My SciELO
Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista Española de Enfermedades Digestivas
Print version ISSN 1130-0108
Rev. esp. enferm. dig. vol.108 n.8 Madrid Aug. 2016
https://dx.doi.org/10.17235/reed.2016.4103/2016
Usefulness of percutaneous elastography by acoustic radiation force impulse for the non-invasive diagnosis of chronic pancreatitis
Utilidad de la elastografía percutánea con la técnica de radiación acústica de la fuerza de impulso para el diagnóstico no invasivo de la pancreatitis crónica
Camilo Julio Llamoza-Torres1, María Fuentes-Pardo2, Francisco Javier Álvarez-Higueras3, Fernando Alberca-de-las-Parras3,4 and Fernando Carballo-Álvarez3,4
1Unit of Digestive Diseases. Department of Internal Medicine. Hospital General Universitario Rafael Méndez. Lorca, Murcia. Spain.
2Family and Community Medicine. Área III de Salud de la Región de Murcia. Murcia, Spain.
3Department of Digestive Diseases. Unit of Clinical Management of Digestive Diseases. Hospital Clínico Universitario Virgen de la Arrixaca. Murcia, Spain.
4Instituto Murciano de Investigación Biosanitaria (IMIB-Arrixaca). Murcia, Spain.
ABSTRACT
Objective: To determine the accuracy of the acoustic radiation force impulse (ARFI) technique for the diagnosis of chronic pancreatitis.
Materials and methods: We present an observational, single-center study that included patients with suspected chronic pancreatitis in the period between October 2012 and September 2015 who underwent endoscopic ultrasound (EUS) and were classified according to the standard and Rosemont criteria. The same group of patients was assessed by the ARFI technology using ACUSON S2000™ equipment with Virtual Touch™ Quantification software for the assessment of pancreatic stiffness by obtaining the shear wave velocity (SWV) in the head, body and tail of the pancreas. Data were analyzed using ANOVA and nonparametric correlation methods. Diagnostic accuracy was obtained by analyzing receiver operating characteristic (ROC) curve.
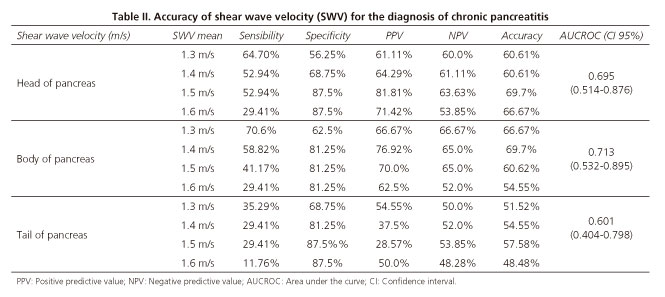
Results: A total of thirty-three patients were studied (45.5% women); mean age was 58.3 ± 11.8 years. Seventeen with a diagnosis of chronic pancreatitis and sixteen classified as normal according to standard criteria. A significant difference was detected between the means of SWV in pancreatic body of patients without (SWV: 1.27 m/s) and with chronic pancreatitis (SWV 1.57 m/s), p = 0.037. The area under the ROC curve was 71.3% (95% CI 0.532-0.895) and the accuracy of ARFI for diagnosing chronic pancreatitis was 69.7% for a SWV of 1.4 m/s in the pancreatic body. The SWV means in head (r = 0.421, p < 0.05) and body (r = 0.455, p < 0.05) of the pancreas showed a significant correlation with the number of EUS criteria for chronic pancreatitis.
Conclusion: Quantification of pancreatic stiffness with ARFI elastography technique has proven to be useful for the non-invasive diagnosis of chronic pancreatitis.
Key words: Chronic pancreatitis. Elastography. Acoustic radiation force impulse.
RESUMEN
Objetivo: determinar la precisión diagnóstica con la técnica de la radiación acústica de la fuerza de impulso (ARFI) para el diagnóstico de pancreatitis crónica (PC).
Material y métodos: estudio observacional, monocentro, de pacientes a quienes, por sospecha de pancreatitis crónica entre octubre de 2012 y septiembre de 2015, se les realizó un estudio por ultrasonografía endoscópica (USE) y se les clasificó según los criterios estándar y de Rosemont. Evaluamos los mismos pacientes mediante un equipo de ultrasonido ACUSON S2000™ equipado con el software Virtual Touch™ Quantification para obtener la velocidad de onda de corte (Vc) en la cabeza, cuerpo y cola del páncreas. Los datos fueron analizados mediante análisis de la varianza y técnicas de correlación no paramétricos; la precisión diagnóstica fue obtenida mediante el análisis de las curvas ROC.
Resultados: evaluamos 33 pacientes (45,5% mujeres) con media de edad de 58,3 ± 11,8 años. Diecisiete con PC y dieciséis clasificados como normal según los criterios estándar. Detectamos diferencias significativas en las medias de Vc en cuerpo de páncreas de pacientes sin (1,27 m/s) y con PC (1,57 m/s) (p = 0,037). El área bajo la curva ROC fue de 0,713 (IC 95% 0,532-0,895) y la precisión diagnóstica, de 69,7% para una Vc de 1,4 m/s en cuerpo pancreático. La media de la Vc en la cabeza (r = 0,421/p < 0,05) y cuerpo de páncreas (r = 0,455/p < 0,05) mostró correlación significativa con el número de criterios USE para PC.
Conclusión: la cuantificación percutánea de la rigidez pancreática mediante la técnica ARFI resulta de utilidad para el diagnóstico no invasivo de la pancreatitis crónica.
Palabras clave: Pancreatitis crónica. Elastografía. Radiación acústica de la fuerza de impulso.
Introduction
Chronic pancreatitis (CP) is a complex chronic inflammatory process of the pancreas, characterized by the presence of chronic inflammatory infiltrates. The activation of pancreatic stellate cells produces parenchymal fibrosis and progressive loss of acinar cells and, to a lesser extent, of the islets of Langerhans (1-5). Endoscopic ultrasound (EUS) has become the technique of choice for the diagnosis of pancreatic disease such as CP, especially in early stages, in which other methods do not achieve the same accuracy (1-6).
Morphological criteria that characterize the disease by EUS have been established, dividing them into parenchymal and ductal types; the presence of multiple criteria leads to a definitive diagnosis in clinical practice. A cut-off of four criteria (3) is often used. The standard classification consists of 9 pancreatic criteria: hyperechoic foci, hyperecoic strands, lobularity, cysts, calcifications, main duct dilation, dilation of the side branches of Wirsung, irregular hyperechoic main pancreatic duct and pancreatic duct walls (3). The hypothesis that all these criteria have the same value for the diagnosis was proposed in the Rosemont classification (4), which defines the EUS-based criteria for CP and their specific validity, establishing different patients groups. However, the interobserver variation of this classification does not improve the aforementioned (3-5).
There is a significant correlation between the number of criteria of CP by EUS and the degree of pancreatic fibrosis histologically evaluated (7-11). Another way to indirectly assess the degree of tissue stiffness is by elastography, which is the representation of images of the mechanical properties of objects (elasticity) when exposed to a force tending to deform (12). Elastography can be classified based on the physical measurement valued at: a) strain or deformation; and b) wave velocity distortion or shear wave velocity (SWV); or depending on the driving method used: a) by manual compression (using direct force by hand or cardiovascular pulsations); b) acoustic radiation force impulse (ARFI); and c) by mechanical impulse (e.g., Fibroscan®) (12,13).
The literature on pancreatic elastography has been evaluated by three different approaches: percutaneous, endoscopic and magnetic resonance imaging (14). In 2005, Hirooka et al. published ??the first elastographic report of the pancreas by EUS-elastography (15), and Uchida et al. by percutaneous elastography (16), both using qualitative methods. The ARFI technique, developed by the group of Katty Nightingale (17), is a dynamic method that produces a mechanical tissue excitation by acoustic pulses of short duration (< 1 ms) in a region of interest (ROI), which produce a tissue distortion and waves transverse cut to ROI (a defined area of 10 x 6 mm). SWV is expressed in meters per second (m/s) with a range of from 0.5 to 5.5 m/s (18).
Reports on pancreatic elastography by ARFI technique have been increasing over the past 3 years (12-22), having noted that currently it can only be used with a percutaneous ultrasound equipment and not by EUS. Yashima et al. (19) presented the first study of diagnostic accuracy by ARFI between healthy volunteers and patients with chronic pancreatitis, taking standard magnetic resonance imaging (MRI), MR cholangiopancreatography (MRCP) and/or endoscopic retrograde cholangiopancreatography (ERCP) as a reference, and describing for SWV of 1.4 m/s sensitivity and specificity of 75% and 72%, respectively. Goya et al. (20) also evaluated the diagnostic accuracy for acute pancreatitis at the time of hospital admission, describing for SWV of 1.63 m/s sensitivity and specificity of 100% and 98%, respectively. Furthermore, Mateen et al. (21) used the same technique to evaluate 166 individuals diagnosed with acute, chronic pancreatitis and normal controls, and were unable to differentiate healthy controls from chronic pancreatitis, only distinguishng acute pancreatitis.
Pancreatic elastography associated with EUS is currently considered to be a complementary and useful tool in diagnosing CP, and could even classify patients according to severity (23,24). However, whether less invasive capacity techniques, such as percutaneous ARFI based elastography, could be equally relevant needs to be systematically determined. In this sense, the present study aims to evaluate the diagnostic accuracy of this technique for the diagnosis of CP.
Materials and methods
Descriptive, observational, cross, open-label, single-center study that evaluates patients over an 18-year period with suspected chronic pancreatitis by EUS from October 2012 to September 2015 in the Department of Digestive Diseases of a universitary general hospital. Inconclusive cases were evaluated with a secretin-stimulated MRCP (s-MRCP) and a contrast-enhaced pancreas MRI with intravenous gadolinium. We excluded patients with pancreatic focal lesions, biliopancreatic prosthesis or previous pancreatic surgical procedure, and those who declined to participate.
EUS was performed mostly under deep sedation assisted by a specialist in anesthesia. EUS was performed by using the radial EUS probe Olympus GF-UE160-AL5 attached to the Aloka console model Alpha7 (although a radio equipment Olympus GF-UM 160 and mini-console EXERA EU-M160 was used in the first year). This equipment did not have elastography software. The use of a contrast agent was not contemplated.
CP was determined by the type and number of standard criteria described by Wiersema et al. (3), defined as the presence of > 4 criteria and non-diagnostic with < 4 criteria, except for those who met the criteria for s-MRCP using the Cambridge classification (6), peak of pancreatic contrast enhancement in the venous or portal phase, or gland signal intensity ratio after intravenous gadolinium administration (enhanced/unenhanced) lower than 1.7. The severity of chronic pancreatitis according to Rosemont criteria (4) was also evaluated.
For performing percutaneous elastography (ARFI) we used ACUSON S2000™ equipment with Virtual Touch™ Quantification (VTQ) mode (Siemens Medical Solutions USA, Inc.). All patients were fasting for at least 6 hours and examined whilst lying in the decubitus position by a convex 1.5-6 MHz 6C1 HD transducer placed on the epigastric region. The patient was asked to relax and keep breathing in for each measurement. Ten measurements of ARFI-VTQ were made in the head, body and tail of the pancreas after a defined margin and echotexture of the same B-mode image was obtained to minimize interposition of gas (mainly gastric chamber) and secretions between the probe and ROI.
The shear wave velocity was expressed in m/s with a theoretical range of 0.5 to 5.5 m/s. The ROI allows measurements in depth of up to 6 cm. In the case of incorrectly recorded measurement, results were given as X.XX m/s. The VTQ software has a report function, whereby the values of a plurality of measurements made in one place are automatically averaged and presented as the mean, median, standard deviation and interquartile range. Measurements were included in the analysis with a success rate of at least 60% for each pancreatic area. The time spent per patient was 10 to 15 minutes.
Continuous variables were expressed as means and standard deviation (SD), discrete variables as proportions or rates, and categorical variables in proportions. For data with a non-normal distribution, non-parametric tests were used. The diagnostic accuracy was calculated after the analysis of the receiver operating characteristic (ROC) curve. For the association between SWV and number of total parenchymal and ductal EUS criteria, the Spearman correlation coefficient was obtained when the data did not follow a normal distribution. Elasticity values in the different groups of the Rosemont classification were compared by one-way single-factor analysis of variance (ANOVA) when following a normal distribution of data by the Kolmogorov-Smirnov test. Heterogeneity of variances was evaluated by the Levene statistical test; post hoc analysis was performed by the Games-Howell test to assess differences between groups. Statistical analyses were performed using the software package version 21.0 IBM® SPSS® Statistics for Macintosh®.
The study was approved by the local research committee and was conducted in accordance with the Declaration of Helsinki. All participants completed and signed a written informed consent form.
Results
From October 2012 to September 2015, a total of 53 patients suspicious of CP were included in the study. Thirty-one patients were diagnosed with CP; 30 patients by EUS and 1 patient by s-MRCP 1 (Cambridge 3).
Of the 31 patients diagnosed with CP, 14 patients were excluded (1 refused the elastography for personal reasons, 6 were unable go to the hospital due to work-related issues, 4 had biliopancreatic prosthesis and 3 had undergone surgical intervention due to local complications of CP). Of the 22 patients with a normal pancreas by EUS, there was one death due to variceal upper gastrointestinal bleeding, 4 declined to participate and one patient had a body mass index of 35 and was excluded since it was not possible to reach the pancreas with ROI due to its deep location; thus 16 people with normal EUS findings were finally evaluated.
Baseline characteristics of the study population
Of the 53 patients with suspected chronic pancreatitis, 33 patients were evaluated. There were 15 males and 18 females with a mean age of 58.3 ± 11.8 years, range 32-84. All of them reported a history of epigastric discomfort or post prandial distress in the upper abdomen and 60.6% of them (20 people) had epigastric pain as a predominant symptom. Chronic diarrhea defined as increased defecation rate of more than 2 weeks duration and steatorrhea were reported in only 6.06% (2 patients).
At the time of exploration, 24 patients (72.7%) were non-drinkers or previous light drinkers with alcohol consumption lower than 20 g/day; 19 patients (57.6%) did not smoke. Eight patients (24.24%) presented previous episodes of acute pancreatitis and 7 (21.2%) had diabetes mellitus type 2. Only 3 patients (9.1%) had a previous technique image with pancreatic calcifications.
In the group diagnosed with CP, the mean age was 58.76 years (SD ± 13.58) whereas in the non-CP group it was 57.81 (SD ± 10.08), which was not a significant difference (p = 0.821). It was possible to identify a relevant risk factor in 13 cases (76.5%) diagnosed with CP (4 [23.5%] associated with tobacco, 7 [41.2%] associated with tobacco and alcohol, 2 [11.8%] related to recurrent pancreatitis and hyperlipidemia); in 4 patients (23.5%) it was not possible to determine the cause.
Endoscopic characteristics
Of the sixteen patients with CP evaluated by EUS, 8 (50%) had four EUS criteria; 6 (37.5%) had 5, and 2 (12.5%) presented 7 criteria. Likewise, using the Rosemont classification, 7 patients were diagnosed as consistent with CP (43.8%), 6 (37.5%) as suggestive of CP, and 3 as indeterminate for CP (18.8%). These last 3 patients had four standard EUS criteria for CP (3 minor parenchymal criteria and 1 minor ductal criterion). In the group of patients with non-CP by EUS (16 patients), 2 (12%) had only 1 or 2 parenchymal EUS criteria; the remainder (14 patients: 87.5%) did not present any alteration by EUS.
Pancreatic elastography by acosutic radiation force impulse (ARFI)
Mean, median, standard deviation and range were obtained in each of the 30 measurements of SWV by subject; 10 in head, body and tail of the pancreas, respectively.
Significant differences were found between the means of SWV of the body of pancreas in patients with CP and non-CP (p = 0.037); although not for the measurements made in the head (p = 0.058) and pancreatic tail (p = 0.326), taking into account the statistical Mann-Whitney U for samples with heterogeneity of variances by the Levene test (Table I).
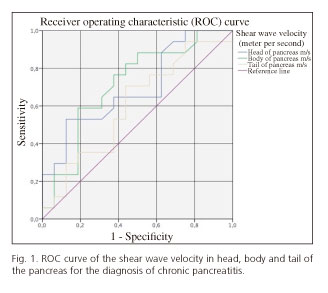
ROC curve analysis of SWV in the head, body and tail of the pancreas was performed and an area under the curve (AUC) of 0.695 (95% CI, 0.514-0.876), 0.713 (95% CI 0.532-0.895) and 0.601 (95% CI 0.404-0.798), respectively, was obtained (Fig. 1).
The diagnostic accuracy of SWV for the diagnosis of CP is shown in table II. The ROC curves analysis, by applying the cut-off for SWV of 1.4 m/s measured at the pancreatic body, had a sensitivity of 58.8%, a 76.92% positive predictive value and a 65.0% negative predictive value for the diagnosis of CP, with an area under the curve (AUCROC 95% CI) of 0.713 (0.53-0.89).
The correlation between the total number of EUS criteria and SWV in the head, body and tail was evaluated, with a Spearman correlation coefficient rs of 0.417, 0.367 and 0.229, respectively (Table III). The patient with CP diagnosed by s-MRCP and contrasted with gadolinium was excluded from the analysis.
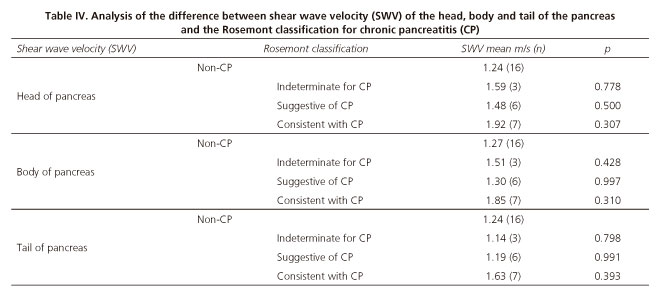
SWV differences in the head, body and tail of the pancreas between patients with and without CP according to the Rosemont classification were evaluated. Heterogeneity of variances was found by the Levene statistic, so a post ad hoc analysis was performed using Games-Howell corrections (Table IV).
No differences were detected in the SWV of the head, body and tail of the pancreas in the group of patients with CP according to the Rosemont classification when etiology was evaluated. SWV in different parts of the pancreas in patients with moderate/high alcohol consumption was similar to that of patients with none or light alcohol consumption. Likewise, no differences between the number of EUS criteria among those with minimal or no alcohol consumption (< 20 g/day) and moderate to severe (> 20 g/day) consumption were found.
SWV in CP did not depend on age. Neither SWV nor the number of EUS criteria was significantly different when patients were divided into 2 categories of under 65 and > 65 years of age.
Discussion
We present the results of the diagnostic accuracy of a non-invasive method based on percutaneous elastography for the diagnosis of chronic pancreatitis. Of the 31 cases of CP recruited, 14 were excluded for different reasons, half of them for local complications of the disease, so the most severe cases of CP could not be evaluated by percutaneous elastography and could have given higher values in the elastographic measurements, which could have improved diagnostic accuracy.
SWV was obtained from measurements taken at the three main parts of the pancreas (head, body and tail) in both normal and CP patients. Between the three parts of the pancreas of non-CP group patients there were no differences. However, those with CP presented differences, although they were not significant, probably due to the limited number of patients.
There were differences in SWV between different parts of the pancreas of patients with and without CP, but significant differences were only found in the measurements made in the body of pancreas (1.27 m/s vs. 1.57 m/s, p = 0.037) (Table I). The sensitivity described for conventional ultrasonography ranged from 52% to 86% (24) mainly due to the presence of gross morphological changes such as calcifications or marked dilation of the pancreatic ducts, mainly reflecting assessments made in advanced stages. It should be noted that, in our series, the mean number of EUS criteria was 4.6 and more advanced stages were excluded as they did not meet the inclusion criteria. The optimal number of parenchymal and ductular abnormalities for a diagnosis of CP by EUS have been established with a high sensitivity and specificity (3,4), but when evaluating patients with early stage disease sensitivity it was reduced to 86% (24).
Using histology as a reference, EUS criteria have been evaluated for non-calcifying CP, reporting sensitivities between 75-91% and 85-100% of specificity (7-11). But these studies have many limitations such as the inclusion of patients with a high probability pre-test for CP in the presence of high risk factors (7), including patients with surgical resection for cancer (8), and variability in the number of criteria used to define CP by EUS and histology (9). Our series found a significant correlation between the number of EUS criteria, mainly parenchymal and SWV in the head and body of the pancreas (Table III). However, when CP severity was evaluated, no significant differences were found for SWV at each stage (Table IV). Nevertheless, it can be seen that there is a general tendency to differentiate non-CP from advanced CP (Fig. 2). This crude differentiation is reminiscent of the discriminative power of the studies of liver with transient elastography, clearly differentiating the absence of fibrosis (F0) and early (F1) from advanced stages (F4), although it is still difficult to determine the intermediate stages (F2,F3).
Elastography associated with EUS is a technique that shows high accuracy for the differential diagnosis of solid tumors and inflammatory pancreatic masses with sensitivity of 100% and specificity of 88-96% (23-28). Itokawa et al. (27) introduced the concept of strain ratio (SR) as a semiquantitative method to objectively evaluate tissue hardness, which is simply the ratio of the distortions produced by a force on two adjacent areas. In 2013, Iglesias-García et al. (23) obtained a diagnostic accuracy of 91.1% by EUS-elastography using the SR for CP with a cut-off of 2.25, describing a direct correlation between the SR and the different stages of CP according to the Rosemont classification. EUS-elastography, despite obtaining a high diagnostic accuracy using indirect quantification methods, is a dependent operator technique and has a relative degree of invasiveness.
In 2012, Yashima et al. (19) presented a study with 52 healthy subjects and 46 patients with CP using the ARFI technology. The cut-off values to differentiate healthy patients had a sensitivity, specificity, positive predictive value and negative predictive value of 75%, 72%, 69% and 78%, respectively. However, the technique used to diagnose CP was based on ERCP and/or MRI analysis, without a morphological description by EUS, as more than 70% of CP patients presented an advanced stage. Following the analysis of the inclusion criteria, the lack of morphological data by EUS in patients classified as normal but with a history of abdominal discomfort in the study by Mateen et al. (21) could explain the absence of differences in the SWV between these patients and those classified as CP, meaning there was an initial unintentional misclassification.
We could not avoid selection bias because patients with local complications, requiring some pancreatic surgery or endoscopic procedures, which could influence pancreatic stiffness data, were excluded. Another bias of our study was the fact that it was a non-blinded single center study. This is mainly due to the clinical setting of the practice and to the fact that the disease has a low incidence and prevalence. Likewise, the period between performance of the EUS and elastography should be considered.
Pancreatic fibrosis, which is probably the main cause of pancreatic stiffness in CP, is not considered as synonymous with disease, since there are other causes of pancreatic fibrosis, including advanced age, alcohol consumption, tobacco, etc. In our group of patients with CP, no differences were detected between age (over or under 65 years), alcohol consumption (less than or > 20 g/day), etiology of CP, and the mean of SWV and the number of EUS criteria, which may be due to the limited number of patients included in the study.
In conclusion, despite having a small number of patients, we could significantly correlate morphological findings obtained by EUS for CP with SWV by non-invasive percutaneous elastography. If diagnostic accuracy of CP for the latter technique was improved with a larger number of patients, the absence of adverse effects and excellent tolerance would justify its widespread use immediately after a suspected diagnosis, and even for the follow up of this disease.
Acwnowledgments
We thank all the staff of the Department of Digestive Diseases of the Hospital Universitario Rafael Méndez, who facilitated the implementation of this study.
References
1. Luaces-Regueira M, Iglesias-García J, Lindkvist B, et al. Smoking as a risk factor for complications in chronic pancreatitis. Pancreas 2014;43:275-80. DOI: 10.1097/01.mpa.0000437324.52598.ee. [ Links ]
2. Domínguez-Muñoz E, Lucendo A, Carballo F, et al. Estudio español multicéntrico de estimación de la prevalencia e incidencia de la pancreatitis crónica y sus complicaciones. Rev Esp Enferm Dig 2014;106(4):239-45. [ Links ]
3. Wieserma MJ, Hawes RH, Lehman GA, et al. Prospective evaluation of endoscopic ultrasonography and endoscopy retrograde cholangiopancreatography in patients with chronic abdominal pain of suspected pancreatic origin. Endoscopy 1993;25:555-64. DOI: 10.1055/s-2007-1010405. [ Links ]
4. Catalano MF, Sahai A, Levy M, et al. EUS-based criteria for the diagnosis of chronic pancreatitis: The Rosemont classification. Gastrointest Endosc 2009;69:1251-61. DOI: 10.1016/j.gie.2008.07.043. [ Links ]
5. Stevens T, López R, Adler DG, et al. Multicenter comparison of the interobserver agreement of standard EUS scoring and Rosemont classification scoring for the diagnosis of chronic pancreatitis. Gastrointest Endosc 2010:71:519-26. DOI: 10.1016/j.gie.2009.10.043. [ Links ]
6. Balci C. MRI assessment of chronic pancreatitis. Diagn Interv Radiol 2011;17:249-54. [ Links ]
7. Albashir S, Bronner MP, Paris MA, et al. Endoscopic ultrasound, secretin endoscopic pancreatic function test, and histology: Correlation in chronic pancreatitis. Am J Gastroenterol 2010;105:2498-503. DOI: 10.1038/ajg.2010.274. [ Links ]
8. Varadarajulu S, Eltoum I, Tamhane A, et al. Histopathologic correlates of noncalcific chronic pancreatitis by EUS: A prospective tissue characterization study. Gastrointest Endosc 2007;66:501-9. DOI: 10.1016/j.gie.2006.12.043. [ Links ]
9. Chong AK, Hawes RH, Hoffman BJ, et al. Diagnostic performance of EUS for chronic pancreatitis: A comparison with histopathology. Gastrointest Endosc 2007;65:808-14. DOI: 10.1016/j.gie.2006.09.026. [ Links ]
10. Le Blanc LJ, Chen JH, Al Haddad M, et al. Endoscopy ultrasound and histology in chronic pancreatitis: How are they associated? Pancreas 2014;43(3):440-4. [ Links ]
11. Vega-Peralta J, Attam R, Arain MA, et al. Correlation of EUS with histopathology in 50 patients undergoing total pancreatectomy (TP) with islet autotransplantation (IAT) for minimal change chronic pancreatitis (MCCP). Gastrointest Endosc 2011;73Suppl:AB324. DOI: 10.1016/j.gie.2011.03.675. [ Links ]
12. Shiina T. JSUM ultrasound elastography practice guidelines: Basics and terminology. J Med Ultrasonics 2013:40:309-23. DOI: 10.1007/s10396-013-0490-z. [ Links ]
13. Itoh Y, Takehara Y, Kawase T, et al. Feasibility of magnetic resonance elastography for the pancreas at 3T. J Magn Reson Imaging 2015 Jul 7. DOI: 10.1002/jmri.24995. [ Links ]
14. Hirooka Y, Kuwahara T, Irisawa A, et al. JSUM ultrasound elastography practice guideline: Pancreas. J Med Ultrasonics 2015;42:151-74. DOI: 10.1007/s10396-014-0571-7. [ Links ]
15. Hirooka Y, Itoh A, Hashimoto S, et al. Utility of EUS:elastography in the diagnosis of pancreatic diseases. Gastrointest Endosc 2005;61:AB282. DOI: 10.1016/S0016-5107(05)01447-1. [ Links ]
16. Uchida H, Hirooka Y, Itoh A, et al. Utility of elastography in the diagnosis of pancreatic diseases using transabdominal ultrasonography. Gastroenterology 2005;128:A536. DOI: 10.1016/S0016-5107(05)01447-1. [ Links ]
17. Gennisson JL, Deffieux T, Fink M, et al. Ultrasound elastography: Principles and techniques. Diagnostic and Interventional Imaging 2013;94:487-95. DOI: 10.1016/j.diii.2013.01.022. [ Links ]
18. Friedrich-Rust M, Schlueter N, Smaczny C, et al. Non-invasive measurement of liver and pancreas fibrosis in patients with cystic fibrosis. J Cyst Fibros 2013;12(5):431-9. DOI: 10.1016/j.jcf.2012.12.013. [ Links ]
19. Yashima Y, Sasahira N, Isayama H, et al. Acoustic radiation force impulse elastography for noninvasive assessment of chronic pancreatitis. J Gastroenterol 2012:47:427-32. DOI: 10.1007/s00535-011-0491-x. [ Links ]
20. Göya C, Hamidi C, Hattapoğlu S, et al. Use of acoustic radiation force impulse elastography to diagnose acute pancreatitis at hospital admission. J Ultrasound Med 2014;33:1453-60. DOI: 10.7863/ultra.33.8.1453. [ Links ]
21. Mateen M, Muheet K, Mohan R, et al. Evaluation of ultrasound based acoustic radiation force impulse (ARFI) and eSie touch Sonoelastography for diagnosis of inflammatory pancreatic diseases. J Pancreas 2012;13(1):34-44. [ Links ]
22. Kawada N, Tanaka S, Uehara H, et al. Potencial use of point shear wave elastography for the pancreas: A single center prospective study. Eur J Radiol 2014;83:620-4. DOI: 10.1016/j.ejrad.2013.11.029. [ Links ]
23. Iglesias-García J, Domínguez-Muñoz E, Castiñeira-Alvariño M, et al. Quantitative elastography associated with endoscopic ultrasound for the diagnosis of chronic pancreatitis. Endoscopy 2013;45:781-8. DOI: 10.1055/s-0033-1344614. [ Links ]
24. Kelly KA, Hollingsworth MA, Brand RE, et al. Advances in biomedical imaging, bioengineering, and related technologies for the development of biomarkers of pancreatic diseases. Pancreas 2015;44(8):1185-94. DOI: 10.1097/MPA.0000000000000552. [ Links ]
25. Giovannini M, Hookey LC, Bories E, et al. Endoscopic ultrasound elastography: The first step towards virtual biopsy? Preliminary results in 49 patients. Endoscopy 2006;38:344-8. DOI: 10.1055/s-2006-925158. [ Links ]
26. Janssen J, Schlörer E, Greiner L. EUS elastography of the pancreas: Feasibility and pattern description of the normal pancreas, chronic pancreatitis, and focal pancreatic lesions. Gastrointest Endosc 2007;65:971-8. DOI: 10.1016/j.gie.2006.12.057. [ Links ]
27. Itokawa F, Itoi T, Sofuni A, et al. EUS elastography combined with the strain ratio of tissue elasticity for diagnosis of solid pancreatic masses. J Gastroenterol 2011;46:843-53. DOI: 10.1007/s00535-011-0399-5. [ Links ]
28. Mei M, Ni J, Liu D, et al. EUS elastography for diagnosis of solid pancreatic masses: A meta-analysis. Gastrointest Endosc 2013;77(4):578-89. DOI: 10.1016/j.gie.2012.09.035. [ Links ]
![]() Correspondence:
Correspondence:
Camilo Julio Llamoza-Torres.
Unit of Digestive Diseases. Department of Internal Medicine.
Hospital General Universitario Rafael Méndez.
Ctra. Nacional 340, km. 589. 30817 Lorca, Murcia. Spain
e-mail: info@llamozatorres.com
Received: 17-11-2015.
Accepted: 12-04-2016











 text in
text in