INTRODUCTION
Esophageal atresia (EA) is a congenital malformation associated with prematurity and tracheoesophageal fistulas. It is mandatory to surgically repair the disease during the first months of life. The development of refractory anastomotic strictures is not infrequent and endoscopic dilation and corticoid injections are usually required. Some authors have reported a complete resolution of dysphagia in children with EA after the placement of esophageal stents. We present a challenging case of a young woman with a surgically treated congenital EA and a severe anastomotic stricture-related relapsing dysphagia that was definitively resolved after sequential Polyflex(tm) stent placement.
CASE REPORT
We present the case of a 23-year-old female who had been previously diagnosed with type 1 EA at birth (1989). A gastrostomy tube was placed in her stomach within 24 hours of birth and also three months later. As a newborn, she underwent a surgical partial esophagectomy with an esophageal-esophageal anastomosis. A mild postsurgical anastomotic leakage was diagnosed and conservatively managed, and oral feeding was started two weeks later. Three months later, the patient was readmitted due to an aspiration broncopneumonia and severe gastroesophageal reflux (GERD). At 14 months of age, the patient was diagnosed with anastomotic stenosis and an important gastroesophageal reflux; bougie dilations were started from an initial diameter of 15 Fr until reaching a maximum diameter of 34 Fr. According to the chronic recurrence of the stricture and the associated GERD, a Nissen fundoplication was performed when she was three years of age. In spite of this surgical procedure, the patient had to be chronically dilated. In 2002, a first hydraulic balloon dilation was performed and the patient was free of dysphagia for almost five years. In 2007, the dysphagia reappeared, caused by an anastomotic stenosis located 26 cm from the teeth. Periodical balloon dilations, up to 18 mm-diameter, were started. After several dilations, the patient was finally referred to our department in 2009. At first, the stricture was successfully dilated several times but the dysphagia recurred. In October 2010, a biodegradable (BD) uncovered stent (X-ELLA, Czech Rep) of 60 x 25-31 mm, which was only available in Spain at that time, was placed. Unfortunately, this stent was not well tolerated and caused a severe thoracic pain of an eight-week duration, probably related to the excessive width of the stent. A computed tomography (CT) scan ruled out a perforation. Three months later, an upper endoscopy revealed that an intense hyperplastic reaction had grown on the upper limit of the stent, with only a mild relief of the dysphagia. Two more dilations had to be performed until the persistent pain was finally relieved. Subsequently, in November 2011, a smaller caliber (18/23 x 90 mm) Polyflex(tm) esophageal stent (Boston Scientific, USA) was inserted with the aim of avoiding pain (Fig. 1). Only mild chest pain was reported by the patient (Fig. 2). After 12 weeks, the stent was retrieved. Hyperplastic tissue had grown both over the upper and the distal esophagus, burying the proximal end of the stent and making its extraction rather tricky. Despite this new grown tissue, the patient had a full resolution of the dysphagia after stent removal. An endoscopic review was performed two weeks later and the insertion of another Polyflex(tm) stent was discarded, due to the fact that the dysphagia had completely disappeared and a significant stricture was not observed. However, the dysphagia started again during the following summer and a new dilation program was scheduled, with a mild success. A second Polyflex(tm) stent (18-23 mm x 90 mm) was placed during December 2012 and the dysphagia decreased significantly. Eight weeks later, an attempt to retrieve the stent failed due to the fact that the upper tip of the stent was buried in the cervical esophagus. Therefore, a coaxial fully covered stent (Ultraflex(r), 24 x 140 mm) was inserted to induce tissue necrosis and facilitate the extraction of the plastic stent. Both stents were finally extracted together with some ease with rat-teeth forceps in February 2013.
Since then, the patient has not presented any dysphagia after five years of follow-up. During this time, two upper endoscopies (Fig. 3) and two barium esophagograms (Fig. 4) have been performed and the stenosis has not recurred. The endoscopy two years after the second Polyflex(tm) extraction in February 2015 shows a complete resolution of the esophageal stricture and hyperplastic tissue remnants at the proximal edge of the stent. There is no stricture on the barium esophagogram four years after stenting in June 2017 and the patient remains asymptomatic.
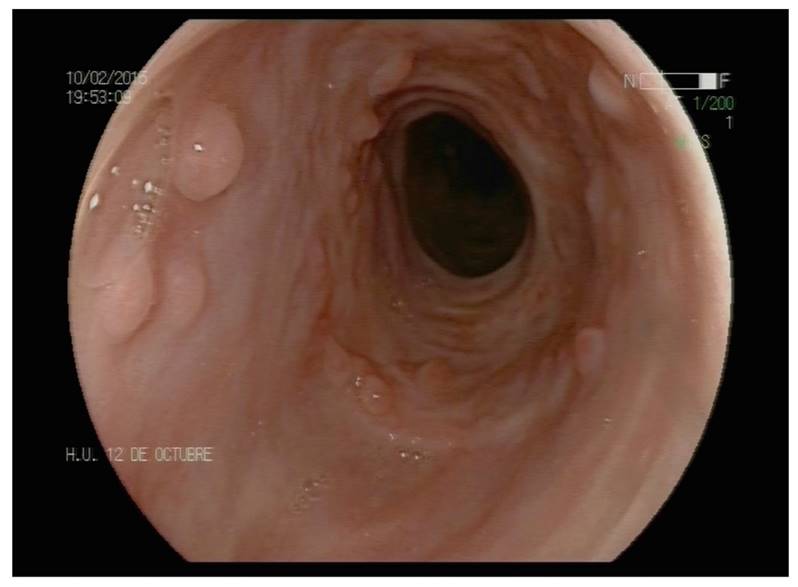
Fig. 3 Endoscopic view two years after the second Polyflex(tm) extraction in February 2015. There is a complete resolution of the esophageal stricture and hyperplastic tissue remnants at the proximal edge of the stent.
DISCUSSION
Esophageal atresia (EA) encompasses a group of congenital anomalies that results in an interruption of the continuity of the esophagus, which occurs in 1/2,500-3,500 of live births and can be associated with tracheoesophageal fistula (86%) 1. It is associated with vertebral, anal, cardiovascular, tracheoesophageal, renal and limb congenital disorders in 50% of cases 2. The absence of esophageal continuity renders the infant unable to swallow saliva and suffer excessive salivation. Oral feeding is impossible and bronchial aspiration is frequent. Surgical correction is mandatory. Several surgical techniques can be performed and if there is a "long gap" between the ends of the esophagus and delayed primary repair should be attempted. Usually, a gastrostomy tube must be inserted to permit feeding. The most usual technique is the reconstruction of the digestive tract via an esophageal-esophageal anastomosis. Several complications can develop after surgical repair: a) early complications, such as anastomotic leakage, anastomotic stricture and recurrent fistula; and b) delayed complications, such as gastroesophageal reflux, tracheomalacia and dysmotility.
Anastomotic leaks occur in 15-20% of the patients and major disruptions can be life-threatening in only one third of cases 3. In contrast, minor leaks can be detected 5-7 days after surgery and tend to seal spontaneously, although increasing the risk of delayed stricture formation. An esophageal-esophageal anastomotic stenosis can develop in 30 to 40% of cases, most of which will usually respond to one or two endoscopic dilatations 4. Risk factors include anastomotic tension, anastomotic leakage and gastroesophageal reflux.
Endoscopic dilations can be performed with progressively larger bougies (Savary-Gillard) or under fluoroscopy with hydraulic balloons. However, stricture recurrence is not infrequent and several other therapies such as steroid injection, local application of mitomycin C and mucosal cutting have been used 5,6. GERD has been implicated in the pathogenesis of anastomotic strictures. Its incidence in post-repair esophageal atresia-transesophageal fistula (EA-TEF) patients is variable (27-85%) and about 20% will require surgical correction 7. Acute or chronic respiratory problems may be suspicious of a significant case of GERD. Recurrent vomiting is also frequent. Proton-pump inhibitors are useful in less than 50% of cases and, sometimes, a partial fundoplication is required. An adequate correction of GERD may lead to a resolution of the relapsing anastomotic stricture formation that maintains refractory to endoscopic dilatations.
The use of stents in esophageal atresia is not rare in children 8. A lot of devices have been used, from fully-covered self-expandable metallic stent to biodegradable and plastic stents 5,6,9. However, there is no consensus with regard to the most appropriate approach and stent for this indication. There are previous reports of polypropylene stents (Polyflex(tm), Boston Sci.) inserted in children suffering from postsurgical dysphagia in the setting of an EA 10, but not in adults. BD stents have been successfully used in children in this setting 8, with the advantage that they do not need to be removed. However, the side effects can be a limiting factor; an excessive stent width may influence thoracic pain severity and currently these stents can be individually customized.
Polyflex(tm) stents have been used for several different indications such as malignant and benign gastrointestinal and colonic strictures, treatment of digestive fistulas and even the retrieval of buried stents. The stents are self-expanding cylinders of polypropylene filaments covered with silicone. The upper tip is conical and the rest is cylindrical. Thus, plastic stents must be loaded in the applicator just before their placement. The diameters and lengths are different (30-42 French and 40-150 mm), so they can adapt to the size of the patient and the length of the stenosis. Dilation is required until a diameter of 1 mm or 3 French less than the open stent diameter is achieved. The stent applicator is inserted under fluoroscopic guidance. The length of the stent should exceed the damaged area by 1 cm. Once the stent is inserted, it produces a progressive radial dilation until it reaches its maximum diameter in 24 hours. Pain can occur but it is not frequent. It is prudent to remove Polyflex(tm) stents after six to eight weeks in order to avoid burying the conical upper tip of the prostheses beyond hyperplastic tissue.
Our patient had been dilated during many years, with disease-free periods during adolescence, and required dilations for almost all her life. The dysphagia decreased dramatically since the first Polyflex(tm) stent was placed and even completely disappeared after the second plastic stent was removed. Both stents were well tolerated with no complications. Accordingly, it can be concluded that the use of sequential Polyflex(tm) stenting may be a safe and appropriate alternative for the treatment of refractory anastomotic strictures in adults with EA. Nevertheless, this kind of stents are expensive, removal can be difficult and they have higher rates of migration than other stents.

















