Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Nutrición Hospitalaria
versión On-line ISSN 1699-5198versión impresa ISSN 0212-1611
Nutr. Hosp. vol.28 no.4 Madrid jul./ago. 2013
https://dx.doi.org/10.3305/nh.2013.28.4.6461
ORIGINAL/Otros
Cytotoxicity of Agaricus sylvaticus in non-tumor cells (NTH/3T3) and tumor (OSCC-3) using tetrazolium (MTT) assay
Citotoxicidad de Agaricus sylvaticus en células no tumorales (NIH/3T3) y el tumor (CCCA-3) usando tetrazolio (MTT)
Joice Vinhal Costa Orsine1, Luíssa Marques Brito2, Renata Carvalho Silva3, Maria de Fátima Menezes Santos Almeida4 and Maria Rita Carvalho Garbi Novaes5
1Professor. Instituto Federal Goiano-Campus Urutai. Brazil
2Estudante do Curso de Medicina. Escola Superior de Cièncias da Saùde-ESCS. Brasilia, DF
3Aluna de Doutorado. Universidade de Brasilia. Campus Universitario Darcy Ribeiro. Brasilia. Brazil
4Professor. Universidade de Brasilia. Campus Universitario Darcy Ribeiro. Brasilia. Brazil
5Professor. School of Medicine. Escola Superior de Cièncias da Saùde. ESCS-FEPECS; Universidade de Brasilia. Brazil
We thank ESCS to support this work.
ABSTRACT
The purpose of this study was to assess the cytotoxic effect of the non-fractionated aqueous extract of A. sylvaticus mushroom in cultures of non-tumor cells (NIH3T3) and tumor cells (OSCC-3). The cells were maintained in DMEN cell culture medium added of 10% of fetal bovine serum and 1% antibiotic. For the cytotoxicity test we prepared the aqueous mushroom extract at concentrations of 0.01 mg.ml-1, 0.02 mg.ml-1, 0.04 mg.ml-1, 0.08 mg.ml-1, 0.16 mg.ml-1, and 0.32 mg.ml-1. For the culture, 2 x 105 cells/ml was deposited in 96-well microplates during 24 hour incubation with subsequent exchange of medium by another containing the mushroom concentrations. After 24 hour incubation the medium was discarded and 100 ml of tetrazolium blue (MTT) was added at a concentration of 5 mg.ml-1. The microplates were incubated for 2 h at 37o C. Spectrophotometric analysis was performed using 570 nm wavelength. From the values of the optical densities we determined the drug concentration capable of reducing cell viability by 50%. Therefore, the mushroom A. sylvaticus, at all concentrations tested, did not show cytotoxic effects, once the inhibitory concentration (IC50) obtained for tumor cells OSCC-3 was 0.06194 mg.ml-1, and the IC50 checked for non-tumor cells NIH3T3 was 0,06468 mg.ml-1. This test made it possible to determine that A. sylvaticus mushroom has no cytotoxic effects, suggesting its use safe for human consumption.
Key words: Toxicity. Food safety. Agaricus sylvaticus.
RESUMEN
El objetivo de este estudio fue evaluar el efecto citotóxico de un extracto acuoso no fraccionado de la seta A. sylvaticus en cultivos de células no tumorales (NIH3T3) y tumorales (OSCC-3). Se mantuvo a las células en un medio de cultivo celular DMEN al que se añadió suero de ternera fetal al 10% y antibiótico al 1%. Para la prueba de toxicidad, se preparó el extracto acuoso del hongo a concentraciones 0,01 mg.ml-1, 0,02 mg.ml-1, 0,04 mg.ml-1, 0,08 mg.ml-1, 0,16 mg.ml-1 y 0,32 mg.ml-1. Para el cultivo, 2 x 105 células/ml se depositaron en microplacas de 96 pocillos durante 24 horas de incubación con el subsiguiente recambio del medio por otro que contenía las concentraciones del hongo. Tras 24 horas de incubación, se desechó el medio y se añadieron 100 ml de azul de tetrazolio (MTT) a una concentración de 5 mg.ml-1. Se incubaron las microplacas durante 2 h a 37o C. Se realizó un análisis espectrofotométrico con una longitud de onda de 570 nm. A partir de los valores de las densidades ópticas, se determinó la concentración de la droga capaz de reducir la viabilidad celular en un 50 %. La seta A. sylvaticus, a todas las concentraciones probadas, no mostró efectos citotóxicos una vez que la concentración inhibitoria (IC50) obtenida para las células tumorales OSCC-3 fue de 0,06194 mg.ml-1 y la IC50 comprobada para las células no tumorales NIH3T3 fue de 0,06468 mg.ml-1. Esta prueba consiguió determinar que la seta A. sylvaticus no posee efectos citotóxicos lo que sugiere un uso seguro para el consumo humano.
Palabras clave: Toxicidad. Seguridad alimentaria. Agaricus sylvaticus.
Abbreviations
A. sylvaticus: Agaricus sylvaticus.
CaCo-2: Colon cancer cells.
DMEM: Dulbecco's Modified Eagle Medium.
EDTA: Ethylenediamine-tetraacetic acid.
Hep-2 cell line: Human epithelial cells derived from a larynx carcinoma.
IC50: Inhibitory concentration.
MTT: 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide.
NIH/3T3: Non-tumoral fibroblasts cell line.
OSCC-3: Oral squamous cell carcinoma.
Introduction
The mushrooms of the genus Agaricus have long been considered functional foods for their rich chemical composition and high amount of bioactive compounds, bringing many benefits to the health of those who consume it, besides the absence of toxicity (Orsine et al., 2012a).
Studies have been conducted in an effort to utilize mushrooms of the genus Agaricus in the treatment of various ailments. The Agaricus blazei Murill mushroom showed antinociceptive and anti-inflammatory effects in Wistar rats (Carvalho et al., 2011); protective effect against lethal infection with Streptococcus pneumoniae in mice (Bernadshaw et al., 2005); reducing effect on the degree of edema and hemorrhagic halo in bothropic poisoning in experimental rabbits (Ferreira et al., 2003) further to high potential use in the treatment of leishmaniasis (Valadares et al., 2012). A dietary supplementation with A. sylvaticus was able to improve gastrointestinal disorders in post-surgery patients with colorectal cancer as well as the quality of life of these patients (Fortes et al., 2010). The Agaricus bisporus mushroom stimulated the production of immunoglobulin A in saliva samples of healthy volunteers, suggesting that its use was responsible for developing immunity (Jeong et al. 2012).
However, there are few toxicological studies on edible mushrooms and food safety tests. The 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) test has often been used to investigate cytotoxicity caused by medicinal plants (Shoeb et al. Jan 2012; Talib and Mahasneh, 2010) and fungi with antimicrobial activity (Joel and Bhimba, 2012).
The principle of the MTT technique consists in the absorption of yellow tetrazolium salts by mitochondrial reductases of metabolically active cells, resulting in a product called formazan. This product accumulated intracellularly, is extracted by adding an appropriate solvent. This is a low-cost method, yielding fast results in 48 hours (Mosmann, 1983).
The purpose of this study was to perform cytotoxicity screening of the aqueous extract of A. sylvaticus mushroom in non-tumoral fibroblasts cell line (NIH/3T3) and oral squamous cell carcinoma (OSCC-3), using the MTT reduction test.
Materials and methods
Obtaining the sample
The A. sylvaticus mushroom was obtained from a producer in Minas Gerais, Brazil, in 2010. The sample was dried and milled.
Preparation of extract
We weighed 10 g of dehydrated minced mushroom, and diluted it in 100 ml of distilled water. The solution was stirred in a mechanical shaker for 30 minutes and was then filtered through filter paper.
The filtered solution was then distributed into eppendorfs 1mL previously weighed and identified, frozen, and subsequently taken to a liophylization chamber. After complete sublimation of water, we weighed again the eppendorfs containing the soluble solids in mushroom A. sylvaticus' water.
We prepared the non fractionated aqueous extract of the mushroom A. sylvaticus at concentrations: 0.33 mg.ml-1, 0.16 mg.ml-1, 0.08 mg.ml-1, 0.04 mg.ml-1, 0.02 mg.ml-1, and 0.01 mg.ml-1.
In vitro study
In vitro studies were carried out following the methodology proposed by Saldanha (2007), from the MTT assay.
Culture and proliferation of non-tumor fibroblast cell line (N1H/3T3) and oral squamous cell carcinoma (OSCC-3)
Cell lines NIH/3T3 (non-tumor fibroblasts) and OSCC-3 (immortalized cells in culture from a human oral squamous cell carcinoma) were maintained separately in culture medium DMEM (Dulbecco's Modified Eagle Medium), GIBCO-BRL, supplemented with 10% fetal bovine serum (GIBCO-BRL) and 1% of antibiotics (penicillin-streptomycin).
The cultures were set up from an initial passage of 2 x 105 cells in 75 cm2 culture flasks, maintained in an incubator at 37 oC with saturated humidity of 5 % CO2-atmosphere. Upon reaching 80-90% confluence, cells were released from the bottom of the flask by treatment with 0.125% trypsin solution/0.02% EDTA (ethylenediamine-tetraacetic acid) for two minutes, centrifuged at 1,000 rpm for three minutes, using Neubauer counting chamber and transferred to a new culture flask.
Treatment of NIH/3T3 cells and OSCC-3 with non-fractioned aqueous extract of mushroom A. sylvaticus
After 24 hours of cultivation in the presence of non-fractioned aqueous extract of mushroom A. sylvaticus sample, cells were subjected to MTT test to determine viability of the isolated cells. Concentrations of the non-fractioned aqueous extract were added to the cultures, which were maintained for 24 hours under the conditions described in section 2.3.1. We used solution DMEN only as negative control. The NIH/3T3 cells and OSCC-3 were maintained at the Nanobiotechnology laboratory, Genetics and Morphology Department, Brasilia University.
Analysis of cell viability
Cell viability was assessed after two hours contact of NIH/3T3 cells and OSCC-3 with MTT in spectrophotometer. For the reading we used wavelength of 570 nm. The result obtained indicates the optical density, since the darker the color obtained, the greater the MTT metabolism of the cells under study. Consequently, a higher optical density results in less toxicity of the extract tested. We used the Prism Graph Software to analyze the results.
The cytotoxicity of each concentration of the non-fractionated aqueous extract of the mushroom A. sylvaticus was expressed by cell death, calculated in relation to negative control, according to the methodology proposed by Zhang et al. (2004).
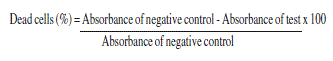
The data generated were used to plot a dose-response curve which determines the extract concentration capable of killing 50% of the cell population tested, indicating IC50 (inhibitory concentration).
Statistical analysis
Data were expressed as the mean percentage of toxicity. Significance levels among concentrations of non-fractionated aqueous extract of A. sylvaticus mushroom tested were analyzed using analysis of variance (ANOVA), with Software Graphpad PRISM® 4.0. For multiple comparisons among groups, control group and intra-group, we used the Newman-Keuls test, with significance set at p < 0.05.
Results
Agaricus sylvaticus mushroom have a rich chemical composition, highlighting the variety and quantity of minerals as well as its high protein content (Orsine et al., 2012b). But, to be approved in the in vitro cytotoxicity assays, the sample to be tested must not cause cell death nor affect its cellular functions. Therefore, tests using cell culture can detect cell lysis, growth inhibition and other effects that can be triggered onto these cells (Daguano et al., 2007).
In figure 1 we presented the results for the OSCC-3 cells treated with different concentrations of mushroom A. sylvaticus. The IC30 determined was of 0.06194 mg.ml1, that is, the A. sylvaticus non-fractionated water extract does not show toxicity in tumor cells used in this study.
In figure 2 the results were expressed regarding NIH3T3 cell culture treated with different concentrations of mushroom A. sylvaticus. The IC50 found was 0.06468 mg.mr1, that is, the A. sylvaticus non-fractionated water extract showed no toxicity in non-tumor cells analyzed.
Discussion
This study investigated the mushroom A. sylvaticus and its safe use in food. These results may contribute towards research done with A. sylvaticus, toxicity testing and food safety, supplement, or as an adjunct in cancer treatment, since very low toxicity of the extract was observed in two types of cells tested.
Mushrooms of the genus Agaricus have been widely studied by several authors, in search of answers to their toxicity (Chang et al., 2012; Orsine et al., 2012c; Bellini et al., 2008; Novaes et al., 2007; Singi et al. 2006; Sugui et al. 2006; Kuroiwa et al. 2005; Costa et al. 2003).
Table I presents studies on the toxicity of edible mushrooms of different genres, performed worldwide in the period from 2003 to 2012 in order to support the discussion of this work.
Plants used in folk medicine in Jordan were tested for cytotoxic effects using the MTT assay on Vero cell line. The Rosa damascena plant showed IC50 value of 454.11 mg.ml-1, whereas the Ononis hirta plant showed IC50 of 72.50 mg.ml-1 (Talib and Mahasneh, 2010).
The cytotoxicity of five strains of fungus Penicillium thiomii (named as IR-1, IR-2, IR-4, IR-6 and IR-7) isolated from the medicinal plant Terminalia chebula Retz, in Bangladesh, was evaluated by the MTT assay. The ethyl acetate extract of the fungus strains inhibited the growth of colon cancer cells CaCo-2. Values were obtained for the IC50 ranging from 44 to 67 mg.ml-1 (Shoeb et al. 2012).
The cytotoxicity caused by the extract of fungi Pestalotiopsis Microspora VB5 was screened using the MTT test. As a result, the authors observed that the concentration of the extract tested was inversely proportional to Hep-2 cell line (human epithelial cells derived from a larynx carcinoma) growth (Joel and Bhimba, 2012).
Conclusion
The non-fractionated aqueous extract of the mushroom A. sylvaticus showed no cytotoxic effect on tumor cells OSCC-3 and non-tumor cells NIH/3T3, showing to be safe for use in food and/or dietary supplementation.
References
1. Bellini MF, Cabrioti LN, Terezan AP, Jordão BQ, Ribeiro LR, Mantovani MS. Cytotoxicity and genotoxicity of Agaricus blazei methanolic extract fractions assessed using gene and chromosomal mutation assays. Genet Mol Biol 2008; 31 (1). [ Links ]
2. Bernardshaw S, Johnson E, Hetland G. An extract of the mushroom Agaricus blazei Murill administered orally protects against systemic Streptococcus pneumoniae Infection in Mice. Scandinavian Journal of Immunology 2005; 62: 393-8. [ Links ]
3. Carvalho C, Alves NC, Monteiro AC, Pelógia NCC. Antinociceptive and anti-inflammatory effect of Agaricus blazei Murill in rats submitted to the modified formalin test. Rev Dor São Paulo 2011; 12 (1): 50-3. [ Links ]
4. Chang JB, Lu HF, Liao NC, Lee CS, Yeh MY, Liu CM, Chung MT, Man-Kuan A, Lin JJ, Wu MF, Chung JG. Evaluation of genotoxicity and subclinical toxicity of Agaricus blazei Murrill in the Ames test and in histopathological and biochemical analysis. In Vivo 2012; 26 (3): 437-45. [ Links ]
5. Costa WF, Nepomuceno JC. Efeito protetor do chá de cogumelo do sol (Agaricus blazei Murill) contra a ação genotóxica do uretano em células somáticas de Drosophila melanogaster. Rev Ciênc Farmac 2003; 24: 153-8. [ Links ]
6. Daguano JKMF, Santos C, Rogero SO. Avaliação da citotoxicidade de biocerâmicas desenvolvidas para uso em sistemas de implantes. Matéria 2007; 12 (1): 134-9. [ Links ]
7. Ferreira KM, Melo MM, Dantas-Barros AM. Tratamento tópico de coelhos com Agaricus blazei Murril após envenenamento botrópico experimental. Revista Brasileira de Farmacognosia 2003; 13: 77-80. [ Links ]
8. Fortes RC, Recova VL, Melo AL, Novaes MRCG. Alterações gastrointestinais em pacientes com câncer colorretal em ensaio clínico com fungos Agaricus sylvaticus. Rev Bras Coloproct 2010: 30 (1): 45-54. [ Links ]
9. Gill SK, Rieder MJ. Toxicity of a traditional Chinese medicine, Ganoderma lucidum, in children with cancer. Can J Clin Pharmacol 2008; 15 (2): 275-85. [ Links ]
10. Jeong SC, Koyyalamudi SR, Pang G. Dietary intake of Agaricus bisporus white button mushroom accelerates salivary immunoglobulin A secretion in healthy volunteers. Nutrition 2012; 28: 527-31. [ Links ]
11. Joel EL, Bhimba BV. Fungi from mangrove plants: their antimicrobial and anticancer potentials. International Journal of Pharmacy and Pharmaceutical Sciences 2012; 4 (3): 139-142. [ Links ]
12. Kim MO, Moon D, Jung JM, Lee WS, Choi YH, Kim G. Agaricus blazei Extract Induces Apoptosis through ROS-Dependent JNK Activation Involving the Mitochondrial Pathway and Suppression of Constitutive NF- kB in THP-1 cells. Evid Based Complement Alternat Med 2011. [ Links ]
13. Kuroiwa Y, Nishikawa A, Imazawa T, Kanki K, Kitamura Y, Umemura T, Hirose M. Lack of subchronic toxicity of an aqueous extract of Agaricus blazei Murrill in F344 rats. Food and Chemical Toxicology 2005; 43 (7): 1047-53. [ Links ]
14. Luo H, Liu Y, Fang L, Li X, Tang N, Zhang K. Coprinus comatus Damages Nematode Cuticles Mechanically with Spiny Balls and Produces Potent Toxins To Immobilize Nematodes' Appl Environ Microbiol 2007; 73 (12): 3916-23. [ Links ]
15. Mantovani MS, Matuo R, Bellini MF, Oliveira RJ, Ribeiro LR. Atividade clastogênica e genotóxica de altas concentrações do extrato aquoso de Agaricus brasiliensis e diferentes respostas quando associado aos inibidores de reparo de DNA, ARA-C e 3DEOT, in vitro. Semina: Ciências Biológicas e da Saúde 2006; 27 (1): 13-22. [ Links ]
16. Mosmann T. Rapid colorimetric assay for cellular growth and survival. J Immunol Methods 1983; 65: 55-63. [ Links ]
17. Motoi M, Ohno N. Safety study of culinary-medicinal Royal Sun Agaricus, Agaricus brasiliensis S. Wasser, et al. KA21 (higher Basidiomycetes) assessed by prokaryotic as well as eukaryotic systems. Int JMed Mushrooms 2012; 14 (2): 135-48. [ Links ]
18. Nieto RIJ, Salama AM, Cataño PJE, Chegwin AC. Determination of Pleurotus ostreatus, Pleurotus pulmonarius and Paxillus involutus toxicity over Artemia salina. Rev Iberoam Micol. 2008; 25 (3): 186-7. [ Links ]
19. Novaes MRCG, Novaes LCG, Melo AL, Recôva VL. Avaliação da toxicidade aguda do cogumelo Agaricus sylvaticus. Comunicação em Ciências da Saude 2007; 18 (3): 227-36. [ Links ]
20. Orsine JVC, Costa RV, Novaes MRCG. Mushrooms of the genus Agaricus as functional foods. Nutr Hosp 2012a; 27 (4): 1017-24. [ Links ]
21. Orsine JVC, Novaes MRCG, Asquieri ER. Nutritional value of Agaricus sylvaticus; mushroom grown in Brazil. Nutr Hosp 2012a; 27 (2): 449-55. [ Links ]
22. Orsine JVC, Costa RV, Silva RC, Santos MFMA, Novaes MRCG. The acute cytotoxicity and lethal concentration (LC50) of Agaricus sylvaticus through hemolytic activity on human erythrocyte. International Journal of Nutrition and Metabolism 2012b; 11 (4): 19-23. [ Links ]
23. Postemsky PD, Palermo AM, Curvetto NR. Protective effects of new medicinal mushroom, Grifola gargal singer (higher Basidiomycetes), on induced DNA damage in somatic cells of Drosophila melanogaster. Int J Med Mushrooms 2011; 13 (6): 583-94. [ Links ]
24. Saldanha CA. Avaliação in vitro da citotoxicidade e genotoxicidade dos polímeros de albumina magnéticos. Dissertação (Mestrado em Biologia Animal), Universidade de Brasília. 2007, 71p. [ Links ]
25. Savi T, Patenkovi A, Sokovi M, Glamoclija J, Andjelkovi M, van Griensven L. The effect of royal sun agaricus, Agaricus brasiliensis S. Wasser, et al., extract on methyl methanesulfonate caused genotoxicity in Drosophila melanogaster. Int J Med Mushrooms 2011; 13 (4): 377-85. [ Links ]
26. Shoeb M, Thoo-Lin PK, Nahar N. Anticolon cancer activity of endophytic fungal strains from Terminalia chebula Rezt. Bangladesh J Pharmacol 2012; 7: 47-9. [ Links ]
27. Singi G, Damasceno DD, D'Andréa ED, Alexandre GMB, Singi MB, Lira C Alves LC, Simões TI. Efeitos agudos da aplicação endovenosa do cogumelo-do-sol (Agaricus blazei Murill) sobre a pressão arterial mèdia e a freqüência cardiaca de ratos anestesiados. Rev Bras Farmacogn 2006; 16 (4): 480-4. [ Links ]
28. Sugui MM. Mecanismos de antimutagenicidade do cogumelo Agaricus brasiliensis sobre lesões no DNA induzidas in vivo e in vitro. Tese Apresentada à Universidade Estadual Paulista. Faculdade de Medicina de Botucatu para obtenção do grau de Doutor. Botucatu. 2006, 43 p. [ Links ]
29. Talib WH, Mahasneh AM. Antimicrobial, cytotoxicity and phytochemical screening of Jordanian plants used in traditional medicine. Molecules 2010; 15: 1811-24. [ Links ]
30. Valadares DG, Duarte MC, Ramírez L, Chávez-Fumagalli MA, Lage PS, Martins VT, Costa LE, Ribeiro TG, Régis WC, Soto M. Fernandes AP, Tavares AP, Coelho EA. Therapeutic efficacy induced by the oral administration of Agaricus blazei Murill against Leishmania amazonensis. Parasitol Res 2012; 111: 1807-16. [ Links ]
31. Yoshioka Y, Tamesada M, Tomi H. A repeated dose 28-day oral toxicity study of extract from cultured Lentinula edodes mycelia in Wistar rats. Journal of Toxicological Sciences 2010; 35 (5): 785-91. [ Links ]
32. Zhang Y, Ong CN, Shen HM. Involvent of proapoptotic Bcl-2 family members in parthenolide induced mitochondrial dysfunction and apoptosis. Cancer Lett 2004; 211 (2): 175-88. [ Links ]
![]() Correspondence:
Correspondence:
Joice Vinhal Costa Orsine
Professor. Instituto Federal Goiano-Campus Urutai
Rodovia Geraldo Silva Nascimento, km 2.5
CEP 75790-000 Urutaí - Goiás - Brazil
E-mail: joicevinhal@gmail.com
Recibido: 30-I-2013
Aceptado: 13-II-2013














