Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Nutrición Hospitalaria
versión On-line ISSN 1699-5198versión impresa ISSN 0212-1611
Nutr. Hosp. vol.30 no.4 Madrid oct. 2014
https://dx.doi.org/10.3305/nh.2014.30.4.7301
ORIGINAL / Alimentos funcionales
Effects of flaxseed flour on the lipid profile of rats submitted to prolonged androgen stimuli
Efectos de la harina de semilla de linaza en el perfil lipídico de ratas sometidas a estímulos androgénicos prolongados
Ilma Cely de Amorim Ribeiro1, Carlos Alberto Soares da Costa2, Vivian Alves Pereira1, Gilson Teles Boaventura2 and Mauricio Alves Chagas1
1 Department of Morphology, Laboratory of Cellular and Extracellular Biomorphology Biomedic Institute, Federal Fluminense University. Brasil.
2 Department of Nutrition and Dietetics, Experimental Nutrition Laboratory, College of Nutrition, Federal Fluminense University, Rio de Janeiro, Brazil.
This work was supported by the Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ).
ABSTRACT
Background: The chronic use of steroid hormones can lead to alterations in the lipid profile such as an increase in LDL and decrease in HDL levels . The effect of flaxseed on lipid profiles has been widely investigated.
Aim: Evaluate the lipid profile of adult male Wistar rats fed with flax based meals and submitted to androgenic hyperstimulation.
Material and Methods: Forty Wistar rats were divided into 4 groups of 10 animals: the Control group (CG); Flax group (FG) fed a flaxseed flour-based meal; Induced group (IG); and the Induced group (IGF) that was fed a flaxseed flour-based meal. The induction was done by using silicone pellets filled with testosterone propionate (1mg), sealed with a surgical adhesive and substituted every 4 weeks.
Results: Triglycerides (FG: 71.16 ± 21.95; IG: 99.16 ± 26.00 and IGF: 86.33 ± 27.16 mg/dL) and HDL-cholesterol (FG: 23.05 ± 1.67; IG: 29.06 ± 7.24 and IGF: 26.06 ± 3.56 mg/dL) were significantly lower in the experimental groups. The FG and IGF (41.16 ± 3.97 and 49.66 ± 11.25 mg/dL, respectively) showed significantly lower levels of cholesterol than the other groups(CG: 78,85 ± 11.58 and IG: 70,83 ± 14.85 mg/dL). Regarding LDL levels, the IG showed significantly higher concentrations (21,93 ± 8,84 mg/dL) than the others groups (CG: 7,81 ± 5,37; FG: 3,88 ± 1,32 and IGF: 6,66 ± 7,24 mg/dL).
Conclusions: The flaxseed has a relevant effect on the lipid profile of animals submitted to androgenic hyperstimulation.
Key words: steroid hormone, rat, cholesterol, flaxseed.
RESUMEN
Introducción: El uso crónico de hormonas esteroides puede causar alteraciones en el perfil lipídico como el aumento de las LDL y reducción de las HDL. Los efectos de la linaza en el perfil lipídico han sido extensivamente investigados.
Objetivo: Evaluar el perfil lipídico de ratas Wistar machos adultos alimentados con piensos a base de linaza y sometidos a hiperestimulaciones androgénicas.
Materiales y Métodos: Cuarenta ratas Wistar fueron divididos en 4 grupos de 10 animales: Grupo control (GC); Grupo de linaza (GL), alimentados con piensos a base de harina de linaza ; Grupo Inducido (GI); y Grupo Inducido (GIL) alimentados con piensos a base de harina de linaza. La inducción fue realizada utilizando pellets de silicona rellenados con propionato de testosterona (1 mg) cerrados con un adhesivo quirúrgico y sustituidos cada 4 semanas.
Resultados: Los triglicéridos (GL: 71.16 ± 21.95; GI: 99.16 ± 26.00; GIL: 86.33 ± 27.16 mg/dL) y colesterol-HDL (GL: 23.05 ± 1.67; GI: 29.06 ± 7.24; GIL: 26.06 ± 3.56 mg/dL) estaban significativamente más bajos en los grupos experimentales. EL GL (41.16 ± 3.97 mg/dL) y GIL (49.66 ± 11.25 mg/dL) presentaron niveles menores de colesterol que los otros grupos (GC: 78,85 ± 11.58; GI: 70,83 ± 14.85 mg/dL) y el GI concentraciones significativamente mayores de LDL (21,93 ± 8,84 mg/dL) que los otros grupos (GC: 7,81 ± 5,37; GL: 3,88 ± 1,32; GIL: 6,66 ± 7,24 mg/dL).
Conclusión: La linaza presenta efectos relevantes en el perfil lipídico de animales sometidos a hiperestimulaciones androgénicas.
Palabras clave: hormonas esteroides, rata, colesterol, semilla de linaza.
Introduction
Those who chronically use steroid hormones show changes in their lipid profile, including an increase in LDL levels (low density lipoproteins) e decrease in HDL levels (high density lipoproteins)1. Anabolic steroids have been used in medicine for at least five decades and their therapeutic indications are associated to hypogonadism, hormone replacement and protein metabolism deficiencies. They are also used in sports for the enhancement of athletic performance2. Similar to endogenous hormones, synthetic steroid hormones also have anabolic and androgenic effects3.
Strength athletes that consume steroid hormones have an increased risk of atherosclerosis due to changes in their lipid profile, increase in LDL concentrations and decrease of HDL cholesterol. The increased risk is due to the deposition of cholesterol plates on vessel walls along with an increase in platelet aggregation and probable endothelial dysfunction. Both the lipid profile changes and clotting and the endothelial dysfunction can lead to an increased risk of coronary spasm4. Some observational studies, case reports and literature reviews link the use of steroid hormones to important changes in the lipid profile5,6,7,8. Although some studies have suggested that testosterone reduces serum levels of high density lipoproteins (HDL), there are other studies that show that testosterone does not have any effect on HDL levels9,10.
Testosterone is a steroid hormone that originates cholesterol and is the main androgen hormone in men with an important anabolic role. Around 95% of it is secreted by Leydig cells located in testicles and 5% by the cortex of the adrenal glands11. Testosterone can be administered through injections, adhesives, gels, topically, pills or implants9,12. Testosterone levels gradually decline with age and a deficiency of it can cause significant morbidity and a substantial reduction in the quality of life13.
Flaxseed is the richest vegetable source of omega 3 fatty acids α linolenic acid) and of the phytohormone lignan. It is also an essential source of high quality proteins and fibers, as well as a source of phenolic compounds14. It can also contribute to the reduction of many diseases such as diabetes mellitus, atherosclerosis, cancer15, improvement of prostate health16, breast cancer17 and cardiovascular protection through improvements in the lipid profile18. Due to the presence of components that have physiologically beneficial effects on one's health, as well those that are part of a basic nutrition, flaxseed is included in on the of the following categories: functional foods, bioactive foods and/or food with endocrine effects19,20,21,22.
The effect of flaxseeds on lipid profiles has been widely investigated and attributed not only to the seed flour but also to SDG (secoisolariciresinol), to its oil and its protein. In mice, SDG reduces hyperlipidemia and hypercholesterolemia23. Flaxseed oil reduced total and LDL cholesterol in rats that consumed a diet that was rich in fat, as well as protected them from renal lesions associated to hypercholesterolemia24. Also, the consumption of diets with flax protein reduced serum cholesterol and triglycerides in rats with normal lipid profiles25.
The aim of our study is to evaluate the lipid profile of adult male Wistar rats fed with flaxseed-based and casein-based meals and compare them with animals that were submitted to androgenic hyperstimulation to verify the feasibility of this seed as a regulator of lipid function.
Material and methods
Experimental Procedure
The research project was approved by the Ethical Committee on Animal Use (CEUA - Comité de Ética no Uso de Animais) of the Federal Fluminense University under the number 236. Forty Wistar rats were selected and divided into 4 groups of 10 animals: the Control group (CG) that received a casein-based meal; the Flax group (FG) that received a flax flour-based meal; the Induced group (IG) that received a casein-based meal; and the Induced group (IGF) that was fed a flax flour-meal. The study was done on young adult male rats (42-50 days old) that were kept in plastic cages with a constant cycle of 12 hours of light and 12 hours of darkness at a temperature of 22± 1oC. Hyperplasia induction was done by using silicone pellets (Dow Chemicals) filled with testosterone propionate (1mg) and sealed with a surgical adhesive. These pellets were inserted in the dorsoscapular region (incision of approximately 10mm) with intraperitoneal anesthesia (xylazine 2% and ketamin 10%) and substituted every 4 weeks12.
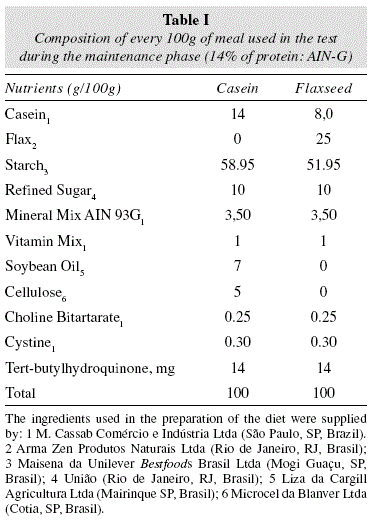
Experimental meal
The seed was ground in a blender to obtain the flour that was then weighed and bagged to be used immediately to produce the meal. The prepared experimental meal was isocaloric and had a vitamin and mineral mix added to it according to the recommendations of the American Institute of Nutrition (AIN-93M)[26] during the experimental period. The meal that was offered to the flax group had a 25% flax flour concentration with the objective of offering all the recommended input of fiber. The ingredients in the experimental meal (table I) were weighed and homogenized with a Hobart® industrial mixer (São Paulo, SP, Brazil) with boiling water for the gelatinization of the starch. The obtained dough was transformed into pellets and dried in a ventilated incubator (Fabbe-Primar® no171, São Paulo, SP, Brazil) at 60oC for 24h and, after identification, stored under refrigeration until use. All animals were weighed at the beginning of the biological test and, from that moment on, twice every week throughout the entire experiment. The weighing was done on a digital scale (Gehaka) with a precision of 0.05 g.
Biochemistry Analysis
After 20 weeks of the experiment, the rats were euthanized with a high dosage of sodium thiopental intraperitoneously for the collection of blood through cardiac puncture. The blood collected without EDTA was centrifuged at 3500 rpm during 15 minutes to obtain the serum that was stored at -20oC. The analysis of albumin, total proteins, cholesterol, triglycerides, LDL and HDL was done using colorimetric kits of the LabTest device (LabMax, Belo Horizonte, Brazil).
Hormone Indicators
Estradiol was analyzed using a radioimmunoassay method (Perkin Elmer equipment, "WIZARD2 Automatic Gamma Counter" device and Siemens kit). While testosterone was analyzed with a chemoluminescence method (Elecsys equipment, Roche brand and Roche kits)
Statistical Analysis
The data was shown using averages and standard deviations. The normal distribution of the values found were evaluated using the Kolmogorov-Smirnov test. For the present study the univariate ANOVA Test associated with the Tukey-Kramer multiple comparison test was used. The significance level in all tests was established as p<0.05. The statistical analyses were done by the program Graph Pad Prism statistical package version 5.0, 2007 (San Diego, CA, USA).
Results
It was found that the flax induced group had a lower serum triglyceride concentration (-12%) than the casein induced group. The flax control group had a lower triglyceride average than the flax induced group. The flax induced group had higher triglyceride concentration (+21%) when compared to the flax control group. The experimental groups had lower triglyceride levels than the casein control group.
Regarding cholesterol, the flax induced group and flax control group had lower average cholesterol values (P<0.05) than the casein induced group. The flax control group and flax induced group also had lower a serum cholesterol concentration (P<0.05) than the casein control group.
The experimental groups had significantly lower serum HDL-cholesterol levels (P<0.05) when compared to the casein control group.
The serum LDL concentration in the flax induced group and the flax control group was lower (P<0.05) than in the casein induced group. The casein induced group had a higher LDL content (P<0.05) than the casein control group.
The flax induced group had a higher serum albumin concentration (P<0.05) than the casein induced group. The casein induced group had a lower albumin content (P<0.05) than the casein control group. The flax control group had a higher serum albumin concentration (P<0.05) than the casein induced group.
When evaluating the serum levels of testosterone, it was observed that the casein induced group and the flax induced group had higher results (P<0.05) when compared to the flax control group. The casein induced group had higher levels of testosterone (P<0.05) than the casein control group and flax induced group.
The flax induced group had significantly higher serum levels of estradiol (P<0.05) when compared to the other groups.
The numerical data are expressed in Table II.
Discussion
In our study, it was found that the levels of HDL in both induced groups (casein and flax) were lower than in the casein control group. This augments the idea that the administration of testosterone reduces serum levels of HDL. These results are in agreement with Tikkanen & Nikkila (1987) that suggested that testosterone increases the activity of the hepatic lipase enzyme (HL) and lipoprotein lipase (LPL) that catabolize HDL27. Similar results of these testosterone effects on HDL levels were reported by Frisch & Sumida(1999) in rats28. Flax did not show any improvement in the HDL profile and other studies have reported similar results. Bloedon et al. (2008) demonstrated the negative effect of the consumption of flax on HDL levels when it was offered at a dosage of 40g/day29. No improvement was verified in HDL levels in rats after the use of flax oil along with a diet rich in fat24.
The effects of flax were clearer on LDL levels. In this study, the LDL levels increased in the casein induced group when compared to the flax induced group. The administration of testosterone increases the levels of LDL on Rhesus monkeys30 and humans1. However, Frisch & Sumida(1999) in their study on rats did not find any significant difference in LDL levels until the seventh week of testosterone administration28. This discrepancy can be explained by the longer duration of our experiment (12 weeks) and form of induction through pellets that conveys a continuous release of the hormone throughout the entire study period.
Flax was able to improve the lipid profile in the group that suffered testosterone induction, decreasing the levels of LDL. The serum levels of LDL in the flax induced group did not have any significant difference to the flax control group, therefore the consumption of flax seems to interfere directly on this indicator and revert the effect of testosterone on LDL. This is in agreement with previous studies with flaxseeds that proved its efficiency in improving lipid profile. Prasad (2005) demonstrated that the use of 40mg/kg of weight/day of SDG associated to a diet that is rich in cholesterol reduces total cholesterol and LDL levels31.
Since normally the serum levels of HDL decrease and the LDL levels increase during treatment with testosterone, the level of triglycerides can suffer alterations6. The increase in triglyceride levels in the casein control group when compared to the other groups is linked to the increase of HDL levels32. Among the control groups, the group that received the flax flour-based meal had lower triglyceride levels than the control group that received the casein-based meal. Another study that supports the effect of flax on triglycerides was the use of 20g of flax during 60 days in hyperlipidemic patients that resulted in the modification of cardiovascular risk factors with a significant decrease of cholesterol, LDL and triglycerides33.
The administration of testosterone did not change the levels of cholesterol. No significant differences between the flax induced group and flax control group was found or between the casein induced group and casein control group either. Similar results were found in rats28. The group that received the flax-based meal as well as the induced group that also received the flax-based meal had lower levels of cholesterol than the other studied groups, suggesting that flax has a direct regulating roll on lipid indicators34.
The effect of flaxseeds on lipid profiles has been credited not only to the flaxseed flour, but also to SDG and flaxseed oil. Riediger et al. (2008) offered flaxseed oil along with a saturated fat-rich diet to male mice and found that a reduction in serum levels of triglycerides and cholesterol, crediting this result to the lower rate of n6:n3 in the diet35. In men, the ingestion of 100 mg of SDG during 12 weeks resulted in a significant decrease of the LDL/HDL cholesterol rate, which is an important predictor of the risk of cardiovascular diseases in hypercholesterolemic men36.
The liver is the only organ that is capable of synthesizing albumin37. Due to its metabolizing function, the liver is one of the main organs that are affected by the abusive use of anabolizing steroids which can evolve from small disorders to cancer38,39. The present study did not evaluated the possible histological and enzymatic alterations regarding to androgenic hyperstimulation. Even so, we observed that the induced group that was fed the flax-based meal, the serum albumin levels did not show significant differences with the casein and flax control groups. This result suggests that the diet with flax also has a regulating effect on liver metabolism. Mello et al.(2012) in their study verified that the supplementation of flax oil not only diminishes serum cholesterol levels but also increases the proportion of α-linolenic acid (C18:3 ω-3) in the liver of rats with a dose-dependent response40.
The rats that suffered androgenic hyperstimulation through subcutaneous implants had higher levels of testosterone which validates the efficiency of the method. However, the induced group fed with the flax-based meal had a lower level of testosterone than the induced group fed with the casein-based meal. The same happened with the flax control group when compared to the control group fed with the casein meal. This dietary effect with flax, even in the group that underwent hormonal induction, suggests that flax may have an anti-androgenic effect similar to soybeans41. In another study, Almeida et al.(2012) verified that the ingestion of canola oil as well as alpha linolenic acid (ALA) rich flax flour decreases testicular mass in rats42. Okuyana et al.(2010) reported a lower serum concentration of testosterone in rats treated during 84 days with a diet that contained 12% of canola oil43.
The individuals that use these steroid hormones show high levels of estradiol (E2)44. These authors suggest that the elevated levels of testosterone are bio-transformed by the aromatase enzyme (present in the liver and fat tissue), increasing estradiol levels. This process probably aided in the increase of estradiol levels visualized in the flax control and flax induced groups, along with the presence of phytoestrogen (SDG) in the flax diet45.
This study suggests that flax can have a direct regulating effect on the levels of cholesterol and LDL in animals submitted to high levels of testosterone, efficiently acting on the maintenance of a normal lipid profile.
Conflict of interest
The authors declare that they have no conflict of interest.
References
1. Kuipers H, Wijnen JA, Hartgens F, Willems SM . Influence of anabolic steroids on body composition, blood pressure, lipid profile and liver functions in body builders. Int J Sports Med 1991; 12(4):413-418. [ Links ]
2. Cunha TS, Cunha NS, Moura MJCS, Marcondes F K. Esteróides anabólicos androgênicos e sua relação com a prática desportiva. Rev Bras Cienc Farm (online) 2004; 40(2):165-179. [ Links ]
3. Silva PRP, Danielski R, Czepielewski MA. Esteróides anabolizantes no esporte. Rev Bras Med Esporte 2002; 8(6):235-243. [ Links ]
4. Sullivan ML, Martinez MC, Gennis P, JE Gallagher. The cardiac toxicity of anabolic steroids. Prog Cardiovasc Dis 1998; 41:1-15. [ Links ]
5. Hartgens F, Rietjens G, Keizer HA, Kuipers H, Wolffenbuttel BH. Effects of androgenic-anabolic steroids on apolipoproteins and lipoprotein (a). Br J Sports Med 2004; 38(3):253-259. [ Links ]
6. Glazer G . Atherogenic effects of anabolic steroids on serum lipid levels. A literature review. Arch Intern Med 1991;151(10):1925-1933. [ Links ]
7. Palatini P, Giada F, Garavelli G, Sinisi F, Mario L, Michieletto M, Baldo-Enzi G . Cardiovascular effects of anabolic steroids in weight-trained subjects. J Clin Pharmacol 1996; 36(12):1132-1140. [ Links ]
8. Sachtleben TR, Berg KE, Cheatham JP, Felix GL, Hofschire PJ. Serum lipoprotein patterns in long-term anabolic steroid users. Res Q Exerc Sport 1997; 68(1):110-115. [ Links ]
9. Margo K, Winn R. Testosterone treatments: why, when, and how? Am Fam Physician 2006; 73(9):1591-1598. [ Links ]
10. Zgliczynski S, Ossowski M, Slowinska-Srzednicka J, Brzezinska A, Zgliczynski W, Soszynski P, Chotkowska E, Srzednicki M, Sadowski Z. Effect of testosterone replacement therapy on lipids and lipoproteins in hypogonadal and eldery men. Atherosclerosis 1996; 121: 35-43. [ Links ]
11. Kadi F, Bonnerud P, Eriksson A, Thornell LE. The expression of androgen receptors in human neck and limb muscles: effects of training and self-administration of androgenic-anabolic steroids. Histochem Cell Biol 2000; 113:25-29. [ Links ]
12. Hsu A, Bruno RS, Lohr CV, Taylor AW, Dashwood RH, Bray TM, Ho E. Dietary soy and tea mitigate chronic inflammation and prostate cancer via NFkB pathway in the Noble rat model. J Nutr Biochem 2011; 22(5):502-510. [ Links ]
13. Zhao C, Moon du G, Park JK. Effect of testosterone undecanoate on hematological profiles, blood lipid and viscosity and plasma testosterone level in castrated rabbits. Can Urol Assoc J 2013; 7(3-4):E221-5.doi: 10.5489/cuaj.507. [ Links ]
14. Singh KK, Mridula D, Rehal J, Barnwal P. Flaxseed: a potential source of food, feed and fiber. Crit Rev Food Sci Nutr 2011; 51(3):210-222. [ Links ]
15. Giada MDE. L. Food applications for flaxseed and its components: products and processing. Recent Pat Food Nutr Agric 2010; 2(3):181-186. [ Links ]
16. Demark-Wahnefried W, Polascik TJ, George SL, Switzer BR, Madden JF, Ruffin MT 4th, et al. Flaxseed supplementation (not dietary fat restriction) reduces prostate cancer proliferation rates in men presurgery. Cancer Epidemiol Biomarkers Prev 2008; 17(12): 3577-3587. [ Links ]
17. Lee J, Cho K. Flaxseed sprouts induce apoptosis and inhibit growth in MCF-7 and MDA-MB-231 human breast cancer cells. In Vitro Cell Dev Biol Anim 2012; 48(4):244-250. [ Links ]
18. Zhao G, Etherton TD, Martin KR, Gillies PJ, West SG, Kris-Etherton PM. Dietary alpha-linolenic acid inhibits proinflammatory cytokine production by peripheral blood mononuclear cells in hypercholesterolemic subjects. Am J Clin Nutr 2007; 85: 385-391. [ Links ]
19. Brzezinsk A, Debi A. Phytoestrogens: the natural selective estrogen receptor modulators? Eur J Obstet Gynecol Reprod Biol 1999; 85(1): 47-51. [ Links ]
20. Hasler CM, Kundrat S, Wool D. Functional foods and cardiovascular disease. Curr Atherosckler Rep 2000; 2:467-475. [ Links ]
21. Kurzer MS, Xu X. Dietary phytoestrogens. Annu Rev Nutr 1997; 17:353-381. [ Links ]
22. Thompson, LU. Potential Health benefits and problems associated with antinutrients in foods. Food Res Int 1993; 26:131-149. [ Links ]
23. Fukumitsu S, Aida K, Ueno N, Ozawa S, Takahashi Y, Kobori M. Flaxseed lignan attenuates high-fat diet-induced fat accumulation and induces adiponectin expression in mice. Brit J Nutr 2008; 100: 669-676. [ Links ]
24. Akpolat M, Kanter M, Topcu-Tarladacalisir Y, Aydogdu N. Protective Effect of Flaxseed Oil on Renal Injury in Hyperlipidaemic Rats: The Effect of Flaxseed Oil on Hyperlipidaemia. Phytother Res 2011; 25(6): 796-802. [ Links ]
25. Bhathena SJ, Ali AA, Mohamed AI, Hansen CT, Velasquez MT. Differential effects of dietary flaxseed protein and soy protein on plasma triglyceride and uric acid levels in animal models. J Nutr Biochem 2002; 13: 684-689. [ Links ]
26. Reeves PG, Nielsen FH, Fahey Jr GCF. AIN-93 purified diet of laboratory rodents: final report of the American Institute of Nutrition ad hoc Writing Committee on the Reformulation of the AIN-76A rodents diet. J Nutr 1993; 123:1939-1951. [ Links ]
27. Tikkanen MJ, Nikkilä EA. Regulation of hepatic lipase and serum lipoproteins by sex steroids. Am Heart J 1987; 113:562-567. [ Links ]
28. Frisch F, Sumida KD. Temporal effects of testosterone propionate injections on serum lipoprotein concentrations in rats. Med Sci Sports Exerc 1999; 31(5):664-669. [ Links ]
29. Bloedon LT, Balikai S, Chittams J, Cunnane SC, Berlin JA, Rader DJ, Szapary PO. Flaxseed and Cardiovascular Risk Factors: Results from a Double Blind, Randomized, Controlled Clinical Trial. J Am Coll Nutr 2008; 279 (1): 65-74. [ Links ]
30. Tyagi A, Rajalakshmi M, Jeyaraj DA, Sharma RS, Bajaj JS. Effects of long-term useof testosterone enanthate. II. Effects on lipids, high and low density lipoproteincholesterol and liver function parameters. Int J Androl 1999;22(6):347-355. [ Links ]
31. Prasad K . Hypocholesterolemic and antiatherosclerotic effect of flax lignan complex isolated from flaxseed. Atherosclerosis 2005; 179:269-275. [ Links ]
32. Aydilek N, Aksakal M. Effects of testosterone on lipid peroxidation, lipid profiles and some coagulation parameters in rabbits. J Vet Med A Physiol Pathol Clin Med 2005; 52(9):436-439. [ Links ]
33. Mandasescu S, Mocanu V, Dăscalita AM, Haliga R, Nestian I, Stitt PA, Luca V. Flaxseed supplementation in hyperlipidemic patients. Nat Rev Med Chir Soc Med Iasi 2005; 109(3):502-506. [ Links ]
34. Cardozo LFMF, Chagas MA, Soares LL, Troina AA, Bonaventura GT. Exposure to flaxseed during lactation does not alter prostate area or epithelium height but changes lipid profile in rats. Nutr Hosp 2010; 25(2):250-255. [ Links ]
35. Riediger ND, Othman R, Fitz E, Pierce GN, Suh M, Moghadasian MH . Low n-6:n-3 fatty acid ratio, with fish- or flaxseed oil, in a high fat diet improves plasma lipids and beneficially alters tissue fatty acid composition in mice. Eur J Nutr 2008; 47:153-160. [ Links ]
36. Fukumitsu S, Aida K, Shimizu H, Toyoda K. Flaxseed lignan lowers blood cholesterol and decreases liver disease risk factors in moderately hypercholesterolemic men. Nutr Res 2010; 30 (7):441-446. [ Links ]
37. Santos NSJ, Draibe SA, Kamimura MA, Cuppari, L. Albumina sérica como marcador nutricional de pacientes em hemodiálise. Rev Nutr 2004; 17(3): 339-349. [ Links ]
38. Campos DR, Yonamine M, Alves MJNN, Moreau RLM. Determinação de esteroides androgênicos anabólicos em urina por cromatografia gasosa acoplada à espectrometría de massas. Braz J Pharm Sci 2005; 41:467-476. [ Links ]
39. Yoshida EM, Karim MA, Shaikh JF, Soos JG, ERB SR. At what price, glory? Severe cholestasis and acute renal failure in an athlete abusing stanozolol. CMAJ 1994; 151:791-793. [ Links ]
40. Melo ILP, Carvalho EBT, Silva AMO, Akiau DM, Mancini-Filho J. Influência do óleo de linhaça sobre o lipidograma no soro e perfil de ácidos graxos poliinsaturados no fígado de ratos. Nutrire 2012; 37:43-43. [ Links ]
41. Ekaluo UB, Udoh PB, Ikpeme EV, Udensi O. Effect of soybean (Glycine max L.) on the hormonal milieu of male rats. Pak J Biol Sci 2011;14(14):752-754. [ Links ]
42. Almeida AFG, Soares da Costa CAS, Gaspar de Moura E, Ferreira Farias Lancetta C, Alves Nascimento-Saba CC. Effects of soybean or canola oil intake on seminiferous tubules structure in young rats. Nutr Hosp 2012; 27(5):1668-1669. [ Links ]
43. Okuyama H, Ohara N, Tatematsu K, Fuma S, Nonogaki T,Yamada K. Testosterone-lowering activity of canola and hydrogenated soybean oil in the stroke-prone spontaneously hypertensive rat. J Toxicol Sci 2010; 35 (5): 743-747. [ Links ]
44. Venâncio DP, Nóbrega ACL, Tufik S, Mello MT. Descriptive Assessment on the Use of Anabolic Steroids and Their Effect on the Biochemical and Neuroendocrine Variables in Practitioners of Resisted Exercise. Rev Bras Med Esporte 2010; 16(3):191-195. [ Links ]
45. Troina AA, Figueiredo MS, Moura EG, Boaventura GT, Soares LL, Cardozo LF, Oliveira E, Lisboa PC, Passos MA, Passos MC. Maternal flaxseed diet during lactation alters milk composition and programs the offspring body composition, lipid profile and sexual function. Food Chem Toxicol 2010; 48:697-703. [ Links ]
![]() Correspondence:
Correspondence:
Mauricio Alves Chagas.
Universidade Federal Fluminense.
Laboratório de Biomorfologia Celular e Extracelular.
Departamento de Morfologia/ Instituto Biomédico.
Rua Hernani Melo, 101, São Domingos, Niterói,
Rio de Janeiro, RJ 24210-130, Brasil.
E-mail: chagas.m@gmail.com
Recibido: 23-I-2014.
1.a Revisión: 03-VII-2014.
Aceptado: 22-VII-2014.