My SciELO
Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista Española de Enfermedades Digestivas
Print version ISSN 1130-0108
Rev. esp. enferm. dig. vol.105 n.7 Madrid Aug. 2013
https://dx.doi.org/10.4321/S1130-01082013000700002
Intermittent boluses versus pump continuous infusion for endoscopist-directed propofol administration in colonoscopy
Administración de propofol mediante bolus intermitente frente a infusión continua con bomba controlada por el endoscopista en la colonoscopia
Jesús M. González-Santiago, Elisa Martín-Noguerol, Gema Vinagre-Rodríguez, Moisés Hernández-Alonso, Carmen Dueñas-Sadornil, Belén Pérez-Gallardo, José M. Mateos-Rodríguez, Miguel Fernández-Bermejo, Pilar Robledo-Andrés and Javier Molina-Infante
Department of Gastroenterology. Hospital San Pedro de Alcántara. Cáceres, Spain
ABSTRACT
Background: non-anesthesiologist administration of propofol (NAAP) using continuous infusion systems may achieve a more sustained sedative action.
Aim: to compare intermittent boluses (IB) with pump continuous infusion (PCI) for NAAP, targeted to moderate sedation, for colonoscopy.
Methods: 192 consecutive outpatients were randomized to receive IB (20 mg propofol boluses on demand) or PCI (3 mg/kg/h plus 20 mg boluses on demand). Sedation could be stopped at cecal intubation at the discretion of the endoscopist. Satisfaction rates of the patient, nurses and endoscopist, propofol doses, depth of sedation (at the beginning, at cecal intubation and at the end), recovery times, complications and were collected.
Results: there were no differences between groups regarding patient, nurse or endoscopist satisfaction rates with procedural sedation. Propofol doses (mg) were significantly higher during the induction phase (86 [30-172] vs. 78 [30-160], p 0.03) and overall (185 [72-400] vs. 157 [60-460], p = 0.003) for PCI group. 81 % of assessments of the depth of sedation were moderate. The level of sedation (O/AAS scale) was borderline significantly deeper at cecal intubation (2.38 vs. 2.72; p = 0.056) and at the end of the procedure (4.13 vs. 4.45; p = 0.05) for PCI group, prolonging thus early recovery time (6.3 vs. 5.1 minutes, p = 0.008), but not discharge time. Complications, all of them in minors, were non-significantly more frequent in the PCI group (9 vs. 7 %, p = 0.07).
Conclusions: NAAP for colonoscopy was safely administered with comparable satisfaction and complication rates with either IB or PCI.
Key words: Propofol. Colonoscopy. Sedation. Continuous infusion. Bolus administration. Moderate sedation.
Introduction
Endoscopic sedation for colonoscopy increases overall rates of cecal intubation and polyp detection and provides better patient tolerability, satisfaction and adherence to further surveillance (1). Propofol (2,6 diisopropyl phenol) is an ultrashort-acting sedative agent with no analgesic properties, which provides sedative and amnestic effects. Non-anesthesiologist administration of propofol (NAAP), including endoscopists, has evolved and grown during the last decade, as shown by recent American and European guidelines (2,3). Compared with traditional sedation (benzodiacepines plus opioids), NAAP presents similar rates of adverse effects, provides higher postprocedure patient satisfaction, decreases time to sedation and recovery times (2-4), whereas its safety is endorsed by overwhelming evidence (5). Intermittent administration of propofol bolus is the current standard administration technique for NAAP in colonoscopy. An initial bolus of propofol (dose adapted to patient weight, age or comorbidity) is administered intravenously, followed by repeated boli according to desired sedation depth and the patient condition. Scarce scientific evidence is available on NAAP using continuous infusion systems for colonoscopy so far now (6-8). According to our hypothesis, continuous infusion of propofol might exert a more sustained sedative effect by maintaining a preset concentration of propofol in the blood or brain, providing a higher patient satisfaction with the procedural sedation. On the other hand, titration of NAAP to a level of conscious sedation with a continuous infusion system during colonoscopy might potentially avoid having a nurse exclusively devoted to administration of sedative agents (9).
However, no data are available comparing both administration techniques by non-anesthesiologists and titrated to a moderate level of sedation up to now. Therefore, the aims of this study are: a) to compare two different NAAP regimens -intermittent boluses (IB) vs. pump continuous infusion (PCI) for colonoscopy; and b) to determinate the satisfaction rates and to evaluate the feasibility of moderate sedation with these regimens.
Methods
This study is a prospective, open-label, unicenter, randomized study. It was performed according to good clinical practice and the Declaration of Helsinki, and was approved by the institutional Ethical Committee. From March to July 2011, 245 consecutive patients presenting for outpatient morning colonoscopy were eligible to enroll in this study (Fig. 1). Exclusion criteria were age less than 18 yrs, allergy to soybeans, eggs, peanuts, propofol, history of colorectal surgery, obstructive sleep apnea syndrome, ASA (American Society of Anesthesia) class IV, short and tick neck, difficult intubation due to inability to open the mouth widely (Mallampati III-IV), previous colonoscopy, refusal, inability or unwillingness to give written consent, patients scheduled for advanced therapeutic colonoscopy or for more than one endoscopic procedure and pregnancy. All patients provided written informed consent.
All colonoscopies were air-insufflated performed, after split-dose bowel preparation (two sodium picosulphate, magnesium oxid and citric acid (Citrafleet®, Laboratorios CasenFleet, Zaragoza, Spain) powder sachets, administered at 7:00 pm the day before and 2 to 6 hours before the procedure. Using the standard technique with the patients initially on the left side, before sedation all patients were given supplemental oxygen (2 L/min) through nasal cannula and monitored by electrocardiogram, pulse oximetry, heart rate and blood pressure monitoring. All patients had an intravenous line with continuously running normal saline infused by gravity. The procedures were performed by seven different attending gastroenterologists with more than five years of experience. Two fellows (JMGS, EMN) were in charge of recording all the measurements throughout the study time. Besides one endoscopist, a nurse was dedicated to monitor the depth of sedation and the infusion of sedatives and saline, whereas a second nurse was present to assist the endoscopist with technical maneuvers. All nurses and physicians were certified in advanced cardiac life support and a minimum of 12 months experience using propofol was required to participate in the study.
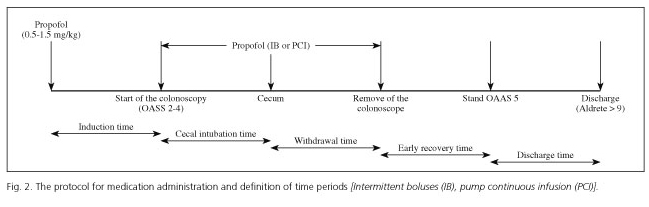
The protocol for medication administration and definition of time periods are listed in figure 2. Patients were randomized in a blinded-fashion using a computer-generated numeric sequence, of which the endoscopist was unaware, to receive propofol sedation (generic product 10 mg/ml) with either IB or PCI. IB was defined by intermittent use of 20 mg propofol boluses on demand, with a minimum period of 30 seconds between boluses to evaluate the effect of the drug. PCI was defined by continuous propofol infusion at a 3 mg/kg/h rate, via the Perfusor® Space (B. Braun, Melsungen AG; Fig. 3), with additional 20 mg boli administered if necessary. Boluses administration occurred upon pushing sequentially two buttons and this task could be performed by either the endoscopist or the nurses. Minimal, moderate and deep sedation were defined by ASA criteria and its correspondence with the OAA/S (Observer Assessment of Alertness/Sedation) scale (10,11). Deep sedation was defined as OAA/S 0-1, moderate sedation as OAA/S 2-4 and minimal sedation as OAA/S 5. The target of sedation throughout the whole endoscopic procedure was moderate sedation as OAA/S 2-4. To determine the level of sedation, we followed the sequence of verbal and tactile stimuli for assessment of sedation reported by Cohen et al. (11). Briefly, four step progressive stimulations (call the patient's name in a normal voice, call it in a louder voice, a light stimulus in the shoulder and shaking) were attempted. Patients who failed to respond to these 4 stimuli were considered to be deeply sedated.
Propofol could be stopped up to cecal intubation at the discretion of the endoscopist. After completion of the procedure, the patient stood stretched out in the procedure room with similar monitoring, accompanied by the nurse. When patients responded to normal verbal command, they were asked to sit up and were offered a drink. This was considered the early recovery time. If they were able to stand unassisted by the bed and had stable hemodynamics parameters (saturation > 90 % on room air and blood pressure and heart rate within 20 % of baseline), they were transferred to a locker room accompanied by a relative. The discharge criteria included ability to stand unassisted and tolerate clear liquids once dressed, until the patient reaches Aldrete score of ≥ 9 or returns to baseline Aldrete score. Once a patient met discharge criteria, they were allowed to leave at their own discretion.
Measurements
Before the endoscopic procedure, patients fulfilled a detailed questionnaire regarding demographic, anthropometric, anxiety, clinical and medication data. The primary endpoint in the study was to determine the satisfaction rates of patients, endoscopists and nurses with procedural sedation. Secondary endpoints were to evaluate depth of sedation, induction and intraprocedural propofol requirements, time to sedation, recovery times and rate of complications. The depth of sedation was estimated at the introduction of the endoscope, at cecal intubation and at the removal of the endoscope (OAA/S). The following events were considered complications: a decline in oxygen saturation to less than 85 % longer than 30 seconds, a heart rate less than 40 beats per minute and blood pressure less than 80/50 mmHg, need for mechanical ventilation or any cardiorespiratory event requiring anaesthesiologist assistance. Endoscopists and nurses rated their satisfaction with sedation in a 10-cm visual analogue scale (VAS) after discharge. An interviewer who was blind to the propofol sedation scheme received, contacted with the patients 24-48 hours after the procedure to answer a questionnaire regarding if they remembered scope insertion or scope removal and willingness to repeat it with a similar protocol and rated their satisfaction and pain perception on a scale of 0-10.
Statistical analysis and sample size
The SPSS 17.0 statistical analysis package (Chicago, IL) was used. Categorical variables are described with percentages and continuous variables are described with mean (standard deviation) or median (range) as appropriate. Associations between categorical was tested with the χ2 test (with Fisher's correction when necessary) and continuous data were assessed using the 2 sample t-test or Mann Whitney U, for parametric and non parametric data, respectively. Both intention-to-treat (ITT) and per protocol (PP) were performed for all variables.
The main evaluation criterion was the overall average score of patients' satisfaction for the two groups, which was assessed 24-48 hours after colonoscopy. The predetermination of the sample size (n = 192) was estimated, assuming an 90 % power at a 5 % significance level and a 5 % of loss rate, to detect a mean 10mm between-group difference on the VAS between control (IB) and experimental (PCI) groups based on mean and spread values of VAS satisfaction scores on previous studies. A total number of 90 patients were required in each group. Once the sample size was determined for primary outcome measures, we analysed the effects of the secondary outcome measures such as depth of sedation, induction and intraprocedural propofol requirements, time to sedation, recovery times and rate of complications.
Results
During the study period, a total of 237 colonoscopies were performed in an outpatient basis. A total of 45 patients refused or were not eligible for enrolment and 192 patients (81 %) were finally recruited (Fig. 1). Colonoscopy was not completed in six patients, who were excluded from the PP analysis (4 in group IB, 2 in group PCI). All analyzed variables had a similar tendency and level of statistical significance on both the ITT and PP analyses. The trial profile is detailed in figure 1.
Demographics
The baseline characteristics of the study population are summarized in table I. Patients were similar at baseline without differences regarding age, gender, body mass index, prior intake of serotonin selective reuptake inhibitors, ASA class or indication for colonoscopy.
Pain and satisfaction scores
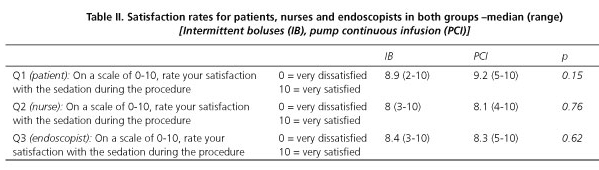
Patient, nurse and physician perception of the endoscopic experience is shown in table II. All patients completed the postprocedure questionnaire. Sixteen patients were not contacted successfully until up to 1-2 weeks after the procedure. There were non-statistically significant differences regarding patient satisfaction among all the involved endoscopists.
Sedation at the end of the procedure, evaluated by the ability to recall the removal of the instrumental, was non-statistically significant different between groups, neither discomfort or pain during the procedure. Accordingly, the quality of the sedation in the two groups was estimated similar by both patients and physicians. For both groups, 100 % of patients were willing to repeat with the same sedation protocol. Patient, nurse and physician satisfaction rates were comparable between both regimens.
Depth of sedation
There were 1,116 assessments of the depth of sedation during 186 colonoscopies (mean 6 per case). Of these, 89 (8 %) were categorized as deep sedation, 904 (81 %) as moderate and 123 (11 %) as minimal sedation. Patients from the PCI group had significantly lower OAA/S scores at cecal intubation (2.38 vs. 2.72; p = 0.05), and at the removal of the colonoscope (4.13 vs. 4.45; p = 0.05) (Fig. 4).
Propofol dosing
Median total propofol dose (mg) was significantly higher in group PCI (157 mg [92-222] vs. 185 [120-250], p = 0.003) on account of significantly higher doses of propofol during the induction phase (78 mg [55-101] vs. 86 mg [60-112], p = 0.03).
Procedure and recovery times
The details of procedure and recovery times, as defined in figure 2 are listed in table III. Median duration of the procedure was 19 min (8-40) for IB and 21 min (9-57) for PCI. No differences were observed in induction sedation, cecal intubation or withdrawal times between both groups. Early recovery time was significantly longer for PCI (Table II), but there were no differences between both groups in time to meet discharge criteria after early recovery.
Complications
Eleven patients (6 %) suffered from 16 minor adverse effects (9 in group PCI, 7 in group IB). Complication rates were non-significantly more frequent in the PCI group (9 vs. 7 %, p 0.07). They consisted of transient oxygen desaturation (n = 11) (< 85 % oxygen saturation and longer than 30 s) solved by increasing the oxygen flow rate and jaw thrust, bradycardia < 35 bpm which required the administration of atropine (n = 1) and transient hypotension (< 70/40 mmHg) rescued with fluids (n = 4). No mechanical ventilation or endotracheal intubation was necessary during the study.
Discussion
The present study is the first randomized trial comparing two different ways of administering propofol (boluses vs. pump infusion) by endoscopists for procedural sedation in colonoscopy. Both sedation schemes were safely administered by trained personnel and resulted in high rates of satisfaction, consistent with evolving consensus regarding NAAP. On account of significantly higher propofol doses, PCI provided a deeper and longer sedation, mostly within a moderate range, compared to IB. As such, early recovery time was longer for PCI but discharge time was not prolonged.
We did not observe major differences in satisfaction and complication rates when propofol was administered by either a continuous infusion pump or a nurse. Our results, in fact, absolutely coincide with a report from Spain, published in abstract form (9). In this report, propofol boluses were administered in one group by an external certified nurse (apart from other two nurses), whereas in the other group, propofol was directly administered using a pump infusion system controlled by the endoscopist, getting by without the external nurse. No differences were found among groups, leading to question the need of a registered nurse exclusively in charge of sedation in colonoscopies, which usually can be performed with a moderate sedation level. In this regard, consensus European guidelines for NAAP (3) recommend a person, commonly a nurse, dedicated to monitor the patient and administer sedation, whereas a second nurse should assist the endoscopist with the technical performance of the procedure. The American Guidelines (2) state that "once the patient's level of sedation and vital signs are stable, the registered nurse may perform minor, interruptible tasks", albeit if "deep sedation is performed by the administration of propofol by a registered nurse ... a second assistant is required for technical assistance during the procedure". Indeed, the need of two or three persons (and their cost) in the endoscopy room for sedation in upper endoscopy and colonoscopy is currently a matter of debate (13,14). Overall, the present study shows that both schemes are feasible and safe in clinical practice. We prefer using PCI targeted to moderate sedation in order to avoid having a nurse exclusively devoted to administration of propofol, allowing minor interruptible tasks for nurses or even exceptionally the performance of the procedure without a second nurse, when not able to be present in the endoscopy room, temporarily or permanently.
As experience with NAAP grows, endoscopists should not only get trained for advanced cardiac life support or airway assessment and management of complications, but may also gain some technical skills from anesthesiologists, like PCI. Continuous infusion via a perfusor pump may be preferable to take advantage of propofol pharmacokinetics in order to maintain a constant sedation level, so that we can avoid the peaks and valleys of the propofol plasma concentration once achieved the desired target of sedation and minimizing the risk of propofol overdosing as well. In this regard, NAAP through PCI may fit especially with endoscopic procedures longer than colonoscopy, such as endoscopic ultrasonography or endoscopic retrograde cholangiopancreatography, as recently shown (15). Of note, similar results than ours were obtained in this study when comparing both ways of administering propofol, titrated to deep sedation, for interventional endoscopy.
Propofol alone has been usually titrated to deep sedation for colonoscopy on the belief that a target of moderate sedation may hamper the performance of colonoscopy due to posturing and alarming movements in response to pain (6). The present study shows that NAAP using single-agent propofol can be safely and effectively used for endoscopic procedures requiring moderate sedation, in accordance with our previous results (16), with high rates of satisfaction and willingness to undergo repeat procedure. Despite overwhelming evidence demonstrating the safety and efficacy of NAAP for endoscopic procedures, it has been recently shown to be used only in one third of settings in an European survey (17). Its expansion is clearly burdened by medico-legal and cost concerns regarding potential deep sedation related adverse events. An interesting recent study comparing propofol sedation for colonoscopy in healthy individuals (ASA I/II class), administered by either the endoscopist or the anaesthesiologist, disclosed a higher level of satisfaction and lower side effects for the endoscopists group (18). In line with these studies, our results support the idea that routine use of anesthesiologist-administered propofol for low-risk individuals undergoing elective colonoscopy is not evidence-based and appears clearly unsustainable in cost terms.
The present study has also several drawbacks. The sample size was calculated to demonstrate differences regarding satisfaction rates between groups. This fact might have led to find statistically significant, but not clinically relevant, differences between groups, such as propofol doses or depth of sedation. Moreover, the possibility of interrupting the administration of propofol up to cecal intubation, at the discretion of the endoscopist, might have biased the final conclusions of the study, despite both groups had a similar rate of interruption at cecum. We usually provide individualized regimen sedations and as such, the target of sedation (moderate or deep) and consequently, propofol doses, depend on patient movements and posturing during the procedure. If comfort is guaranteed under moderate sedation, we do not intend to progress to deep sedation using propofol. Once reached the cecum, we usually stop propofol administration if therapeutic maneuvers are not expected, making the withdrawal period coincide with the drug wash-out phase. In our experience, this strategy ensures moderate sedation during most of the withdrawal period and allows shortening recovery times, without impacting on satisfaction rates significantly. Finally, there were only 3 assessments of sedation levels along the procedure and this might have biased as well the results obtained in the study.
In conclusion, both intermittent boluses and pump continuous infusion with propofol were safely administered by trained nurses and endoscopists, providing successful sedation for colonoscopy. Propofol PCI might have several advantages, such as promoting a deeper and longer moderate sedation and avoiding having a nurse exclusively devoted to propofol administration. Further controlled studies are required to investigate whether administering propofol by PCI might shorten the burden in nursery for colonoscopies.
Acknowledgements
We are indebted to the hard work and dedication of the endoscopy nurses (Rosa Benito, Elena Luengo, Sara Martínez y Paula Santisteban) who participated in this study.
References
1. Cohen LB. Sedation issues in quality colonoscopy. Gastrointest Endosc Clin N Am 2010;20:615-27. [ Links ]
2. Vargo JJ, Cohen LB, Rex DK, Kwo PY. Position statement: Nonanesthesiologist administration of propofol for GI endoscopy. Am J Gastroenterol 2009;104:2886-92. [ Links ]
3. Dumonceau JM, Riphaus A, Aparicio JR, Beilenhoff U, Knape JT, Ortmann M, et al. European Society of Gastrointestinal Endoscopy, European Society of Gastroenterology and Endoscopy Nurses and Associates and the European Society of Anaesthesiology Guideline: Non-anesthesiologist administration of propofol for GI endoscopy. Endoscopy 2010;42:960-74. [ Links ]
4. Singh H, Poluha W, Cheung M, Choptain, Baron KI, Taback SP. Propofol for sedation during colonoscopy. Cochrane Database Syst Rev 2008;4:CD006268. [ Links ]
5. Rex DK, Deenadayalu VP, Eid E, Imperiales TF, Walker JA, Sandhu K, et al. Endoscopist-directed administration of propofol: A worldwide safety experience. Gastroenterology 2009;137:1229-37. [ Links ]
6. Pohlmann S, Herden HN, Hagenmüller F. Propofol narcosis for endoscopy -more dangerous than midazolam? Bildgebung 1993;60(Supl. 1):61-3. [ Links ]
7. Külling D, Fantin AC, Biro P, Bauerfeind P, Fried M. Safer colonoscopy with patient-controlled analgesia and sedation with propofol and alfentanil. Gastrointest Endosc 2001;54:1-7. [ Links ]
8. Martínez JF, Aparicio JR, Compañy L, Ruiz F, Gómez-Escolar L, Mozas I, et al. Safety of continuous propofol sedation for endoscopic procedures in elderly patients. Rev Esp Enf Dig 2011;103:76-82. [ Links ]
9. Igea F, Gonzalez-Huix F, Figa M, Huertas C. Propofol sedation in colonoscopy: Bolus Administered by a nurse (BAN) vs pump continuous infusion (PCI) controlled by the endoscopist. It is possible to replace the nurse? Gastrointest Endosc 2008;67:AB248. [ Links ]
10. Cohen LB, Dubovsky AN, Aisenberg J, Miller KM. Propofol for endoscopic sedation: A protocol for safe and effective administration by gastroenterologists. Gastrointest Endosc 2003;58:725-32. [ Links ]
11. Cohen LB, Hightower CD, Wood DA, Miller KM, Aisenberg J. Moderate level sedation during endoscopy: A prospective study using low-dose propofol, meperidine/fentanyl and midazolam. Gastrointest Endosc 2004;59:795-803. [ Links ]
12. Cohen LB, Ladas SD, Vargo JJ, Paspatis GA, Bjorkman DJ, Van der Linden P, et al. Sedation in digestive endoscopy: The Athens international position statements. Aliment Pharmacol Ther 2010;32:425-42. [ Links ]
13. Külling D, Bauerfeind P, Heuss LT. How much personnel and monitoring are really necessary for non-anesthesiologist administration of propofol for gastrointestinal endoscopy? Endoscopy 2011;43:636. [ Links ]
14. Walder B, Riphaus A, Dumonceau JM. Minimal personnel and monitoring requirements for safe nonanesthesiologist administration of propofol (NAAP). Endoscopy 2011;43:637-8. [ Links ]
15. Riphaus A, Geist C, Schrader K, Martchenko K, Wehrmann T. Intermittent manually controlled versus continuous infusion of propofol for deep sedation during interventional endoscopy: a prospective randomized trial. Scand J Gastroenterol 2012;8-9:1078-85. [ Links ]
16. Molina-Infante J, Dueñas-Sadornil C, Mateos-Rodriguez JM, Perez-Gallardo B, Vinagre-Rodríguez G, Hernandez-Alonso M, et al. Nonanesthesiologist-administered propofol versus midazolam and propofol, titrated to moderate sedation, for colonoscopy: a randomized controlled trial. Dig Dis Sci 2012;57:2385-93. [ Links ]
17. Riphaus A, Macias-Gomez C, Deviere J, Dumonceau JM. Propofol, the preferred sedation for screening colonoscopy, is underused. Results of an international survey. Dig Liv Dis 2012;44:389-92. [ Links ]
18. Poincloux L, Laquiere A, Bazin JE, Monzy F, Artigues F, Bonny C, et al. A randomized controlled trial of endoscopist vs. anaesthetist-administered sedation for colonoscopy. Dig Liver Dis 2011;43:553-8. [ Links ]
![]() Correspondence:
Correspondence:
Jesús M. González-Santiago.
Department of Gastroenterology.
Hospital San Pedro de Alcántara.
C/ Pablo Naranjo, s/n
10003 Cáceres, Spain
e-mail:
jmgonzalezsantiago@gmail.com
Received: 05/02/2013
Accepted: 25/07/2013