INTRODUCTION
Osteoporotic fractures are a major health problem in developed countries, and are among the most frequent causes of disability and medical costs worldwide (1). Up to 20 % of patients die within the first year following hip fracture (2).
Vitamin D deficiency is one of the main determinants of osteoporotic fractures, producing an imbalance in bone remodeling, contributing to bone mass loss and fracture, reducing intestinal absorption of calcium (especially in subjects with low-to-moderate calcium intake), increasing parathyroid hormone (PTH) levels, and stimulating bone resorption (3). Vitamin D coordinates the functions of osteoclasts and osteoblasts in the regulation of bone remodeling (4).
An association has been demonstrated between vitamin D deficiency and increased fracture risk, independent of other risk factors (5), and between the degree of vitamin D deficiency and the severity of osteoporotic hip fracture (OHF) (6). In contrast, a recent meta-analysis concluded that vitamin D supplementation did not reduce fracture risk (7), although some authors have questioned this conclusion and suggested possible research biases (8); the conclusion could also be due to the fact that an insufficient dose of vitamin D was administered.
Vitamin D is involved in all stages of healing and recovery after a fracture: it modifies the expression of the cytokines interleukin 1 (IL-1), interleukin 6 (IL-6), and TNF-α during the initial inflammation phase, both in vitro and in vivo; it influences callus formation (soft and hard stages); and it participates in bone remodeling (9). Various metabolites of vitamin D are increased at the fracture site, and the administration of supplemental vitamin D after a fracture could assist the healing process (10). The extra-skeletal effects of vitamin D deficiency on cognitive status and immunity, among other factors (11), could also have an impact on the clinical outcomes of these patients.
The behavior of vitamin D levels during the early post-OHF phase has not been fully elucidated, but it may depend on the pre-fracture vitamin D status of patients (12). It has been reported that serum vitamin D levels decrease during the fracture healing process after traumatic or surgical bone injury (13), but it is not known whether these changes in vitamin D levels influence clinical outcomes.
The objectives of this study were to evaluate: a) vitamin D levels in patients hospitalized for hip fracture at admission and 8 days later; and b) the relationship of vitamin D levels with clinical outcomes (length of hospital stay and mortality) and with cognitive and functional status.
METHODS
STUDY DESIGN
This prospective study consecutively enrolled osteoporotic hip fracture patients aged over 65 years admitted to the Orthopedics Department of Jaén University Hospital (Spain, latitude 37º 46' 0”) between 2013 and 2016. Bias due to differences in sunlight exposure was mitigated by recruiting during the Spring and Autumn (March, April, October and November).
Inclusion criteria were: a) low-energy hip fracture (caused by fall from standing height or lower), and b) signed consent to participation in the study. Exclusion criteria were: a) pathological or atypical fracture; b) high-impact fracture (trauma, fall down multiple steps, fall from a height > 30 cm); c) periprosthetic fracture; d) neoplastic disease; e) moderate or severe kidney disease; f) calcium and/or phosphorus metabolism disorder; g) all types of secondary osteoporosis except for vitamin D deficiency; h) malabsorption syndrome and/or ileal resection; and i) current treatment with antiepileptics, rifampin, cyclophosphamide, bisphosphonates, other antiresorptive agent, or sodium fluoride.
All participants were interviewed within 24 hours post-admission. Data were gathered on sex; age; residence; cognitive status as assessed by the Mini-Mental State (MMS) exam (14); functional status as evaluated by the Activities of Daily Living (ADL) questionnaire (15); previous morbidity (Charlson index) (16); pre-anesthetic risk (American Society of Anesthesiologists [ASA] score) (16); medication received at the time of the fracture or before, with a specific question on oral anticoagulants; and nutritional status according to the long-form Mini-Nutritional Assessment (MNA) questionnaire (17). Dietary intake was assessed by administering a 24-hour dietary recall on three non-consecutive days, and calculating the average daily intake.
During their hospital stay, patients did not receive any anti-osteoporosis medication, calcium or vitamin D supplementation.
BLOOD PARAMETERS
Two fasting venous blood samples were drawn: the first within 24 hours of admission and the second on day 8 post-admission, usually 6 days post-surgery (the length of hospital stay was longer than 8 days in all patients). Blood counts and biochemical parameters, including serum calcium (corrected for albumin), serum phosphorus, albumin, IL-6, 25OHD3, and intact PTH (iPTH), were determined. Plasma 25OHD3 was analyzed by chemiluminescence using a Liaison Day Sorin® analyzer. 25OHD3 levels ≥ 30 ng/ mL were considered adequate; levels were deemed inadequate when they were within the range 20-29 ng/mL, and deficient when < 20 ng/mL (18). Serum iPTH was analyzed by chemiluminescence using an Access 800® Unicel DXL analyzer (reference range: 15.0- 88.0 pg/mL).
CLINICAL VARIABLES
Data were gathered on fracture type, dietary intake, length of preoperative stay, type of surgery, complications (nosocomial infections and severe anemia requiring transfusion), and clinical outcomes (length of hospital stay and mortality).
STATISTICAL ANALYSIS
A t-test and analysis of variance were used to compare continuous variables. Factors influencing the length of hospital stay were evaluated by univariate analysis and by multiple linear regression analysis, adjusting for potential confounders. Data on preoperative and total hospital stays were non-normally distributed, and were therefore logarithmically transformed. The Stata 14 SE (College Station, TX, USA) package was used for data analyses.
RESULTS
The patients (81.8 % female, mean age of 84.0 years) mostly lived in the community (86.4 %), and only 13.6 % were institutionalized. Data on their cognitive status (MMS), dependence (ADL), nutritional risk (MNA), comorbidities, preoperative stay, type of surgery, need for transfusion, hospital stay, and mortality are included in table I. A major degree of dependence was observed in 66.7 % of the patients, who had ADL index scores of E, F, G, or H, indicating severe or total dependence for activities of daily living.
Table I. Demographic and clinical characteristics of the included population at the beginning of the study
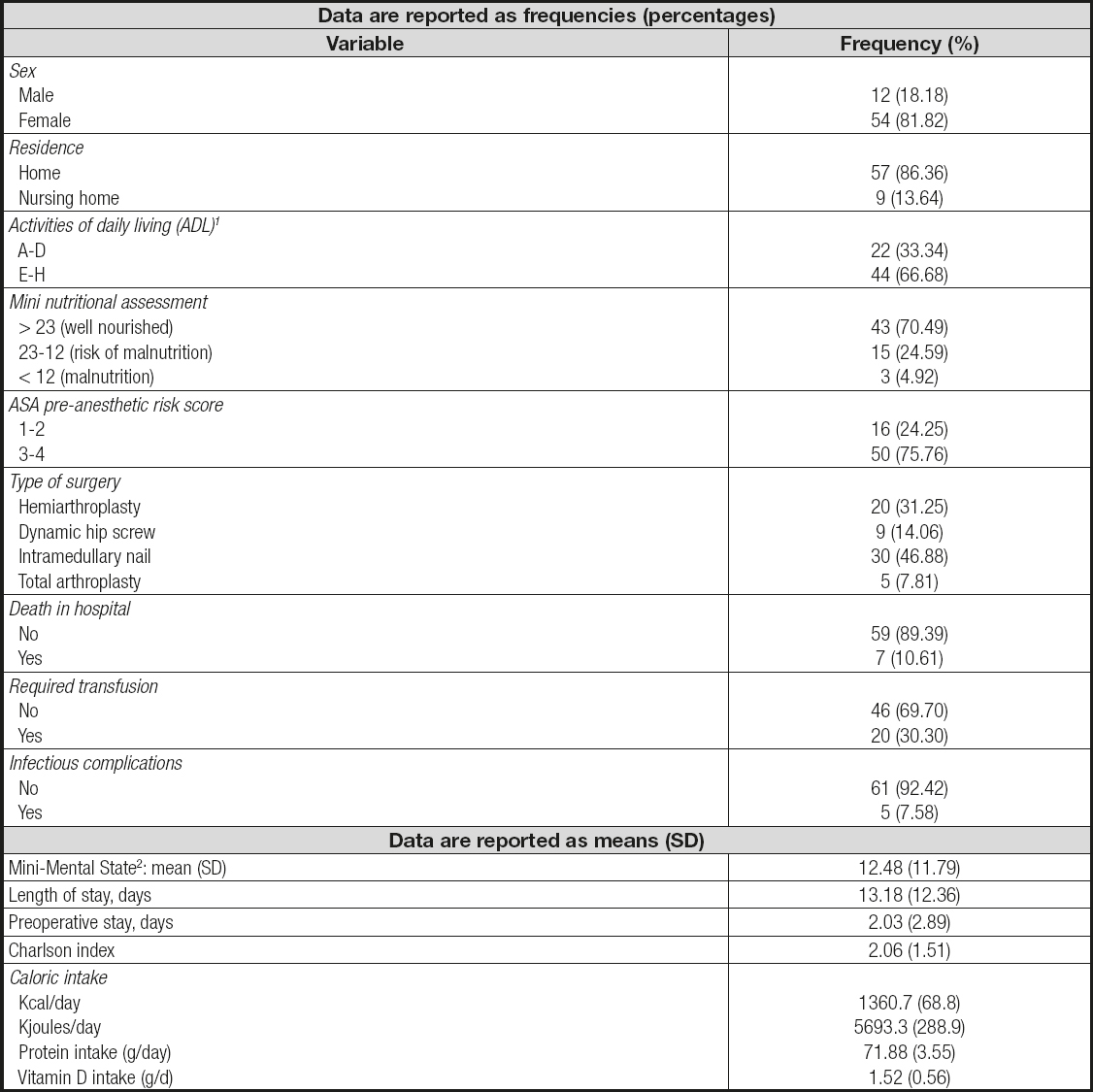
1Patients in categories A, B, C, and D are considered independent or moderately dependent for ADL. Patients in categories E, F, G and H are considered severely or totally dependent for ADL.
2Mini-Mental State (cutoff point for abnormality: < 23). SD: standard deviation
A normal nutritional status (determined by MNA) was found in 70.5 % of the patients. The ASA anesthetic risk was class three in 54.6 % and class four in 21.2 %. The mean Charlson index score (comorbidities) was 2.1; mean preoperative length of stay was 2.0 days, and total hospital stay was 13.2 days. Five patients (7.6 %) showed signs of nosocomial infection during their hospital stay, and seven (10.6 %) died before hospital discharge. The mean pre-admission caloric intake was 1,361 kcal/day, mean protein intake was 71.9 g/ day, and mean vitamin D intake was 1.52 g/day (608 IU).
The regression analysis showed no relationship between vitamin D intake at admission [b coefficient (standard error of the mean (SEM)] of 0.43 [0.44], p = 0.33; data not shown in a table). Table II reports the results for bone metabolism parameters and energy and protein intakes at admission according to the patients' ADL-assessed functional status. Although it may be observed that serum 25OHD3 levels were lower in those with worse functional status, the difference was not statistically significant. No significant differences in other serum markers were observed. Table III displays the results for bone metabolism and energy and protein intakes classified according to MMS-assessed cognitive status, showing significantly lower serum 25OHD3 levels in patients with worse cognitive status (MMS score < 24, p = 0.04). No significant differences were found for the other bone metabolism markers or for IL-6.
Table II. Bone metabolism parameters and energy and protein intakes in the first 24 h of admission according to functional status as assessed by the Activities of Daily Living (ADL) tool

Low: corresponds to patients in categories A, B, C, and D, i.e., independent or moderately dependent for ADL; High: corresponds to patients in categories E, F, G, and H, i.e., severely or totally dependent for ADL; 25OHD3: 25-hydroxyvitamin D3; iPTH: intact PTH; IL-6: interleukin 6.
Table III. Bone metabolism parameters and energy and protein intakes in the first 24 h of admission according to cognitive status as assessed by the Mini Mental State (MMS) examination

MMS score < 10 = moderate to severe dementia; 10-23 = mild dementia; 24-35 = no cognitive impairment; 25OHD3: 25-hydroxyvitamin D3; iPTH: intact PTH; IL-6: interleukin 6.
Table IV includes the mean differences in bone metabolism parameters between blood samples drawn < 24 h and 8 days post- admission. Admission 25OHD3 levels were low, with a mean (SEM) of 12.04 (1.03) ng/mL. During hospital stay, there was a significant decrease in serum 25OHD3, iPTH, and IL-6 levels (p < 0.001, p = 0.015, and p < 0.001, respectively) and a significant increase in albumin-corrected calcium levels (p = 0.03). Table V shows the relationships of study variables with the length of hospital stay. The difference in 25OHD3 levels between days 1 and 8 post-admission was significantly related to the length of hospital stay, with a greater decrease being associated with a longer stay in both the simple (p = 0.045) and multivariate (p = 0.42) analyses. No significant relationship was observed with the other variables (preoperative length of stay or differences in calcium, phosphorus, iPTH, or IL-6 levels). The only variable significantly associated with longer hospital stay was the decrease in 25OHD3 levels between days 1 and 8 post-admission.
Table IV. Bone metabolism parameters on days 1 and 8 after hospital admission

25OHD3: 25-hydroxyvitamin D3; iPTH: intact PTH; IL-6: interleukin 6; SEM: standard error of the mean.
Table V. Relationship between bone metabolism parameters and length of hospital stay (linear regression analysis)

Multivariate analysis: ajusted for baseline values, preoperative stay, ADL, MMS, energy intake (kcal), and albumin. Diff: difference between days 1 and 8; 25OHD3: 25-hydroxyvitamin D3; iPTH: intact PTH; IL-6: interleukin 6; SEM: standard error of the mean.
Given the low number of deaths (n = 7), it was not possible to establish a significant relationship between bone parameters and hospital mortality (results not shown).
Stratification by sex showed similar trends amongst women and men, although there was a loss of statistical significance for corrected serum calcium, phosphorus, iPTH, and IL-6 values in males because of the much smaller number of men.
DISCUSSION
Our study showed a high prevalence of vitamin D deficiency among elderly patients with osteoporotic hip fracture. A significant decline in 25OHD3, IL-6, and iPTH levels was observed during the first eight days after hip fracture, and a greater decrease in 25OHD3 was significantly associated with longer hospital stay. Serum 25OHD3 levels were associated with MMS-assessed cognitive status but not with ADL-evaluated functional status.
The main limitations of our study were the small sample size and the low number of deaths, providing inadequate statistical power to detect significant associations with this outcome. In addition, IL-6 could not be measured in some elderly patients because it was not possible to draw a sufficient amount of blood for this purpose.
In accordance with previous reported values in Mediterranean people (19), we observed low 25OHD3 levels in our patients. In addition, we found no association between vitamin D intake and serum vitamin D levels. Although we observed a reduction in serum calcium between days 1 and 8 post-admission, this became a significant increase after adjustment for serum albumin, which decreased between days 1 and 8. This finding can be explained by the immobility of patients and an increase in bone resorption during the early post-fracture phase (12,20).
An interesting finding in our study was the significant decline in serum 25OHD and IL-6 levels during hospital stay. Vitamin D is known to induce immune response by inhibiting pro-inflammatory cytokines such as IL-6, TNF-a, and IL-17 (21 22-23). Mellenthim et al. described a U-shaped association between 25OHD3 and high-sensitivity CRP (hs-CRP) in the presence of inflammation; they found a reduction in hs-CRP with lower 25-OHD3 levels until a nadir of 21-25 ng/mL was reached, then observing an increase with 25OHD3 levels above 25 ng/mL (24). Srikanth et al. reported an inverse association between 25OHD3 and IL-6 levels in older men (25), and vitamin D is known to play a key role in fracture consolidation and orthopedic surgery outcomes (26). In agreement with our results, two studies have reported a reduction in 25OHD after orthopedic surgery in patients with knee (13) and tibial or femoral shaft (27) fractures, which might be related to the inflammation produced by the procedure (28). In addition, Reid et al. (13) described a decline in vitamin D levels that persisted after normalization by increased plasma CRP; they also proposed that the fall in vitamin D may be due to an increased uptake of its metabolites by callus bone during the post-fracture healing period (13).
IL-6 has been associated with worse clinical outcomes after hip fracture (29,30), reaching peak levels at 72 hours post-fracture (12).
In the present study we observed a significant reduction of both IL-6 serum levels at 8 days after the fracture, indicating that the acute inflammation induced by the bone tissue injury and by surgery decreased within a few days. In addition, serum 25OHD3 levels remained low at the end of the study, which may correspond to a greater consumption of this vitamin at the fracture site (10,13). In contrast to our results, Briggs et al. observed no fluctuation in serum 25OH levels after long-bone fracture in humans (31). However, a longitudinal study of 205 women with hip fractures reported a fall in 1,25(OH)2D3 levels from day 3 to day 10, followed by a gradual rise over the next 12 months (32).
Our observation of a decline in 25OHD3 levels is relevant because of the possible consequences of vitamin D deficiency on the recovery of patients. Vitamin D promotes consolidation at the fracture site by directly stimulating osteoblasts, which have vitamin D receptors (VDRs) and express the 1-a hydroxylase enzyme (CYP27B1). This leads to the endogenous synthesis of 1,25(OH)2D3, which coordinates the functions of both osteoclasts and osteoblasts, and controls bone remodeling (4). Clinical studies have reported that vitamin D supplementation favors consolidation in vitamin D-deficient patients (12,33).
The vitamin D deficiency observed in our patients at admission, and the decline in serum vitamin D levels during their hospital stay, might delay the early stage of fracture healing. In fact, a greater decline in these levels was associated with longer hospital stay. Vitamin D deficiency has previously been associated with clinical outcomes (34). Low plasma vitamin D levels have been associated with a longer delay in the discharge of critically ill patients (35), and with greater length of stay, mortality, and surgical intensive care unit costs (36). They have also been related to worse clinical outcomes (37) and increased comorbidities requiring hospital treatment, including secondary hyperparathyroidism (SHPT) (38). Indeed, researchers have associated higher 25OHD· levels with the recovery of walking ability after a fracture (39), and lower levels with worse clinical outcomes (19). However, another study of 100 patients with hip fractures observed no relationship between vitamin D levels at admission and hospital stay (40,19). Besides vitamin D deficiency, SHPT was independently associated with longer hospital stay and increased mortality (41). In the present study, we observed a significant reduction in iPTH during hospital stay, despite the decline in serum vitamin D. This may be explained in part by the significant increase in albumin-corrected serum calcium levels, and in part by a state of so-called “functional hypoparathyroidism” (20).
In another study, vitamin D levels < 20 ng/mL or > 60 ng/mL were associated with increased mortality in hospitalized patients (42), and an association has been found between lower levels and increased mortality in patients with hip fracture (43,44). In the present study no significant association was found, which may be attributable to the small number of deaths (n = 7) and consequent lack of statistical power.
We found a statistically significant association between 25OHD3 levels and MMS-assessed cognitive status. Vitamin D levels have previously been related to dementia, Parkinson's disease (45), and mild cognitive impairment (MCI). Thus, Anweilwer et al. associated low 25OHD3 levels with MCI in 95 elderly people without dementia (46), and a longitudinal study found that severe vitamin D deficiency was independently associated with a future risk of MCI and dementia (47). Vitamin D deficiency was also associated with worse cognitive status and depression after traumatic brain injury (48). Vitamin D has been described as playing an important extra-skeletal role in the nervous tissue, among others (11). Finally, different vitamin D receptor (VDR) gene polymorphisms in the brain have been associated with cognitive impairment, Parkinson's disease, and multiple sclerosis (49).
In conclusion, 25OHD3 levels were inadequate in patients with osteoporotic hip fracture within 24 h of their hospitalization, and then significantly declined over the next eight days. A greater decline in vitamin D levels was associated with a longer hospital stay, and lower levels were associated with a worse MMS-assessed cognitive level. No association was found with ADL-evaluated functional status. These findings support the administration of vitamin D supplementation during the hospital stay of patients admitted for low-energy hip fracture. Further research is warranted to establish a definitive conclusion on this issue.














