My SciELO
Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Nutrición Hospitalaria
On-line version ISSN 1699-5198Print version ISSN 0212-1611
Nutr. Hosp. vol.33 n.3 Madrid May./Jun. 2016
https://dx.doi.org/10.20960/nh.253
TRABAJO ORIGINAL / Paciente crítico
Critical energy deficit and mortality in critically ill patients
Déficit crítico de energía y mortalidad en pacientes críticos
Marcia Carolina Siqueira-Paese1, Diana Borges Dock-Nascimento1 and José Eduardo de Aguilar-Nascimento2
1Intensive Care Unit. Santa Rosa Hospital. Cuiaba, Brazil.
2Federal University of Mato Grosso. Campus Sinop, Brazil.
3Food and Nutrition Department. Medical School. Federal University of Mato Grosso. Centro Universitario de Varzea Grande (UNIVAG). Brazil.
Financial support: Conselho Nacional de Desenvolvimento Cientifico e Tecnologico (CNPq) of Brazil (Postgraduate scholarship granted to Márcia C.S. Paese, and Research Scholarship granted to Jose E. de Aguilar-Nascimento).
ABSTRACT
Objective: We investigate the influence of caloric and protein deficit on mortality and length of hospital stay of critically ill patients.
Methods: A cohort prospective study including 100 consecutive patients in a tertiary intensive care unit (ICU) receiving enteral or parenteral nutrition. The daily caloric and protein deficit were collected each day for a maximum of 30 days. Energy deficits were divided into critical caloric deficit (≥ 480 kcal/day) and non-critical caloric deficit (≤ 480 kcal/day); and in critical protein deficit (≥ 20 g/day) and non-critical protein deficit (≤ 20 g/day). The findings were correlated with hospital stay and mortality.
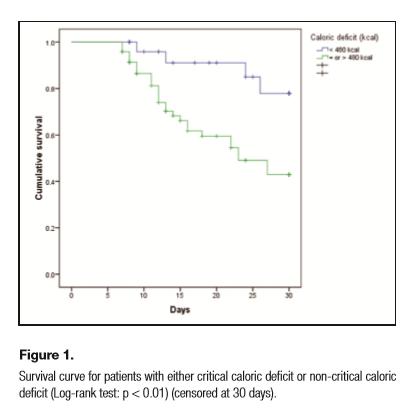
Results: The mortality rate was 33%. Overall, the patients received 65.4% and 67.7% of the caloric and protein needs. Critical caloric deficit was found in 72% of cases and critical protein deficit in 70% of them. There was a significant correlation between length of stay and accumulated caloric deficit (R = 0.37; p < 0.001) and protein deficit (R = 0.28; p < 0.001). The survival analysis showed that mortality was greater in patients with both critical caloric (p < 0.001) and critical protein deficits (p < 0.01). The Cox regression analysis showed that critical protein deficit was associated with higher mortality (HR 0.25, 95% CI 0.07-0.93, p = 0.03).
Conclusions: The incidence of caloric and protein deficit in the ICU is high. Both caloric and protein deficits increase the length of hospital stay, and protein deficit greater than 20 g/day is an independent factor for mortality in critical care unit.
Key words: Critical care. Enteral nutrition. Parenteral nutrition. Energy deficit. Mortality.
RESUMEN
Objetivo: investigar la influencia del déficit de calorías y proteínas en la mortalidad y la duración de la estancia hospitalaria de pacientes críticos.
Métodos: se realizó un estudio prospectivo de cohorte incluyendo 100 pacientes consecutivos en una unidad de cuidados intensivos (UCI) terciaria que recibían nutrición enteral o parenteral. Se registraron diariamente los déficits de calorías y proteínas durante un máximo de 30 días. Los déficit de energía y proteínas fueron divididos en: déficit crítico de calorías (≥ 480 kcal/día), déficit calórico no crítico (≤ 480 kcal/día), déficit crítico de proteínas (≥ 20 g/día) y déficit de proteínas no crítico (≤ 20 g/día). Los resultados se correlacionaron con la estancia hospitalaria y la mortalidad.
Resultados: la tasa de mortalidad fue de 33%. En general, los pacientes recibieron 65,4% y 67,7% de las necesidades calóricas y proteicas. El déficit calórico crítico se encontró en el 72% de los casos y el déficit proteico crítico en el 70% de ellos. Hubo una correlación significativa entre la duración de la estancia con el déficit calórico (R = 0,37; p < 0,001) y el déficit de proteínas (R = 0,28; p < 0,001). El análisis de sobrevida mostró que la mortalidad fue mayor en los pacientes con déficit calórico crítico (p < 0,001) o con déficit proteico crítico (p < 0,01). El análisis de regresión de Cox mostró que el déficit crítico de proteínas se asoció con mayor mortalidad (HR 0.25, IC 95% 0,07-0,93; p = 0,03).
Conclusiones: la incidencia de déficit de calorías y de proteínas en la UCI es alta. Tanto los déficit calóricos como proteicos aumentan la duración de la estancia hospitalaria, y el déficit de proteínas superior a 20 g/día es un factor independiente de mortalidad en la ICU.
Palabras clave: Cuidados críticos. Nutrición enteral. Nutrición parenteral. Déficit energético. Mortalidad.
Introduction
Critically ill patients show high metabolic demands and they frequently have systemic inflammatory response which leads to longer hospital stay, organ failure and increased morbimortality (1-3). Some papers have shown that in critical care settings, underfeeding related to inability to deliver the required nutrients is very common. Repetitive fasting periods, enteral tube complications, and gastrointestinal intolerance are the most frequent reported problems (4-11). A multicentric study showed that the greater quantity of calories and proteins delivered the more significant clinical improvement is found especially in patients with body mass index (BMI) lower than 25 kg/m² or greater than 35 kg/m2 (12).
Energy deficit is common in intensive care but only a few studies have correlated it with mortality among critically ill patients (5,6,11). In this context new data may enhance the perceptions of intensive care physicians on this important issue. Therefore, the aim of this study was to investigate the influence of protein and caloric deficit on both the length of hospital stay and mortality of critically ill patients receiving specialized nutrition therapy.
Material and methods
This was a prospective, cohort study including all the adult patients of both sexes that were admitted to the intensive care unit of the Santa Rosa Hospital in Cuiabá, Mato Grosso (Brazil). The study was approved by the REC (Research Ethical Committee) of the Julio Muller University Hospital under protocol n.o895/CEP - HUJM/2010. Exclusion criteria were: a) length of stay of less than 48 h, b) specialized nutrition therapy for less than seven days and c) oral diet as the first prescription or prior to the first seven days of hospitalization.
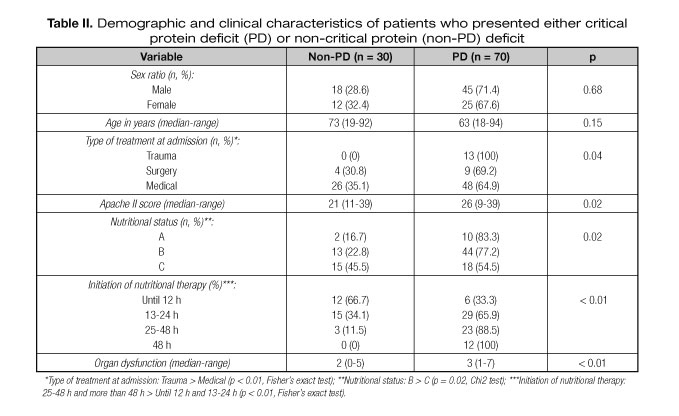
In the first 24 hours after admission, demographic and nutritional data were collected (Table I). Caloric and protein goals, time of initiation of the nutrition therapy (up to 12, 13-24, 25-48 or more than 48 hours after admission), the APACHE II score (13) and the number of dysfunctional organs were also collected (14).
ENERGY CALCULATION
Caloric and protein targets were calculated according to the ESPEN guidelines (20-25 kcal/kg/day and 1.5-2.0 g of protein/kg/day) (15,16). Energy delivered via glucose solutions and intravenous propofol was added to final calculations. Glycemic control was done to maintain glycaemia ranging from 80 to 150 mg/dL. We collected and compared data from the target (amount of calories and proteins calculated from the ESPEN protocol that should be followed), the prescription (amount of calories and protein actually prescribed by the intensivist) and the delivery (amount of calories and proteins received by the patient).
CLINICAL OUTCOME
Mortality during the first 30 days after ICU admission and during all the hospital stay was registered. The length of ICU stay, the appearance of a new organ dysfunction, and the number of days on mechanical ventilation were recorded. The patients were followed up until the 30th day after admission for the energy deficit calculations due to the design of the study. Experienced intensive care physicians who were blind and not participating of the study did all prescriptions and medical decisions.
NUTRITIONAL PROTOCOL
Specialized nutrition therapy was early initiated (first 24 h after admission) in patients unable to intake oral nutrients unless they were hemodynamic unstable. Enteral nutrition (EN) was preferred over parenteral nutrition (PN). Both gastric and post-pyloric routes were prescribed. However, if EN was contra-indicated, impossible or not reaching the nutritional goals after four days, PN was immediately initiated either alone or associated with EN. Target was programmed to the reached on the 3rd day of PN and on the 4th day of EN. The stepwise increase of energy delivery was 1/3 of the goal per day for PN and 1/4 of the goal per day of EN. A wide range of either enteral or parenteral components were prescribed according to the decision of the nutrition team for each individual over time.
DEFICIT CALCULATIONS
We summed up the total amount of calories (kcal) and protein (g) calculated as target (as described above) for each patient during the ICU stay; afterwards we compared with the amount prescribed and the amount actually received. All caloric and protein deficits were registered in daily basis. The total deficit was calculated by summing up all deficits during the ICU stay. The mean deficit per patient was calculated by dividing the total amount of deficit by the number of days of ICU stay. Two levels of energy deficit were considered for analysis: critical caloric deficit defined as the mean caloric deficit above 480 kcal/day and critical protein deficit as the mean protein deficit above 20 g/day. These cutoff points were the 25th percentile of the energy deficit found in all patients.
STATISTICAL METHOD
Sample was calculated estimating that 60% of the patients who received specialized therapy either by enteral or parenteral routes would have a caloric deficit above 25%. For these patients it was estimated that the percentage of complications would be 30% higher. We estimated that for 100 patients it would be necessary to obtain 80% power analysis and 5% of type-1 error. Bivariate comparisons included Student's t tests, Mann-Whitney U, Chi2 test, Pearson's correlation and Fisher's exact tests were used as appropriate. Survival analysis was done and Kaplan-Mayer curves were performed to compare critical deficit and non-critical deficit for mortality using the log-rank test. All subjects were followed overtime either until an event of interest (mortality or discharge) or the end of the study. Drop-outs were censored at the time they dropped out. Distribution of survival times in several subsets of confounding variables such as nutritional status, age, APACHE II score, type of treatment (medical, surgical, or trauma) and time to initiation of nutritional therapy (before or after 24 h of admission) were also analyzed. Cox proportional-hazards regression analysis by enter procedure was used to assess the impact of independent variables on ICU mortality across the time. All variables with p bellow or equal to 0.20 in the log rank test entered the Cox analysis. The continuous variations in caloric and protein deficit were transformed into categorical variables (critical caloric deficit or non-critical caloric deficit, critical protein deficit or non-critical protein deficit) using the mean plus one standard deviation as a cutoff (≥ 480 kcal/day and ≥ 20 g of protein/day). Discrete variables are expressed as counts (percentage) and continuous variables as means ± standard deviation (SD) or median and range. Data from Cox analysis are presented as hazard ratio (HR) and 95% confidence interval (CI). Data were analyzed using SPSS 17.0 (SPSS Inc., Chicago, IL) for Windows.
Results
From April to December 2012, 618 patients were admitted to the ICU. 462 patients were excluded for either having received oral diet or being admitted for less than 48 h. Another 56 patients were ruled out due to short stay (less than 7 days). One-hundred patients (63 males and 37 females) with an average age of 65 (range 18-94) years old remained for analysis.
The median length of ICU stay was 15 (range: 7-96) days. Mechanical ventilation was required at some point of the stay for 86 subjects (median [range]: 13 [3-30] days). During the study, 69 patients presented one new organ failure with a median of one (1-5) new organ failure. The mortality rate in the 30 days period was 33% and during the whole hospital stay was 45%.
NUTRITION VARIABLES
A total of 1772 days of follow-up were observed. In a total of 242 days (13.6%) no nutrients were delivered. During the period of the study 87 patients remained for at least one day (median = 2 [range: 1-10] days) without nutritional therapy. The chosen route was EN in 81%, PN in 7%, and a combination (EN + PN) in 12% of the cases.
CALORIC DEFICIT
Only four (4%) patients did not have a caloric deficit. The patients received 65.4% of the amount of calculated calories. There was a significant decrease (p < 0.01) of total amount of energy provision comparing the target (35,427 ± 19,226 kcal) to the prescribed (26,707 ± 16,699 kcal) and to the delivered (23,187 ± 15,431 kcal) for the patients.
PROTEIN DEFICIT
There was a significant decrease (p < 0.01) of protein intake comparing the calculated (1,652 ± 926 g) to the prescribed (1,294 ± 883 g) and to the delivered (1,118 ± 794 g) for the patients. Ninety-two patients had protein deficit and they received a mean of 67.7% of the total amount of protein calculated.
CRITICAL CALORIC AND PROTEIN DEFICITS
Seventy two patients presented critical caloric deficit (≥ 480 kcal/day) and 70 presented critical protein deficit (≥ 20 g of protein/day). The clinical and demographical characteristics of these patients can be seen in table I and table II. By univariate analysis, critical caloric deficit was most common in patients admitted due to trauma and when nutritional therapy was initiated after 12 h of admission. On the other hand the number of cases of critical protein deficit was greater in the trauma subset, in patients with higher Apache II score, initiation nutritional therapy after the 24 h of admission, and in those with a larger number of organ dysfunctions.
CALORIC DEFICIT ANDLENGTH OS STAY
There was a significant statistical correlation between the length of hospital stay and both the accumulated caloric (R=0.37; p<0.001) and protein (R = 0.28; p < 0.001) deficits.
UNIVARIATE SURVIVAL ANALISYS
There was no statistical significant difference in mortality assessed by log-rank test for comparisons of groups of patients by age (p = 0.12), APACHE II score (p = 0.08), time to initiation of nutritional therapy (p = 0.64), type of treatment (p = 0.82) and nutritional status (p = 0.86). Figure 1 shows the Kaplan-Meier curve for critical caloric and non-critical caloric deficit groups, and figure 2 shows the curve for critical protein or non-critical protein deficit groups. Patients with both non-critical caloric deficit (HR: 0.31 [95% CI 0.13-0.69]) and non-critical protein deficit (HR: 0.31 [95% CI 0.13-0.69]) had a significant lower mortality (p < 0.01).
COX REGRESSION ANALISYS
The Cox regression model included APACHE score, age, critical caloric deficit, and critical protein deficit. The Cox regression analysis showed that non-critical protein deficit (HR 0.25, 95% CI 0.07-0.93, P = 0.03) remained associated with lower mortality (Tabla III).
Discussion
The overall results show that the quantities of calories and proteins delivered to patients were significantly lower than those calculated (target) or prescribed. In fact, more than 90% of the patients received less than they needed. In addition, the impact caused by the above mentioned caloric and protein deficit was a significant factor to increase ICU stay as well as enhance mortality. These findings are relevant and suggest that more nutritional attention to this population of patients is needed.
Caloric and protein deficit in critically ill patients is not uncommon (5,7,12,17,18). Within this context some recent studies have pointed towards the need to follow nutrition protocols aiming at reducing this disparity (19-21) and guidelines (1,15,16,22). Our results show that even following existing guidelines there was still a caloric and protein deficit. A study showed in 22% of 2208 days of observation critically ill patients did not receive any calories (23). Another study showed that in 101 days (15.1%) of 669 observation days, no nutritional supplement was administered (5). Our findings are in agreement with these studies. All together, these numbers are alarming and may be similar in different parts of the world.
Our findings show that survival risk may decrease by 25-30% if caloric/protein deficit is present. In addition the regression analysis showed that protein deficit is an independent cause of mortality in critically ill patients. An earlier study has shown that caloric deficit is associated with complications but not with mortality (5). However, another study with similar findings showed a significant correlation between caloric and protein deficit and mortality, particularly in patients with a BMI below 25 or greater than 35 kg/m2 (12). In another study involving 251 critical patients and a total of 750 days of study, mortality was significantly lower (26.2% vs. 73.3%, p = 0.002) in the group that achieved the caloric target by the 4th day compared with the group that did not achieve the target (24).
Nutritional deficits during the first days after ICU admission have an impact in the outcomes (4,5). Our findings show that all the patients who received nutritional therapy after the first 48 hours of admission presented critical protein deficit. In contrast, few patients who initiated in the first 12 hours of admission presented critical protein deficit. This reinforces the recommendation for early initiation of enteral feeding in critical patients (25-29).
However, this study has limitations. The number of cases in this study is small, the ICU population was not homogeneous and proteins and calories were not calculated using a calorimeter. Nevertheless, the results come from a single hospital, taken by a single researcher and an experienced and well-qualified nutrition therapy team who has worked together for many years took care for all the patients (18,30-31). The nutritional prescriptions closely followed international society recommendations.
Conclusion
The incidence of caloric and protein deficit in ICU is high. Energy deficit increases ICU length of stay and protein deficit greater than 20 g per day is an independent factor of mortality in critically ill patients.
Acknowledgments
We thank the Conselho Nacional de Desenvolvimento Cientifico e Tecnologico (CNPq) for the scholarship given to Márcia Carolina de Siqueira Paese.
References
1. McClave SA, Martindale RG, Vanek VW, et al. Guidelines for the provision and assessment of nutrition support therapy in the adult critically ill patient: Society of Critical Care Medicine (SCCM) and American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.). JPEN J Parenter Enteral Nutr 2009;33:277-316. [ Links ]
2. De Aguilar-Nascimento JE. The role of macronutrients in gastrointestinal blood flow. Curr Opin Clin Nutr Metab Care 2005;8:552-6. [ Links ]
3. De Aguilar-Nascimento JE, Dock-Nascimento DB, Bragagnolo R. Role of enteral nutrition and pharmaconutrients in conditions of splanchnic hypoperfusion. Nutrition 2010;26:354-8. [ Links ]
4. Engel JM, Muhling J, Junger A, et al. Enteral nutrition practice in a surgical intensive care unit: what proportion of energy expenditure is delivered enterally? Clin Nutr 2003;22:187-92. [ Links ]
5. Villet S, Chiolero RL, Bollmann MD, et al. Negative impact of hypocaloric feeding and energy balance on clinical outcome in ICU patients. Clin Nutr 2005;24:502-9. [ Links ]
6. Dvir D, Cohen J, Singer P. Computerized energy balance and complications in critically ill patients: an observational study. Clin Nutr 2006;25:37-44. [ Links ]
7. Soguel L, Revelly JP, Schaller MD, et al. Energy deficit and length of hospital stay can be reduced by a two-step quality improvement of nutrition therapy: the intensive care unit dietitian can make the difference. Crit Care Med 2012;40:412-9. [ Links ]
8. Nozaki VT, Peralta RM. Adequacy of nutritional support provided by enteral feeding: a comparison of two hospitals. Rev Nutr 2009;22:341-50. [ Links ]
9. Stapleton RD, Jones N, Heyland DK. Feeding critically ill patients: what is the optimal amount of energy? Crit Care Med 2007;35(9 Suppl):S535-40. [ Links ]
10. McClave SA, Sexton LK, Spain DA, et al. Enteral tube feeding in the intensive care unit: factors impeding adequate delivery. Crit Care Med 1999;27:1252-6. [ Links ]
11. Rubinson L, Diette GB, Song X, et al. Low caloric intake is associated with nosocomial bloodstream infections in patients in the medical intensive care unit. Crit Care Med 2004;32:350-7. [ Links ]
12. Alberda C, Gramlich L, Jones N, et al. The relationship between nutritional intake and clinical outcomes in critically ill patients: results of an international multicenter observational study. Intensive Care Med 2009;35:1728-37. [ Links ]
13. Knaus WA, Draper EA, Wagner DP, et al. APACHE II: a severity of disease classification system. Crit Care Med 1985;13:818-29. [ Links ]
14. Vincent JL, Moreno R, Takala J, et al. The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. Intens Care Med 1996;22:707-10. [ Links ]
15. Kreymann KG, Berger MM, Deutz NE, et al. ESPEN Guidelines on Enteral Nutrition: Intensive care. Clin Nutr 2006;25:210-23. [ Links ]
16. Singer P, Berger MM, Van den Berghe G, et al. ESPEN Guidelines on Parenteral Nutrition: intensive care. Clin Nutr 2009;28:387-400. [ Links ]
17. Heyland DK, Cahill N, Day AG. Optimal amount of calories for critically ill patients: depends on how you slice the cake! Crit Care Med 2011;39:2619-26. [ Links ]
18. Dock-Nascimento DB, Tavares VM, de Aguilar-Nascimento JE. Evolution of nutritional therapy prescription in critically ill patients. Nutr Hosp 2005;20:343-47. [ Links ]
19. Heyland DK, Cahill NE, Dhaliwal R, Wang M, Day AG, Alenzi A, et al. Enhanced protein-energy provision via the enteral route in critically ill patients: a single center feasibility trial of the PEP uP protocol. Crit Care 2010;14:1-14. [ Links ]
20. Miller KR, Kiraly LN, Lowen CC, et al. CAN WE FEED? Amnemonic to merge nutrition and intensive care assessment of the critically ill patient. JPEN J Parenter Enteral Nutr 2001;35:643-59. [ Links ]
21. Casaer MP, Hermans G, Wilmer A, et al. Impact of early parenteral nutrition completing enteral nutrition in adult critically ill patients (EPaNIC trial): a study protocol and statistical analysis plan for a randomized controlled trial. Trials 2011;24:12-21. [ Links ]
22. Ortiz Leyba C, Montejo González JC, Vaquerizo Alonso C. Guidelines for specialized nutritional and metabolic support in the critically-ill patient: update. Consensus SEMICYUC-SENPE: septic patient. Nutr Hosp 2011;26:67-71. [ Links ]
23. Krishnan JA, Parce PB, Martinez A, et al. Caloric intake in medical ICU patients: consistency of care with guidelines and relationship to clinical outcomes. Chest 2003;124:297-305. [ Links ]
24. Petros S, Engelmann L. Enteral nutrition delivery and energy expenditure in medical intensive care patients. Clin Nutr 2006;25:51-9. [ Links ]
25. Marik PE, Zaloga GP. Early enteral nutrition in acutely ill patients: a systematic review. Crit Care Med 2001;29:2264-70. [ Links ]
26. Montejo JC, Grau T, Acosta J, et al. Multicenter, prospective, randomized, single-blind study comparing the efficacy and gastrointestinal complications of early jejunal feeding with early gastric feeding in critically ill patients. Crit Care Med 2002;30:796-800. [ Links ]
27. Artinian V, Krayem H, DiGiovine B. Effects of early enteral feeding on the outcome of critically ill mechanically ventilated medical patients. Chest 2006;129:960-7. [ Links ]
28. De Aguilar-Nascimento JE, Kudsk KA. Early nutritional therapy: the role of enteral and parenteral routes. Curr Opin Clin Nutr Metab Care 2008;11:255-60. [ Links ]
29. Doig GS, Heighes PT, Simpson F, et al. Early enteral nutrition, provided within 24 h of injury or intensive care unit admission, significantly reduces mortality in critically ill patients: a meta-analysis of randomised controlled trials. Intensive Care Med 2009;35:2018-27. [ Links ]
30. De Aguilar-Nascimento JE, Bicudo-Salomao A, Portari-Filho PE. Optimal timing for the initiation of enteral and parenteral nutrition in critical medical and surgical conditions. Nutrition 2012;28:840-3. [ Links ]
31. De Aguilar-Nascimento JE, Prado-Silveira BR, Dock-Nascimento DB. Early enteral nutrition with whey protein or casein in elderly patients with acute ischemic stroke: a double-blind randomized trial. Nutrition 2011;27:440-4. [ Links ]
![]() Correspondence:
Correspondence:
José Eduardo de Aguilar-Nascimento.
Rodovia Helder Cândia 2577,
Condomínio Country, casa 15.
78048-150 Cuiabá-MT. Brazil
e-mail: aguilar@terra.com.br
Received: 03/01/2016
Accepted: 05/02/2016