Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO  Similares en Google
Similares en Google
Compartir
Revista Española de Enfermedades Digestivas
versión impresa ISSN 1130-0108
Rev. esp. enferm. dig. vol.97 no.2 Madrid feb. 2005
| ORIGINAL PAPERS |
Mortality from colorectal cancer in Andalusia: findings in favor of mass screening
M. Ruiz-Ramos, A. Escolar Pujolar1 and T. Hermosín Bono2
Registro de Mortalidad de Andalucía. Seville. 1Service of Preventive Medicine and Public Health. Hospital Universitario Puerta
del Mar. Cádiz. 2Centro de Salud de Camas. Seville. Spain
ABSTRACT
Objective: to describe mortality from colorectal cancer (CRC) in Andalusia from 1975 to 2001 in order to facilitate decision-making on mass screening.
Design: a population-based descriptive study. Considering mortality data from CRC and the resident population of Andalusia during the studied period, the main indicators of mortality by age and sex groups were calculated, and trends were estimated.
Results: there have been annual increases in mortality of 1.99% in men and 0.15% in women during the study period. In men, this is mainly due to increased mortality in those older than 65 years, and in women in those in the 35- to 64-year-old age group. In the younger age groups of both sexes, there has been a decline in mortality. The trend of mortality from colon cancer was positive in both men and women, but from rectal cancer was negative in women and slightly positive in men.
Conclusions: increased mortality from CRC in Andalusia may justify mass screening measures to potentially change mortality evolution, as has occurred in other countries.
Key words: Colorectal cancer. Mortality. Age groups. Mass screening.
Ruiz-Ramos M, Escolar Pujolar A, Hermosín Bono T. Mortality from colorectal cancer in Andalusia: a contribution to mass screening. Rev Esp Enferm Dig 2003; 95: 104-114.
Recibido: 10-02-04.
Aceptado: 21-07-04.
Correspondencia: Miguel Ruiz Ramos. Servicio de Medicina Preventiva y Salud Pública. Hospital Universitario Puerta del Mar. Avda. Ana de Viya, 21. 11009 Cádiz.
Tel.: 956 002 214. Fax: 956 002 347
INTRODUCTION
Cancer is a major public health problem in Western countries. One of the bases of any prevention and control strategy is the prevalence and trends in the population.
Cancer of the colon and rectum (CRC) is the second most frequent cause of death from cancer in the more developed regions of the world. Annually some 940,000 new cases are diagnosed, and around 490,000 persons die from this cause (1). In Spain colorectal cancer, the second most frequent malignant tumor, is recorded as being responsible for 11.5% of total cancer-related deaths in men and for 14.9% of cancer-related deaths in women according to data for 2001; the number of new cases per year is estimated at around 21,000 in both sexes, compared with some 12,000 deaths (2). Mortality and incidence rates in Spain are lower than in Northern European countries, and below average European rates, but the mortality rate in men is now higher than in France, Italy, or the United Kingdom (3).
In countries like the United States (4) and the United Kingdom, where incidence and mortality rates are relatively high, mortality began to decline several years ago, and there has been a 50% decrease in mortality between 1950 and the end of the 1990's. A similar trend is revealed in most countries in the European Union, except for Greece, Portugal and Spain, where mortality rates in both sexes continues to increase (5); Spain has experienced a consistent mean yearly increase of 2.6% in men since 1975, and a much lower increase of 0.8% in women, albeit a trend towards stabilization has been recently observed (2).
The existence of marked geographic differences in CRC-related incidence and mortality leads to think that, in addition to genetic factors that cannot be modified, there are environmental factors that play a significant role in the pathogenesis of this condition. With respect to a potential relationship between consumption habits and CRC, a certain type of diet (6,7) -rich in red meats, cholesterol, and animal fats- is being increasingly identified as a risk factor, and there is evidence -though still contested to some extent- that the use of tobacco and alcohol (8), specifically beer (9), are also possible risk factors. Dietary fiber in the case of colon cancer (10), antioxidative agents, calcium, exercise, and probably non-steroid anti-inflammatory drugs (11,12) have a protective effect against the development of colorectal adenomas and carcinomas.
In any case, health promotion programs aimed at changing dietary and personal hygiene habits in the population do yield long-term results and should be complemented with health-care action of more immediate impact. In this context, the treatment of CRC cases with clinical manifestations has serious limitations, despite advances made in surgical techniques and adjuvant therapies. It is a well-known fact that the extent of disease at the time of diagnosis is the main prognostic factor for its evolution. For these reasons, it is justified to believe that, currently, the key to reducing mortality from CRC lies in the earliest possible diagnosis of this disease.
The objective of this study is to describe mortality from colorectal cancer (CRC) in Andalusia from 1975 to 2001, and thus to contribute to guiding the decisions that may be taken regarding prevention, and specifically those related to the possible implementation of mass screening programs, for the early detection of CRC in our region.
MATERIAL AND METHODS
The mortality data used were obtained from Death Statistics Bulletins (Boletines Estadísticos de Defunción-BED) supplied by the National Institute of Statistics (INE) for the years 1975 to 1991 (13), and by the Institute of Statistics of Andalusia (IEA) (14) for the period 1992 to 2001. Codes 152, 153 and 154 of the International Classification of Diseases (ICD), 8th revision, and codes 152, 153 and 154 plus 159.0 of ICD 9th revision have been included. This extended grouping was taken because it was seen that, on passing from the 8th to the 9th revision in the year 1980, the disease location "intestinal tract not specified" that in the 8th revision was included with the colon and coded 153.9, was changed to "other poorly-defined sites within the digestive organs and peritoneum" in the 9th revision, coded 159.0. The effect of this change of classification is that the great majority of deaths included as due to "cancer of the colon" in ICD 8th revision have now been recorded as due to "non-specific cancers". The tenth revision of ICD has been used for the period 1999 to 2001, and this classifies cancers of the colon and rectum under codes C18 to C21. In addition to the basic cause of death, other data available in BED that were used for this study are municipality of residence, year of death, sex, and age in years at time of death.
The populations necessary for the calculation of indicators correspond to projections made by INE, grouped in quinquennial age groups except for children younger than 1 year, children from 1 to 4 years of age, and adults aged 85 years or more.
The indicators calculated are: crude rate of mortality per 100,000 persons-year; age-adjusted rates per 100,000 persons-year using the direct method, and taking the European Standard Population as the reference population; and truncated age-adjusted rates of 35 to 64 years. The latter type of rate only takes into account age groups from 35 to 64 years, thus eliminating lack of accuracy in the diagnoses of cancer, particularly in older persons; -furthermore, the probability of dying at ages younger than 35 years is very low and may distort results.
We have also calculated cumulative rates (15) from 0 to 74 years as an approximation of the risk of dying from cancer of the colon and rectum before 74 years of age in the absence of death risks from other causes. A rate adjusted by the direct method may be considered that in which the proportion of individuals is equal in all age groups; it is easy to calculate and, as rates adjusted by age, permits comparisons to be made between populations with different age structures.
Lastly the rates -adjusted for age using the direct method- of Potential Years of Life Lost (16) (PYLL) between 1 and 70 years were obtained as an indicator of premature mortality. Each of these indicators has been calculated for each of the years in the series, and for men and women separately.
The annual percentage change (APC) has been estimated taking the age-adjusted rates of the first (1975-1979) and the last (1997-2001) quinquennium. For this, the Napierian logarithms of these rates were calculated, the value for the first quinquennium was subtracted from that of the last, the difference was then divided by the 27 years in the period studied, and the result was multiplied by one hundred. Calculations were made for those below 35 years of age, those from 35 to 64 years, those older than 64 years, and for all ages, both for CRC and for cancer of the colon and the rectum separately.
RESULTS
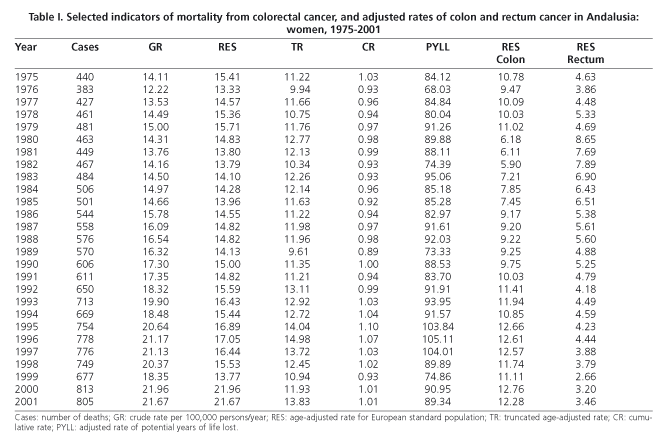
Mortality from CRC in Andalusia during the period studied has undergone a considerable increase in absolute numbers (Tables I and II), rising from 306 deaths in 1975 to 967 in 2001 in men, and from 440 to 805 deaths in women. Crude rates of mortality also suffered a notable increase, from 10.18 to 26.92 per 100,000 persons-year in men, and from 14.11 to 21.67 in women between 1975 and 2001. Age-adjusted rates present a very different behaviour in men and in women. In men, rates have risen from 15.10 to 26.51 per 100,000 persons-year from 1975 to 2001, but in women it has only changed slightly, from 15.41 to 21.67 per 100,000 persons-year, during the same time period.
Indicators of early mortality increased less, especially in women. In men, PYLL has passed from 103.79 in 1975 to 117.9 per 100,000 persons-year in 2001, and truncated rates between 35 and 64 years of age have increased from 12.73 to 18.32 per 100,000 persons-year. In women, PYLL rose from 84.12 to 89.34 per 100,000 persons-year in the same period, and the truncated rate went from 11.22 to 13.83 per 100,000 persons-year. The cumulative rate in men has increased from 1.10 to 1.85% between 1975 and 2001, and in women it has fluctuated between the maximum values of 0.89% in 1989 and 1.04% in 1994.
Figure 1 shows age-adjusted rates. For CRC, men showed a clear rise, especially striking from 1986 onwards; in women, the curve of time evolution has been much smoother. The ratio between men and women was around one in the first quinquennium studied, but then increased to a level 71% higher in men in 1998. It can also be seen that the greater part of the mortality was due to cancer of the colon, which became responsible for up to 80% of deaths in women and a slightly lower percentage in men.

Fig. 1.-Mortality from colorectal, colon, and rectum cancer by sex groups in Andalusia, 1975-2001. Rates adjusted to the
European standard population.
Mortalidad por cáncer colorrectal, de colon y de recto por sexo en Andalucía, 1975-2001. Tasas ajustadas a la población
estándar europea.
Figure 2 shows the curve of mortality from colorectal cancer by age groups for men and women in the two quinquennia of 1975-1979 and 1997-2001. For both men and women, rates increase exponentially with age, and attention is drawn to the substantial increase of mortality in men at all ages in the most recent period of 1997 to 2001.

Fig 2.- Mortality from colorectal cancer by age group, sex and period in Andalusia, 1975 to 2001.
Mortalidad por cáncer colorrectal por grupos de edad, sexo y periodo en Andalucía, 1975 a 2001.
Table III lists annual mean percentage changes in specific mortality rates by age groups. For colorectal cancer, in those younger than 35 years, there has been a decrease in mortality greater than 3% per year in men, and equal to 1.88% in women. In the 35-64 years age group there was a significant increase particularly in men, less steep in women. Above all, the greatest increase in mortality, however, took place in men older than 65; in contrast, in women older than 65 the annual increase in mortality was less than that in the 35-to-64 years age group. Cancer of the colon has increased in both men and women, and cancer of the rectum has declined in women and increased only slightly in men.
DISCUSSION
During the period studied in Andalusia, the trend of mortality from colorectal cancer has increased in men and to a lesser extent in women. The main factors that may be involved in this evolution are diagnostic advances, and changes in exposure to risk factors for this disease. This trend is similar to that presented in other countries in Southern Europe, like Greece and Portugal (5,17), and may be caused by changes in the dietary habits of the population, such as the increased consumption of animal fats that has taken place in these countries over the last twenty years.
The difficulty of ensuring a correct classification of the cause of death selecting between that produced by colon cancer or rectal cancer -since in so many cases cancer is located in the rectosigmoidal region- is the reason why deaths from cancer at both sites have been jointly analyzed. Although epidemiological characteristics are similar, they have peculiarities that make it advisable to study them separately (18). In addition, as can be seen in the results of this study, the mortality trend is different for each disease.
Numerous studies demonstrate that the great majority of CRC cases originate in adenomatous polyps, that risk increases in proportion to the number of adenomas in an individual, and that its most extreme form is familial polyposis (19). These adenomatous polyps are very frequent in developed countries, particularly in persons older than 50 years, and their location can be superimposed on that of CRC (20). The degenerative potential of these adenomas increases as a function of their size (> 2 cm), histological type (villous adenomas), and degree of dysplasia (severe). The set of genetic and cytological transformations that occur in an adenomatous polyp that becomes malignant has been named the adenoma-carcinoma sequence. This sequence has been repeatedly confirmed by epidemiological, histopathological, and more recently by genetic studies.
In any case, it is known that the development of CRC from adenomatous polyps takes some 2-3 years to reach a tumor size of 1 cm, and there is an interval between polyp and cancer of at least four years (21). Thus if the detection and resection of lesions can be performed during the adenomatous phase, the development of CRC may then be interrupted, as has been demonstrated by several studies (22).
Because of these clinical and epidemiological characteristics, one of the key factors to reduce mortality from CRC must be early diagnosis. Although programs for early detection remain controversial (23), two basic techniques for the screening of this cancer have been proposed: presence of fecal occult blood (FOB) and sigmoidoscopy. The first one is relatively simple and inexpensive, but has a low sensitivity: rehydration of samples may increase sensitivity but at the expense of reducing specificity (24,25). In the case of sigmoidoscopy, sensitivity is high and allows 50 to 60% of all cancers to be detected; however, the healthy population poorly accepts the procedure. The accessibility and availability of necessary resources also leads to an effective use of these tests at a populational level, this being low among those adults who, because of their age (in general 50 years or more) are candidates (26).
In the near future, the identification of genetic markers may bring about an improvement of screening techniques for CRC (27). Meanwhile, both the US Preventive Task Force (USPTF) (28) and the Canadian Task Force (29) coincide in their recommendations that the presence of FOB and sigmoidoscopy are effective in the reduction of mortality rates, and can therefore be utilized as early detection tests. The revised European Code Against Cancer, however, only makes a recommendation regarding FOB -that the test for the presence of FOB should be done every 2 years from the age of 50 years on (30).
Before recommending a systematic use of the FOB test and/or sigmoidoscopy, and as stated in the Integral Oncology Plan for Andalusia (31), pilot studies in those areas with high rates of morbidity and mortality from colorectal cancer are needed in order to assess their acceptance by the population and the latter's level of participation, as well as the cost-effectiveness of such screening methods. The performance of these studies should not be delayed, given the magnitude and trends in mortality from this cancer in our region.
REFERENCES
1. Stewart BW, Kleihues P, eds. World Cancer Report. IARCPress. Lyon 2003. [ Links ]
2. López-Abente G, Pollán M, Aragonés N, Pérez-Gomez B. Informe sobre la salud de los españoles. Cáncer. Información disponible a diciembre de 2003. Área de epidemiología ambiental y cáncer. Centro Nacional de Epidemiología. Instituto de Salud Carlos III. Madrid [consulted: 10 June 2004]. Available on: http://193.146.50.130/cancer/salud-cancer-2003.pdf]. [ Links ]
3. Ferlay J, Bray F, Sankila R, Parkin DM. EUCAN: Cancer Incidence, Mortality and Prevalence in the European Union 1998, version 5.0. IARC CancerBase No. 4. Lyon, IARCPress, 1999. [ Links ]
4. Weir HK, Thun MJ, Hankey BF, Ries LA, Howe HL, Wingo PA, et al. Annual report to the nation on the status of cancer, 1975-2000, featuring the uses of surveillance data for cancer prevention and control. J Natl Cancer Inst 2003; 95: 1276-99. [ Links ]
5. Boyle P, d'Onofrio A, Maissonneuve P, et al. Measuring progress against cancer in Europe: has the 15% decline targeted for 2000 come about? Annals Oncology 2003; 14: 1312-25. [ Links ]
6. Berrino F, Krogh V, Riboli E. Epidemiology studies on diet and cancer. Tumori 2003; 89: 581-5. [ Links ]
7. Benito E, Cabeza E. Diet and risk: an overview on Spanish studies. Eur J Cancer Prev 1993; 2: 215-9. [ Links ]
8. Sharpe CR, Siemiatycki J, Rachet B. Effects of alcohol consumption on the risk of colorectal cancer among men, by anatomical subsite (Canada). Cancer Causes Control 2002; 3: 483-91. [ Links ]
9. Ji BT, Dai Q, Gao YT, Hsing AW, McLaughlin JK, Fraumeni JF Jr, et al. Cigarette and alcohol consumption and the risk of colorectal cancer in Shanghai, China. Eur J Cancer Prev 2002; 11: 237-44. [ Links ]
10. Bingham SA, Day NE, Luben R, Ferrari P, et al. Dietary fibre in food and protection against colorectal cancer in the European Prospective Investigation into Cancer and Nutrition (EPIC): an observational study. Lancet 2003; 361: 1496-501. [ Links ]
11. Herbert C. Diet and non-steroidal anti-inflammatory drugs: Role in prevention of colorectal cancer. Gastroenterol Clin Bio 1998; 22 (Supl. 3): S202-17. [ Links ]
12. García-Rodríguez LA, Huerta-Álvarez C. Reduced risk of colorectal cancer among long-term users of aspirin and non-aspirin non-steroidal anti-inflammatory drugs. Epidemiology 2001; 12: 88-93. [ Links ]
13. INE. Movimiento Natural de la Población Española. Defunciones según la causa de muerte. Vol III. Años 1975-1991. Madrid: Instituto Nacional de Estadística. [ Links ]
14. IEA. Movimiento Natural de la Población. Defunciones en Andalucía 1992-1997. Sevilla: Instituto de Estadística de Andalucía. [ Links ]
15. Breslow NE, Day NE. Statistical Methods in Cancer Research. Volume II- The design and analysis of cohort studies. IARC Scientific publications Nº 82. International Agency for Research on Cancer. Lyon 1987. [ Links ]
16. Romeder JM, Mcwinnie JR. Potential years of life lost between ages 1 and 70: an indicator of premature mortality for health planning. Int J Epidemiol 1976; 6: 143-51. [ Links ]
17. La Vecchia C, Lucchini F, Negri E, Boyle P, Maisonneuve P, Levi F. Trends of cancer mortality in Europe, 1955-1989: I, Digestive sites. Eur J Cancer 1992; 28: 132-235. [ Links ]
18. Percy C, Stanek E 3rd, Gloeckler L. Accuracy of cancer death certificates and its effect on cancer mortality statistics. Am J Public Health 1981; 71: 242-50. [ Links ]
19. Muto T, Bussey HTR, Morson BC. The evolution of cancer of the colon and rectum. Cancer 1975; 36: 2251-8. [ Links ]
20. O'Brien NJ, Winawer SJ, and The National Polyp Study. Patient and polyps characteristics associated with high-grade dysplasia in colorectal adenomas. Gastroenterology 1990; 98: 371-80. [ Links ]
21. Fearon ER, Vogelstein B. A genetic model for colorectal tumorogenesis. Cell 1990; 61: 759-67. [ Links ]
22. Winawer SJ, Zauber AG, Ho MN, et al. Prevention of colorectal cancer by colonoscopic polypectomy. N Engl J Med 1993; 329: 1977-81. [ Links ]
23. Boyle P. Faecal occult blood testing (FOBT) as screening for colorectal cancer: the current controversy. Annals Oncology 2002; 13: 16-8. [ Links ]
24. Mandel JS, Bond JH, Timothy R. Reducing mortality from colorectal cancer by screening for faecal occult blood. N Engl J Med 1993; 328: 1365-71. [ Links ]
25. Tárraga López PJ, Marín Nieto E, Celada Rodríguez A, García Molinero MJ, García Olmo D, Solera Albero J. Economic evaluation of colorectal cancer screening with fecal occult blood detection. Rev Esp Enferm Dig 2000; 92: 334-48. [ Links ]
26. Vernon SW. Participation in colorectal cancer screening: a review. J Natl Cancer Inst 1997; 89: 1406-22. [ Links ]
27. Dong SM, Traverso G, Johnson C, Geng L, Favis R, Boynton K, et al. Detecting colorectal cancer in stool with the use of multiple genetic targets. J Natl Cancer Inst 2001; 93: 858-65. [ Links ]
28. Pignone M, Rich M, Teutsch SM, Berg AO, Lohr KN. Screening for colorectal cancer in adults at average risk: a summary of the evidence for the U.S. preventive services task force. Ann Intern Med 2002; 137: 132-41. [ Links ]
29. Recommendation statement from the Canadian Task Force on Preventive Health Care: colorectal cancer screening. CMAJ 2001; 165: 206-8. [ Links ]
30. European Code Against Cancer and scientific justification: third version (2003) [consulted 22/06/2004]. Available on: http://www.cancercode.org/ add_scree01.htm. [ Links ]
31. Consejería de Salud. Plan Integral de Oncología 2002-2006. Junta de Andalucía. [consulted 22/06/2004]. Avalailable on: http://www. csalud.junta-andalucia.es/principal/documentos.asp?pagina=Plan_ Integral_Oncologia. [ Links ]











 texto en
texto en