Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Nutrición Hospitalaria
versión On-line ISSN 1699-5198versión impresa ISSN 0212-1611
Nutr. Hosp. vol.26 no.4 Madrid jul./ago. 2011
Allelic frequency of G380A polymorphism of tumor necrosis factor alpha gene and relation with cardiovascular risk factors and adipocytokines in obese patients
Frecuencia alélica del polimorfismo G380A del factor de necrosis tumoral alpha y relación con factores de riesgo cardiovascular y adipocitoquinas en pacientes obesos
D. A. De Luis, R. Aller, O. Izaola, M. González Sagrado, R. Conde, B. de la Fuente and H. F. Ovalle
Center of Investigation of Endocrinology and Nutrition. Medicine School and Unit of Investigation. Hospital Rio Hortega. University of Valladolid. Valladolid. Spain.
ABSTRACT
Background: The aim of our study was to investigate the allelic frequency of the G308A polymorphism in the TNF alpha gene and the influence of G308A this polymorphism on cardiovascular risk factors and adipokine levels in obese patients.
Design: A population of 834 obesity patients was analyzed. A nutritional evaluation and a blood analysis were performed. The statistical analysis was performed for the combined G308A and A308A as mutant group and type G308G as wild group.
Results: A total of 630 patients (181 males/449 females) (75.5%) had the genotype G308/G308 (wild genotype group) with an average age of 43.5 ± 14.8 years, 188 patients (61 males/127 females) (22.5%) had the genotype G308/A308 (mutant genotype group-heterozygote) and 16 patients (5 males/11 females) (1.9%) with an average age of 44.5 ± 14.2 years had the genotype A308/A308 (mutant group-homorozygote) with an average age of 44.3 ± 11.4 years, without statistical differences in the mean age or sex distribution. Genotypes G308/A308 and A308/A308 was designed (mutant genotype group) as a dominant model. Allelic frequency of the A substitucion -308 was 13.19%. Anthropometric, adipokines, insulin resistance, lipid levels ad dietary intake were similar in both genotypes.
Conclusion: In conclusion, allelic frequency of G308A polymorphism is is in accordance with allelic frequencies observed in other populations. Carries of A308 allele have the same anthropometric and metabolic profile than wild type carriers.
Key words: Adipokines. Cardiovascular risk factors. G308A TNFalpha polymorphism. Obesity.
RESUMEN
Antecedentes: El objetivo de nuestro estudio fue investigar la frecuencia alélica del polimorfismo G308A del gen TNF alfa y su influencia en los factores de riesgo cardiovascular y los niveles de adipocinas en pacientes obesos.
Diseño: Se estudió una población de 834 pacientes obesos. Se realizaron una evaluación nutricional y un análisis de sangre. El análisis estadístico se realizó para el genotipo combinado G308A y A308A como grupo de mutantes y G308G tipo de grupo salvaje.
Resultados: Un total de 630 pacientes (181 varones/449 mujeres) (75,5%) tenían el genotipo G308/G308 (grupo con genotipo salvaje) con una edad media de 43,5 ± 14,8 años, 188 pacientes (61 varones/127 mujeres) (22,5%) con una edad media de 44,5 ± 14,2 años tuvieron el G308/A308 genotipo (grupo de mutantes genotipo heterocigoto) y 16 pacientes (5 varones/11 mujeres) (1,9%) tuvieron la A308/A308 genotipo (mutante grupo homorozygote) con una edad media de 44,3 ± 11,4 años, sin encontrar diferencias en la edad media o la distribución por sexo. La frecuencia alelica de la substitucion A-308 fue 13,19%. Las variables antropométricas, adipoquinas, resistencia a la insulina, perfil lípidico y la ingesta dietética fueron similares en ambos genotipos.
Conclusión: En conclusión, la frecuencia alélica del polimorfismo G308A está de acuerdo con las frecuencias alélicas observadas en otras poblaciones. Los obesos portadores del alelo A308 tienen los mismos perfiles antropométricos y metabólicos que los pacientes obesos con el genotipo salvaje.
Palabras clave: Adipoquinas. Factores de riesgo cardiovascular. Polimorfismo G380A. Obesidad.
Introduction
Some evidences have linked tumor necrosis factor alpha (TNF alpha) to the metabolic abnormalities of obesity and adipose tissue has been shown to be a site for TNF-α synthesis, with a direct correlation between adipokines, adipose tissue, TNF-alpha and insulin levels.1
Mutation analysis has identified a G- > A transition in the promoter region of TNF-alpha gene (-308), this polymorphism has been shown to affect the promoter region of the TNF-alpha gene leading to a higher rate of transcription compared to the wild allele.2 Association studies have been conducted on the G-308 variant, with conflicting results. One study3 has reported a significant association between the G-308A variant and insulin resistance, body mass index and leptin levels. Nevertheless, other studies have not reported correlation between TNF alpha mutation and insulin resistance.4
Adipose tissue is considered an endocrine organ, sending out and responding to signals that modulate appetite, insulin sensitivity and inflammation. Adipocy-tokines (leptin, adiponectin, resistin, IL-6, TNF alpha) are proteins produced mainly by adipocytes.5 These molecules have been shown to be involved in the pathogenesis of insulin resistance and the metabolic syndrome. Some reports suggest that leptin contributes to atheroscleoris and cardiovascular disease in obese patients.6 Hypoadiponectinemia increased risk of coronary artery disease, indicating that adiponectin is a key factor of the metabolic syndrome.7 TNF alpha and interleukin 6 are increased in most animal and humans models with obesity and insulin resistance.8 The role of resistin in linking human obesity with type 2 diabetes mellitus is thus questionable.9
The aim of our study was to investigate the allelic frequency of the G308A polymorphism in the TNF alpha gene and the influence of G308A this polymorphism on cardiovascular risk factors and adipokine levels in obese patients.
Subjects and methods
Subjects
A sample of 834 obese patients (body mass index > 30) was enrolled in a cross-sectional survey. These patients were studied in a Nutrition Clinic Unit and signed an informed consent. Exclusion criteria included history of cardiovascular disease or stroke during the previous 24 months, total cholesterol > 300 mg/dl, triglycerides > 400 mg/dl, blood pressure > 140/90 mmHg, fasting plasma glucose >110 mg/dl, as well as the use of sulphonilurea, thiazolidinedionas, insulin, glucocorti-coids, antineoplasic agents, agiotensin receptor blocker, angiotensin converting enzyme inhibitors and psy-choactive medications.
Procedure
Weight, blood pressure, basal glucose, c-reactive protein (CRP), insulin, total cholesterol, LDL-cholesterol, HDL-cholesterol, triglycerides and adypoci-tokines (leptin, adiponectin, resistin, TNF alpha, and interleukin 6) levels were measured at basal time. Genotype of G308A gene polymorphism was studied.
Genotyping of G308A gene polymorphism
Oligonucleotide primers and probes were designed with the Beacon Designer 4.0 (Premier Biosoft International®, LA, CA). The polymerase chain reaction (PCR) was carried out with 50 ng of genomic DNA, 0.5 uL of each oligonucleotide primer (primer forward: 5'-CTG TCT GGA AGT TAG AAG GAA AC-3'; primer reverse: 5'-TGT GTG TAG GAC CCT GGA G-3'), and 0.25 uL of each probes (wild probe: 5'-Fam-AAC CCC GTC CTC ATG CCC-Tamra-3') and (mutant probe: 5'-Hex-ACC CCG TCT TCA TGC CCC-Tamra -3') in a 25 uL final volume (Termociclador iCycler IQ (Bio-Rad®), Hercules, CA). DNA was denaturized at 95oC for 3 min; this was followed by 50 cycles of denaturation at 95oC for 15 s, and annealing at 59.3o for 45 s). The PCR were run in a 25 uL final volume containing 12.5 uL of IQTM Supermix (Bio-Rad®, Hercules, CA) with hot start Taq DNA polymerase.
Biochemical assays
Serum total cholesterol and triglyceride concentrations were determined by enzymatic colorimetric assay (Technicon Instruments, Ltd., New York, N.Y., USA), while HDL cholesterol was determined enzymatically in the supernatant after precipitation of other lipoproteins with dextran sulphate-magnesium. LDL cholesterol was calculated using Friedewald formula.
Plasma glucose levels were determined by using an automated glucose oxidase method (Glucose analyser 2, Beckman Instruments, Fullerton, California). Insulin was measured by enzymatic colorimetry (Insulin, WAKO Pure-Chemical Industries, Osaka, Japan) and the homeostasis model assessment for insulin sensitivity (HOMA) was calculated using these values.9
Creactive protein and adipocytokines
CRP was measured by immunoturbimetry (Roche Diagnostics GmbH, Mannheim, Germany), with a normal range of (0-7 mg/dl) and analytical sensivity 0.5 mg/dl.
Leptin was measured by ELISA (Diagnostic Systems Laboratories, Inc., Texas, USA) with a sensitivity of 0.05 ng/ml and a normal range of 10-100 ng/ml. Resistin was measured by ELISA (Biovendor Laboratory, Inc., Brno, Czech Republic) with a sensitivity of 0.2 ng/ml with a normal range of 4-12 ng/ml. Adiponectin was measured by ELISA (R & D systems, Inc., Mineapolis, USA) with a sensitivity of 0.246 ng/ml and a normal range of 8.65-21.43 ng/ml. Interleukin 6 and TNF alpha were measured by ELISA (R & D systems, Inc., Mineapolis, USA) with a sensitivity of 0.7 pg/ml and 0.5 pg/ml, respectively. Normal values of IL6 was (1.12-12.5 pg/ml) and TNFalpha (0.5-15.6 pg/ml).
Blood pressure and anthropometric measurements
Blood pressure was measured twice after a 10 minutes rest with a random zero mercury sphygmo-manometer, and averaged. Waist (narrowest diameter between xiphoid process and iliac crest) and hip (widest diameter over greater trochanters) circumferences to derive waist-to hip ratio (WHR) were measured. Body weight was measured to an accuracy of 0.1 kg and body mass index calculated as body weight/ (height2). Tetrapolar body electrical bioimpedance was used to determine body composition11
Dietary assessment
Patients received prospective serial assessment of nutritional intake with 3 days written food records (including a weekend day). Records were analyzed with a computer-based data evaluation system.12 Regular aerobic physical activity (walking was allowed, no other exercises) was maintained during the period study (3 times per week).
Statistical analysis
Sample size was calculated to detect differences over 4 kg in weight loss with 90% power and 5% significance (n = 700). The results were expressed as average ± standard deviation. The distribution of variables was analyzed with Kolmogorov-Smirnov test. Parametric variables were analyzed with a two-tailed, paired Student's-t test. Non-parametric variables were analyzed with the W-Wilcoxon test and U Mann Whitney test. Qualitative variables were analyzed with the chi-square test, with Yates correction as necessary, and Fisher's test. The statistical analysis was performed for the combined G308/A308 and A308/A308 as a mutant group and wild type G308/G308 as second group (dominant model). A p-value under 0.05 was considered statistically significant.
Results
Eight hundred and thirty four obese subjects gave informed consent and were enrolled. This sample has a mean age (44.1 ± 14.2 years and the mean BMI 36.5 ± 6.2, with 247 males (29.6%) and 587 females (70.4%).
A total of 630 patients (181 males/449 females) (75.5%) had the genotype G308/G308 (wild genotype group) with an average age of 43.5 ± 14.8 years, 188 patients (61 males/127 females) (22.5%) had the genotype G308/A308 (mutant genotype group-heterozygote) and 16 patients (5 males/11 females) (1.9%) with an average age of 44.5 ± 14.2 years had the genotype A308/A308 (mutant group-homorozygote) with an average age of 44.3 ± 11.4 years, without statistical differences in the mean age or sex distribution. Genotypes G308/A308 and A308/A308 was designed (mutant genotype group)as a dominant model. Allelic frequency of the A substitucion -308 was 13.19%. The observed genotype frequencies in our sample were in Hardt-Weinberg equilibrium.
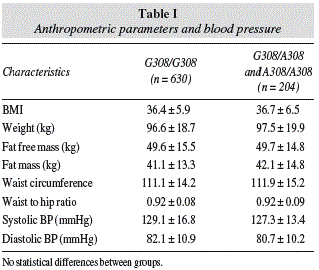
Table I shows anthropometric variables and blood pressure. No statistical differences were detected.
Table II shows cardiovascular risk factors. In mutant genotype group did not have worse metabolic profile tan wild genotype group.
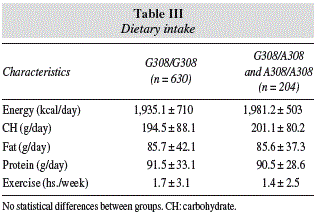
Table III shows nutritional intake with 3 days written food records an exercise. No statistical differences were detected in calory, carbohydrate, fat, and protein intakes.
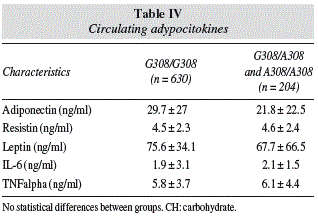
Table IV shows levels of adipocytokines, without statistical differences.
Discussion
In our sample of obese subjects, the alleic frequency of the substitution at position -308 was 13.19%, which is in accordance with allelic frequencies observed in French,13 Britich,14 White American,15 African American,16 Australian17 and Danish subjects18 but lower than the frequency observed in the Irish population.13 There were no differences between genotype groups with respect to estimates of obesity (weight, fat mass by bioimpedance, waist circumference, body mass index), serum insulin, insulin resistance by HOMA, serum lipids or adipocitokines.
Patients with A308 variant did not have higher concentrations of adipocitokines, insulin resistance, IL6 and TNF-α than G308 variant. Data in the literature are contradictories,18-19 some studies did not demonstrate a major role of the -308 substitutions of the TNF alpha gene in the pathogenesis of high levels of TNF alpha or insulin resistance. Others studies (20) have reported that the polymorphism at position -308 (TNF-308 G->A) leads to a higher rate of TNF alpha gene transcription, followed by raised TNF alpha concentrations and decreased insulin sensitivity). The disparity between previous studies might reflect differences in genetic backgraound, sex distribution or age.
In previous studies,18-20 dietary intake has not been controlled. In our study, we reported a similar energy and macronutrient intakes in both groups, and this factor was controlled. However, dietary intake might interact with this polymorphism in previous designs. In this hypothesis, our group21-22 has demonstrated that weight loss secondary to hypocaloric diet had different metabolic response depending of G308A genotype. These results have not been reported after bariatric surgery.23
Relation of blood pressure and G308A polymorphism in TNF alpha gene remains unclear. Some studies24 have found no difference in systolic blood pressure between genotypes. In other work,25 high systolic blood pressure was detected in A variant patients. Our data have shown similar blood pressure in both genotypes.
Our results of adipokines levels are interesting, adipokine levels was not different across genotypes. Only one earlier study has shown an association of the A308 allele with leptin levels.3 The most important variable that determines circulating leptin concentration is body fat mass. These differences in the literature may partially explain by differences in baseline BMI, weight loss and basal leptin levels of participants. Therefore, interaction between gene and diet could explain these differences with bias in previous studies, too. Patients with A 308 allele did not decrease leptin concentrations after weight loss21-22 and this is a variable to consider in further studies, as our design.
Finally, body mass index, fat mass or weight was not different across genotypes. In some studies,26 the allele A was associated with obesity, but this association has been demonstrated in individuals between 50 and 60 years. The mean age of our population was 46 years, this may indicate that obesity may either be related with this polymorphism after the fifth decade of life.
In conclusion, allelic frequency of G308A polymorphism is is in accordance with allelic frequencies observed in other populations. Carries of A308 allele have the same anthropometric and metabolic profile than wild type carriers.
References
1. Qi C, Pekala PH. Tumor necrosis factor alpha induced insulin resistance in adipocytes. Proc Soc Exp Biol Med 2000; 223: 128-125. [ Links ]
2. Wilson AG, Simons JA, McDowell TL, McDevitt HO. Effects of polymorphism of TNF alpha promoter on transcriptional activation. Proc N Atl Acad Sci USA 1997; 94: 3195-3199. [ Links ]
3. Fernández Real JM, Gutiérrez C, Ricart W, Casamitjan R, Fernández Castaner. The TNF alpha gene NCO I polymorphism influences the relationship among insulin resistance, percent body fat, and increased serum leptin levels. Diabetes 1997; 46: 1468-1471. [ Links ]
4. Da Silva B, Gaspur SM, Achenbach YH, Schuh TS, Kotla TJ. Lack of association between the G 308 A polymorphism of the tumor necrosis factor alpha gene and the insulin resistance syndrome. J Investig Med 2002; 48: 236-244. [ Links ]
5. Matsuda M, Shimomura I, Sata M. Role of adiponectin in preventing vascular stenosis. The missing link of adipo-vascular axis. J Biol Chem 2002; 277: 37487-37491. [ Links ]
6. Shimomoura I, Hammer RE, Ikemoto S. Leptin reverses insulin resitance and diabetes mellitus in mice with congenital lipodystrophy. Nature 1999; 401: 73-76. [ Links ]
7. Kumada M, Kihara S, Sumitsuji S. Association of hypoadiponectinemia with coronary artery disease in men. Arterioscler Thromb VascBiol 2003; 23: 85-9. [ Links ]
8. Matsuzawa Y. Adipocytokines: Emerging therapeutic targets. Current Atherosclerosis Reports 2005; 7: 58-62. [ Links ]
9. Steppan CM, Bailey ST, Bhat S. The hormone resistin links obesity to diabetes. Nature 2001; 409: 307-312. [ Links ]
10. Mathews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher Df. Homesotasis model assessment: insulin resistance and beta cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985; 28: 412-414. [ Links ]
11. Lukaski H, Johson PE. Assessment of fat-free mass using bio-electrical impedance measurements of the human body. Am J Clin Nutr 1985; 41 (4): 810-7. [ Links ]
12. Mataix J, Mañas M. Tablas de composición de alimentos españoles. Ed: University of Granada, 2003. [ Links ]
13. Herrmann Sm, Ricard Sm Bicaud V. Polymorphisms of the tumour necrosis factor alpha gene, coronary heart disease and obesity. Eur J Clin Invest 1998; 28: 59-66. [ Links ]
14. Day CP, Grove J, Daly AK, Stewart MW, Avery PJ. Tumour necrosis factor alpha gene promoter polymorphism and decreased insulin resistance. Diabetologia 1998; 41: 430-434. [ Links ]
15. Walston J, Seibert M, Yen C, Cheskin LJ, Andersen RE. Tumor necrosi factor alpha 238 and -308 polymorphisms do not associate with traits related to obesity and insulin resistance. Diabetes 1999; 2096-2098. [ Links ]
16. Hammann A, Mantzoros C, Vidal Puig A, Flier J, Genetic variability in the TNF promoter is not associated with type II diabetes mellitus (NIDDM). Biochem Biophys Res Commun 1995; 212: 833-839. [ Links ]
17. Milner CR, Graig JE, Hussey ND, Norman RJ. No association between the-308 polymorphism in the tumour necrosis factor alpha promoter region and polycictic ovaries. Mol Num Reprod 1999: 5: 5-9. [ Links ]
18. Rasmussen S, Urhammer S, Jensen J, Hansen T, Borch-Johnsem K. The-238 and -308 G->A polymorphisms of the tumor necrosis factor alpha gene promoter are not associated with features of the insulin resistance syndrome or altered birth weight in Danish Caucasians. J Clin Endocr and Metab 2000; 85: 1731-1734. [ Links ]
19. Sheu WH, Lee WJ, Lin LY, Cang RL, Chen YT. TNF alpha -238 amd -308 polymorphisms do not associate with insulin resistance in hypertensive subjects. Metabolism 2001; 50: 1447-1451. [ Links ]
20. Bayley JP, de Rooij H, van den Elsen PJ, Huizinga TW, Verweij CL. Functional analysis of linker scan mutants spanning the -376, -308,-244, and -238 polymrphic sites of the TNF alpha promoter. Cytoine 2001; 14: 316-323. [ Links ]
21. De Luis DA, Aller R, Izaola O, González M, Conde R, Romero E. Influencia del polimorfismo G308A del factor de necrosis tumoral alfa en la resistencia a la insulina en pacientes obesos tras perdida de peso. Med Clin 2007; 129: 401-404. FI 1,327. [ Links ]
22. De Luis DA, Aller R, Izaola O, González M, Conde R. Influence of G308A promoter variant of tumor necrosis factor alfa gene on insulin resistance and weight loss secondary to two hypocaloric diets: a randomized clinical trial. Arch Med Res 2009; 40: 36-41. [ Links ]
23. De Luis DA, Pacheco D, Aller R, González Sagrado M, Izaola O, Terroba MC, Cuellar L, Conde R, Martín T, Perez Castrillón JL. Influence of G308A polymorphism of Tumor necrosis alpha gene on surgical resutls of biliopancreatic diversion. Obes Surg 2010; 20: 221-225. [ Links ]
24. Hoffstedt J, Eriksson P, Hellstrom L, Rossner S, Ryden M, Arner P. Excessive fat accumulation is associated with the TNFalpha -308 G/A promoter polymorphism in women but not in men. Diabetologia 2000; 43: 117-120. [ Links ]
25. Dalziel B, Gosby AK, Richman RM, Bryson JM, Caterson ID. Association of the TNF alpha -308 G/A promoter polymorphism with insulin resistance in obesity. Obesity Research 2002; 10: 401-407. [ Links ]
26. Sookoian SC, González C, Pirola CJ. Meta-analysis on the G308A TNF alpha gene variant and phenotypes associated with Metaboic síndrome. Obesity Research 2005; 13: 2122-2130. [ Links ]
27. De Luis DA, Ballesteros M, Ruiz E, Muñoz C, Penacho A, Iglesias P, López Guzmán A, Maldonado A, Cordero M, San Martín L, Puigdevall V, Romero E, González Sagrado M, Izaola O, Conde R. Polymorphism Trp64Arg of beta 3 adrenoreceptor gene: allelic frequencies and influence on insulin resistance in a multicenter study of Castilla-León. Nutr Hosp 2010; 25 (2): 299-303. [ Links ]
28. De Luis DA, González Sagrado M, Aller R, Izaola O, Conde R, Romero E. G1359A polymorphism of the cannabinoid receptor gene (CNR1) and insulin resistance in patients with diabetes mellitus type 2. Nutr Hosp 2010; 25 (1): 34-8. [ Links ]
![]() Correspondence:
Correspondence:
D. A. De Luis.
Executive Director of Center of Investigation
of Endocrinology and Nutrition.
Medicine School. Valladolid University.
C/ Los Perales, 16.
47130 Simancas. Valladolid. Spain.
E-mail: dadluis@yahoo.es
Recibido: 6-II-2011
Aceptado: 9-II-2011.