Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO  Similares en Google
Similares en Google
Compartir
Revista Española de Enfermedades Digestivas
versión impresa ISSN 1130-0108
Rev. esp. enferm. dig. vol.96 no.5 Madrid may. 2004
| ORIGINAL PAPERS |
Influence of the surgical manipulation of the colon in colonic induced carcinogenesis
in rats
J. F. Noguera Aguilar, I. Amengual Antich1, A. Plaza Martínez2, C. Tortajada Collado3, J. M. Morón Canis3 and J. J. Pujol Tugores
General Surgery Department. Son Llàtzer Hospital. Son Ferriol. 1Pathology Department. Manacor Hospital Foundation. Manacor.
2Vascular Surgery Department. Dr. Peset University Hospital. Valencia. 3Son Dureta University Hospital. Palma de Mallorca
ABSTRACT
Aim: to investigate the influence of different experimental manipulations in a model of colonic experimental carcinogenesis with pharmacological induction in the rat.
Experimental design: a total of 90 Sprague-Dawley male rats, divided into three groups, were used: non-surgical (n = 30); surgical with colonic trauma (n = 20), and surgical with colo-colonic anastomosis (n = 40). Carcinogenic induction was carried out with 1-2 dimethylhydrazine dihydrochloride. Colonic adenocarcinomas were identified and the number of tumors, as well as tumoral surface and percentage of tumoral surface was established. One-way ANOVA and Chi-square were employed for the statistical analysis.
Results: the number of tumors was greater in the surgical group than in the control group, and tumors preferentially develop-ed around the manipulated colon. Surface and tumoral percentage were greater in the surgical group than in the control group, being also greater in the anastomosis group than in the group with colonic trauma. Within anastomosis groups, a greater tumor surface and percentage was found in the group with titanium than in the group with reabsorbable material.
Conclusions: the experimental manipulation of the colon in rats enhances drug-induced colon carcingenesis. The creation of an anastomosis further increases the carcinogenic process compared with simulated anastomosis. This process is also enhanced by the quantity of suture material included in the anastomosis, and by the non-reabsorbable nature of the materials used in the anastomotic line.
Key words: Cancer. Carcinogenesis. Colon. Anastomosis. Rat.
Noguera Aguilar JF, Amengual Antich I, Plaza Martínez A, Tortajada Collado C, Morón Canis JM, Pujol Tugores JJ. Influence of the surgical manipulation of the colon in colonic induced carcinogenesis in rats. Rev Esp Enferm Dig 2004; 96: 322-330.
Recibido: 17-09-03.
Aceptado: 24-11-03.
Correspondencia: José Francisco Noguera Aguilar. Avda. Antonio Maura, 91, bajo 11. 07009 Pont d'Inca. Baleares. e-mail: jnoguera@hsll.es. Tel. 971 60 82 44
INTRODUCTION
Colorectal cancer is the most common digestive cancer in Western Europe and the United States (1), and the second leading cause of death in the Western world (2). In our country, it has an incidence of 20-30 cases per 100,000 inhabitants per year, and is responsible for 20% of deaths caused by malignancies (3,4). Seventy to 80% of patients with colorectal cancer will be candidates to surgery with a curative purpose; in the remaining 20-30% only palliative surgery will be indicated (5,6).
The locoregional recurrence of colorectal cancer is an important problem within the overall recurrence of tumoral illness (7-10). For late anastomotic recurrences, the most adequate explanation may be a metachronous carcinogenesis in the anastomotic zone. This susceptibility of the anastomosis regarding the development of metachronous tumors or local recurrence seems to result from an increased cell proliferation at colonic crypts neighboring the anastomosis area, and to a chronic effect by suture materials employed (11-14).
The importance of this topic is such that experimental standard models permit to assess the results of different manipulations in order to alter the natural history of locoregional recurrences in colorectal cancer (15,16).
The aim of the study was to assess the influence of experimetal colonic manipulation in a model of colonic chemical-induced carcinogenesis in rats. So we aim to determine which experimental maneuvers have a greater influence on this model of carcinogenesis, whether the colonic trauma or the creation of a colo-colonic anastomoses, and to assess the influence of the amount and nature of suture material on the carcinogenesis.
MATERIAL AND METHODS
Ninety-nine Sprague-Dawley male rats were used (Criffa, Spain), with a mean weight of 185 g. They were divided into three groups: non-surgical or control group (n = 30); surgical with colonic trauma (n = 20), and surgical with colo-colonic anastomosis (n = 40). In the latter, we differentiate four types of manipulation in the making of the anastomosis: with continuous suture (n = 10), interrupted suture (n = 10), interrupted suture and addition of reabsorbable material (n = 10), and interrupted suture and addition of non-reabsorbable material (n = 10).
Non-absorbible sutures (polyvynildifluoroethylene, monofilament 5/0, Lorca-Marín, Spain) were used for colo-colonic anastomoses. For the addition of reabsorbable material in the anastomotic line, 8 mg of polyglycolic acid in two laminar fragments were used; for the addition of non-reabsorbable material, 8 mg of laminar titanium in two fragments were used.
The carcinogenic induction of colonic neoplasms was carried out with 1-2 dimethylhydrazine dihydrochloride (Sigma-Aldrich, Spain).
Dietary and environmental conditions
Environmental conditions in the animal storage area were: 12 h/12 h light/dark cycle (light from 8:00 am - 8:00 pm); uniform temperature at 22 ± 2 ºC, and relative humidity of 60-70%. The diet provided was a maintenance diet A.04 (Panlab, Spain).
The study complied with guidelines established by "Royal Decree" 223/1998, and European Directive 86/609/EEC on the protection of animals used in experimentation.
Anesthetic technique
A solution of ketamine, atropine and diazepam (50, 10, 40%, respectively) was administered intramuscularly at a dose of 4 ml/kg.
Surgical technique
A midline laparotomy of 3-4 cm in length was performed in the lower part of the abdomen, identifying the site of colonic manipulation at 4 cm from the anus. In the group with colonic manipulation, a trauma was induced in the first group (n = 20) and a colo-colonic end-to-end anastomosis was made in the other group (n = 40).
Colonic trauma consisted of a colonic wall trauma simulating a colo-colonic anastomosis; it was performed with eight equidistant passes using the needle and thread with which anastomoses were to be carried out, through the whole thickness of the colonic wall, but without colonic section or leaving suture material in the colonic wall. In the colo-colonic anastomosis group, a complete colonic section was carried out with a subsequent suture using non-reabsorbable 5/0 material. Eight points were equidistantly placed and the intraoperative watertightness of the anastomosis was checked using transrectal pressure.
In the groups in which additional material was added, reabsorbable (polyglycolic acid) or non-reabsorbable (titanium), this was included in the form of two 4 mg filaments lateral to the mesenteric margin, which were anchored in the suture point and thus remained within the colonic wall.
Carcinogenic induction, follow-up and sacrifice
After eight postoperative days, colonic tumors were induced with 1-2-dimethylhydrazine dihydrochloride (DMH) by administering 18 weekly subcutaneous injections at a weekly dose of 25mg/kg of weight. Animals were examined weekly, and special attention was paid to their weight, abdominal perimeter, presence and quality of stools, and presence of rectal bleeding. All animals were sacrificed in postoperative week 20, and their colon was searched for colonic tumors. Animals were killed using an anesthetic overdose, and the entire colon was removed. The colon was divided into two pieces: perianastomotic colon (1 cm above and below the anastomotic line), and the remainder of the colon. Pieces were fixed in a 10% formaldehyde solution for histologic examination.
Examination of colonic tumors
Three parameters were determined: number of tumors, tumoral area, and tumoral percentage (percentage of colonic area occupied by tumoral tissue). Only adenocarcinomas were included in the study.
Statistical analysis
Data obtained were analyzed using the SPSS software program. The statistical analysis was carried out using ANOVA models and Chi-square tables. For the comparison of mean values the ANOVA unifactorial test was used, together with LSD test plus Scheffé or Bonferroni tests.
RESULTS
Mortality
Overall mortality in the study included 2 animals out of 90 animals studied, both within the anastomosis group. These deaths resulted from colonic occlusion by induced neoplasms.
Alterations in colonic transit
Alterations of the gastrointestinal transit because of colonic occlusion occurred in 3 animals (3.33%). Two of them died and a postmortem study was performed. Third animal sacrificed because of colonic occlusion. Colonic tumors were the cause of intestinal occlusion.
Number of tumors
A total of 185 colonic adenocarcinomas were obtained from the 90 animals (2.05 tumors per animal). In the group without surgery, a total of 25 tumors developed (X = 0.83 tumors per animal); in the group with colonic trauma without anastomosis, 44 tumors were found (X = 2,2), whereas in the group with colo-colonic anastomosis, a total of 116 tumors were collected (X = 2.9). This difference in mean tumor count between study groups was significant (Fig. 1, p < 0.0005, ANOVA, LSD and Bonferroni tests).
Regarding colonic tumor location, this varied amongst groups. In the groups without surgery, 64% of tumors developed in the proximal colon, whereas in the group with colonic trauma without anastomosis, 68.18% of tumors developed in the proximal colon. When colo-colonic anastomosis was performed, 59.49% of tumors developed in the perianastomotic area.
Tumoral surface area
In all, 79.1 cm2 of tumoral colonic area were obtained. In the group without surgery 9.13 cm2 developed (0.30 cm2 per animal), while in the group with colonic trauma 15.64 cm2 were obtained (X = 0.78 cm2 per animal), and in the group with colo-colonic anastomosis 54.319 cm2 developed, with an average of 1.36 cm2. Differences between the control group and the group with anastomosis were prominent (p < 0.001, ANOVA, LSD and Scheffé tests), unlike differences between the control and colonic trauma groups (Fig. 2).
If we consider the area with manipulation (with colonic trauma or anastomosis), in the group with colonic trauma 17.64% of the surface was tumoral in this colonic area, while in the group with anastomosis 62.06% of the surface was tumoral in the perianastomotic area.
As regards differences obtained between anastomosis subgroups regarding suture material, in the group with continuous suture the average value was 0.9 cm2 per animal, in the group with interrupted suture 1.7 cm2, in the group with titanium 2.2 cm2, and in the group with reabsorbable material 0.6 cm2 (Fig. 3, p < 0.05, ANOVA, LSD and Scheffé tests).
Tumoral percentage
Relating the surface of colonic tumoral tissue with the animal's colonic surface the tumoral percentage is obtained. In the group without surgery, this tumoral percentage was 2.99%, while in the group with colonic trauma it increased to 8.63%, with tumoral percentage reaching 23.54% in the group with colo-colonic anastomosis. Differences between anastomosis and control groups were significant (Fig. 4, p < 0.0005, ANOVA, LSD and Scheffé tests).
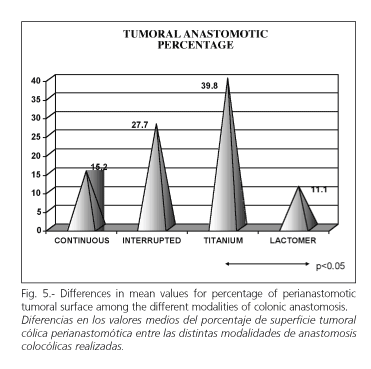
In the group with colonic anastomosis, the perianastomotic area had 18.65% of neoplastic tissue, while in the group with trauma this figure was 2.25% in this area. When the group with colo-colonic anastomosis is considered alone, the highest tumoral percentage appeared in the group with titanium (39.8%), followed by the groups with interrupted suture (27.7%) and continuous suture (15.2%). The group with the smaller percentage of neoplatic tissue was the group with added reabsorbable material (11.1%) (Fig. 5, p < 0.05, ANOVA, LSD and Scheffé tests).
Suture material in the anastomotic line
In the groups in which colo-colonic anastomosis was performed, there was a positive relationship between the length of suture material that remained included in the anastomotic line and the tumoral surface and percentage found in the histological study. The greater quantity of suture material remain included in the anastomotic line, the greater tumoral surface and percentage are found in the histological study. If we consider the group with reabsorbable material added in the anastomotic line, we appreciate that it contains a tumoral surface and percentage smaller than those in the remaining groups, suggesting that this material has a protective effect on colonic carcinogenesis.
Histological study
Except two cases of adenocarcinoma with signet ring cells (1.08%), the rest were non-specific adenocarcinomas. Adenocarcinomas were well differentiated in 56.94% of cases, with moderate differentiation in 35% and poor differentiation in 8.06% of cases. Regarding the level of invasion, it reached the submucosa in most cases (53.33%); in situ invasion was seen in 30% and beyond-the-submucosa invasion in 16.66%. If we analize the invasion level in the study groups, we find that tumors are more invasive in the group with colo-colonic anastomosis in comparison with those in the control group (p < 0.05, Chi-square).
A total of 320 pericolonic lymph nodes (3.5 nodes per rat) were collected. Seven of them contained adenocarcinoma micrometastasis. This represents 2.18% of micrometastases in all nodes evaluated; 3.78% of adenocarcinomas evaluated had nodal involvement, all of them in the group with colo-colonic anastomosis.
DISCUSSION
In the present experimental model, experimentally-induced carcinogenesis can neither result from phenomena related to implanted metastases in the manipulated area, nor to a defficient surgical technique, as no previous colonic tumors were present. All experimentally-induced colonic tumors in animals respond to the induction and promotion of the effects of DMH. This effect will be more intense in some groups of animals depending on the experimental variables used.
We analyzed a variety of experimental manipulations to evaluate the effect of colonic manipulation on induced carcinogenesis, either in generic form (surgical group versus control group), or in a specific form (anastomosis versus control and trauma groups; comparison among the different modalities of anastomosis between them).
All postoperative complications and deaths occurred in the surgical group, and more specifically in the group with anastomosis. All were due to perianastomotic adenocarcinomas. Global mortality in this study was 2.22% and the prevalence of intestinal occlusion was 3.33%.
Tumoral colonic proliferation was evaluated using three parameters: number of tumors, surface of colonic tumoral tissue, and percentage of tumoral tissue. The number of tumors is the least reliable of these three indicators because of the wide variability of both the tumor size and the colon size. Tumoral surface is a better approach to the quantity of colonic tissue involved, but percentage of tumoral tissue is the most adequate parameter for the quantification of the carcinogenic process, and therefore for evaluating tumoral surface in relation to the colon size in every animal.
The number of colonic induced tumors was 185 adenocarcinomas. In the group with experimental manipulation, a greater number of tumors developed compared with the control group (2.55 tumors per animal vs 0.83). This greater prevalence of tumors in the manipulated group was significant. If we consider the control group and the groups with colonic trauma and with colo-colonic anastomosis, differences were also significant.
We observed that experimental colonic manipulation increases the number of pharmacologically-induced tumors when compared with the control group, and that the colo-colonic anastomosis induces more tumors than a simulated anastomosis, also causing a clustering of tumors at the perianastomotic area. There were no significant differences in the number of tumors between groups with different modalities of anastomosis.
If we consider tumoral surface, this is greater in the group with experimental manipulation in comparison with the control group, a difference that is prominent. Comparing control, colonic trauma and anastomosis groups, differences are only significant for the group with anastomosis compared with the control one. When we analyze differences among the different modalities of anastomosis, we only found significant differences be-tween the groups with added titanium or reabsorbable material.
While the colo-colonic anastomosis was the experimental manipulation that originated more tumoral surface, the addition of non-reabsorbable material within the anastomotic line clearly contributes to this induction.
Regarding tumoral percentage, a clear and significant increase in colonic tumoral percentage is seen in the group with experimental manipulation as compared with the control group. If we separate the manipulated groups into a trauma group and a group with anastomosis, and we compare them with the control group, we obtained identical results as those obtained for tumoral surface; the creation of a colo-colonic anastomosis increases significantly the tumoral percentage versus the control group.
The group with colonic anastomosis presented differences between subgroups. Thus, a greater tumoral percentage was found in the group with added titanium (39.8%), followed by the groups with interrupted suture (27.7%), continuous suture (15.2%), and added reabsorbable material (11.1%). Differences between the added reabsorbable material and titanium groups were significant.
These differences support that the quantity of material included in the suture line influences the induction and/or promotion of carcinogenesis, and that this phenomenon increases with the amount of non-reabsorbable material included in the anastomosis.
The histological study of tumors shows differences when we assess the invasion extent of adenocarcinomas. In the group with anastomosis, tumors were more invasive than in the control group, but we did not find significant differences between the different modalities of anastomosis.
Regarding metastatic nodal involvement, nodal invasion was only seen at the epicolic nodes in the group with anastomosis. This highlights the fact that performing anastomosis generates more aggressive and invasive tumors than isolated trauma or no surgery.
We may conclude that the experimental manipulation of the colon in rats enhances drug-induced carcinogenesis; the creation of a colo-colonic anastomosis induces a greater increase in the carcinogenic process as compared with the simulated anastomosis. This process of neoplastic induction and promotion in the perianastomotic area is further enhanced by the quantity of suture material included in the anastomosis and by the non-reabsorbable nature of the material implanted in the anastomotic line.
All these experimental situations can promote the carcinogenic process not only regarding intensity, but also playing a role in the nature of this process, inducing more invasive tumors, and showing a greater capacity of causing lymph node metastasis.
REFERENCES
1. Arnaud JP, Schloegel M, Ollier JC, Adloff M. Colorectal cancer in patients over 80 years of age. Dis Colon Rectum 1991; 34: 896-8. [ Links ]
2. McGregor JR, Galloway DJ, McCulloch P, George WD. Anastomotic suture materials and implantation metastasis: an experimental study. Br J Surg 1989; 76 (4): 331-4. [ Links ]
3. Domínguez F, Riera JR, Junco P, Tojo S, Díaz-Faes M. Influencia en el pronóstico a corto plazo de la sobreexpresión de la proteína p53 en carcinomas colorrectales. Rev Esp Enferm Dig 1994; 86 (5): 796-802. [ Links ]
4. García JC, Cugat E, Angás J, Gonzalez FJ, Reverter JC, Lacy AM. Cáncer colorrectal: resultados de un protocolo de seguimiento. Cir Esp 1993; 53 (6): 430-3. [ Links ]
5. Tissot E, Naouri A, Nasr JR, Feghali B. Récidives locorégionales isolées des cancers coliques : resultats du traitment, facteurs de risque de ces récidives. Lyon Chir 1992 ; 88 (3) : 277-9. [ Links ]
6. Garcia JA, Morcillo MA, Vázquez JL, Zaragoza C, Moltó M, Cámara J. Cáncer colorrectal en el anciano. Experiencia de los 5 primeros años en un Servicio de cirugía de un hospital Comarcal. Cir Esp 1996; 60: 256-7. [ Links ]
7. Törnqvist A, Ekelund G, Leandoer L. The value of intensive follow-up alter curative resection for colorrectal carcinoma. Br J Surg 1982; 69: 725-8. [ Links ]
8. Palmer ML, Herrera L, Petrelli NJ. Colorrectal adenocarcinoma in patients less than 40 years of age. Dis Colon Rectum 1991; 34: 343-5. [ Links ]
9. Delpero JR, LeTreut YP, Granger F, Pol B, Houvenaeghel G, Brandone JM. Chirurgie des récidives locorégionales des cancers colorectaux. Lyon Chir 1992; 88 (6): 475-82. [ Links ]
10. Pol B, Delpero JR, Le Terut YP, Hardwigsen J, Houvenaeghel G. Resections of colorrectal adenocarcinoma recurrentes: a series of 120 patients. Br J Surg 1996; 83 (2): 32-3. [ Links ]
11. Rubio CA, Nylander G. Surgical resection of the rat colon: effects on carcinogenesis by 1,2-Dimethylhydrazine. JNCI 1982; 68 (5): 813-5. [ Links ]
12. Roe R, Fermor B, Williamson RCN. Proliferative instability and experimental carcinogenesis at colonic anastomoses. Gut 1987; 28: 808-15. [ Links ]
13. Umpleby HC, Williamson RCN. Anastomotic recurrente in large bowel cancer. Br J Surg 1987; 74: 873-8. [ Links ]
14. Appleton GVN, Davies PW, Williamson RCN. El efecto de la desfuncionalización sobre la citocinética y el cáncer en las líneas de sutura del colon. Br J Surg 1990; 77: 768-72. [ Links ]
15. Phillips RKS, Cook HT. Effect of steel wire sutures on the incidence of chemically induced rodent colonic tumours. Br J Surg 1986; 73: 671-4. [ Links ]
16. Calderesi RN. Differential effects of surgical suture materials in 1-2 dimethylhydrazine-induced rat intestinal neoplasia. Cancer Res 1984; 44: 2827-30. [ Links ]











 texto en
texto en