N et al. Blockade of the rennin-angiotensin system increases plasma adiponectin levels in type-2 patients with proteinuria. Nephron Clin Pract 2005; 99: c115-c121. [ Links ]
19. Benson C, Perdhadsingh HA, Ho CI. Identification of telmisartan as a unique angiotensin II receptor antagonist with selective PPARgamma modulating activity. Hypertension 2004; 43: 993-1002. [ Links ]
20. Kodama J, Katayama S, Tanaka K, Itabashi A, Kawazu S, Ishii J. Effect of captoprol on glucose concentration. Possible role of augmented postprandial forearm blood flow. Diabetes Care 1999; 13: 1101-11. [ Links ]
21. Folli F, Saad MJ, Velloso L. Crosstalk between insulin and angiotensin II signaling systems. Exp CLin Endocrinol Diabetes 1999; 107: 133-9. [ Links ]
22. Togashi N, Ura N, Higashiura K, Murakami H, Simamoto K. The contribution of skeletal muscle tumor necrosis factor to insulin resistance and hypertension in fructose fed rats. J Hypertens 2000; 18: 1605-10. [ Links ]
23. Higashiura K, Ura N, Takada T. The effect of an angiotensinconverting enzyme inhibitor and an angiotensin II receptor antagonists on insulin resistance in fructose-fed rats. Am J Hypertens 2000; 13: 290-7. [ Links ]
24. Negro R, Formoso G, Hassan H. The effects of irbesartan and telmisartan on metabolic parameters and blood pressure in obese, insulin resistant, hypertensive patients. J Endocrinol Invest 2006; 957-61. [ Links ]
25. Bogan JS, Lodish HF. Two compaments for insulin-stimulated exocytosis in 3T3-L1 adipocytes defined by endocgenous ACRP30 and GLUT4. J Cell Biol 1999: 146: 609-20. [ Links ]
26. Cases JA, Gabrielly I, Ma XH. Physiological increase in plasma leptin markedly inhibits insulin secretion in vivo. Diabetes 2001; 50: 348-52. [ Links ]
27. Schschols AM, Creutzberg EC, Buurman WA. Plasma leptin is related to proinflammatory status and dietary intake in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med 1999; 160: 1220-6. [ Links ]
28. Hollenberg AN, Susulic VS, Madura JP, Zhang B, Moller DE. Tontonoz P et al. Functional anyagonism between CCAAT/enhancer binding protein-alpha and peroxisome proliferatorsactivated receptor-gamma on the leptin promoter. J Biol Chem 1997; 272: 5283-90. [ Links ]
29. Ran J, Hirano T, Adach M. AngiotensinII type 1 receptor blocker ameliorates overprocution and accumulation of triglycerides in the liver of the Zucker fatty rats. Am J Physiol Endocrinol Metab 2004; 287: 227-32. [ Links ]
![]() Correspondence:
Correspondence:
D. A. de Luis.
Professor Associated of Nutrition.
Executive Director of Institute of Endocrinology and Nutrition.
Medicine School. Valladolid University.
Los Perales, 16.
47130 Simancas, Valladolid. Spain.
E-mail: dadluis@yahoo.es
Recibido: 15-X-2009.
Aceptado: 26-X-2009.