Introduction
Mesothelioma is a neoplasm that originates in the cells that line serous cavities. The most frequent location of mesothelioma is the pleura, with pleural mesothelioma comprising up to 80% of cases1. Mesothelioma occurs mainly in elderly patients after decades of exposure to asbestos2. The magnitude of the risk of mesothelioma is directly dependent on the level of exposure to asbestos; while minimal and continuous or brief and intense exposures may result in the development of mesothelioma. The nature and morphology of asbestos fibers also has an influence. Cumulative exposure level of at least 0.1 fibers/mL-year is considered sufficient to produce mesothelioma3.
Mesothelioma is a rare tumor but it is only the tip of the iceberg of asbestos-related diseases (ARDs), representing a serious public health problem3. The intrinsic carcinogenicity of asbestos is compounded by its high capacity to alter the clearance mechanisms of the respiratory tract. Consequently, other agents are retained, forming a highly carcinogenic cocktail. The risk of developing mesothelioma, far from being reduced with the passage of time, increases and continues for more than 40 years after exposure4,5. It is suspected that other inflammation-based nonmalignant pathologies may also be related to exposure to this carcinogen, such as chronic obstructive pulmonary disease (COPD), atherosclerosis and autoimmune diseases3. Of the total mesothelioma deaths that occur in the world, 56% occur in Europe. The majority of European countries that have historically used asbestos have high mortality rates due to mesothelioma; in turn, the rate of ARDs is proportional to the tons of asbestos consumed in the preceding decades.
For large industrialized countries, cancer resulting from asbestos exposure represents a problem due to the high morbidity and mortality involved. ARDs impose a high cost on health systems, they are responsible for high morbidity with a reduced life expectancy and have a large impact on patient quality of life. Occupational exposure can extend beyond asbestos workers, applying to all workers who are involved in the processing and final destination of the material as well as to their family members who live in the home, as asbestos fibers can be carried in the clothing and hair of workers, exposing relatives to mesothelioma development6. Due to the high latency from exposure to the appearance of mesothelioma, it is expected that mesothelioma cases and deaths will continue to increase7,8. Given the low frequency of mesothelioma and its close link to asbestos, this tumor is considered a sentinel tumor, a marker of asbestos exposure2,8. Because of its low survival, mortality from mesothelioma can be used to estimate its incidence and therefore the overall burden of the disease9,10.
Asbestos is a mineral, and therefore, geological soils rich in any of its varieties represent a threat to the health of the population settled in these soils. Given that asbestos is present in many types of industries, it is worth asking whether exposure can also occur with the contaminants that these industries discharge in the geographical area where they are located11,12. In this sense, the studies conducted by López-Abente et al.13 and Tarrés et al.14) detected a higher risk of pleural mesothelioma in residential areas near industrial sources of asbestos. It seems that the distance from residential areas to industries that use asbestos and wind direction influence the development of pleural mesothelioma14. The use of spatial regression models can be useful when evaluating the impact of exposure to asbestos11. According to previous studies, non-occupational exposure to asbestos of any type increases the relative risk (RR) of mesothelioma12,15,16.
The Minimum Basic Data Set (MBDS) in Spain has been operating since 1990. It collects information related to hospital discharges so it can provide data about the specialized sanitary activity of hospitals and the sanitary activity in outpatients clinics. The MBDS has been validated as a tool of epidemiological surveillance of cancer17, and also for the incidence of mesothelioma in Italy, but not in Spain18.
The aim of this study was to determine whether the combined analysis of the MBDS and Statistical Bulletins of Death (SBDs) recorded in the Spanish National Statistics Institute (Instituto Nacional de Estadistica - INE) is an effective tool for monitoring the risk of mesothelioma. The setting chosen is a high-population region of Spain, the Community of Madrid (CM), which lacks a population-based cancer registry (PBCR).
Methods
This is a descriptive epidemiological study that used data from three sources: the MBDS for 2016 and 2017 of the CM; the microdata files from the INE for deaths in 2016 and 2017; and the CM 2017 population census available on the INE website19. The MBDS files for 2016 and 2017 of the CM were obtained via the pertinent request mechanism from the General Directorate of Public Health at the Ministry of Health of the CM. The files were provided in .csv format with anonymized identifying patient codes, the month and year of birth and the date of each hospital discharge, as well as codes for the municipality of residence for municipalities with more than 10,000 inhabitants, following the current legal data protection framework. Each record in the MBDS is a hospital stay, so one patient may have several stays. The anonymized identifying patient code is necessary to check duplicate cases. Although the files provided cover all the care activities of public and private healthcare centers located in the CM, both hospital and extrahospital, only the data of patients with a mesothelioma diagnosis code (C45) were provided, so we did not have control of the denominator. The MBDS file includes International Classification of Diseases 10th revision (ICD-10) codes in 21 fields. There are also 22 fields for procedure codes that were not explored.
Using the pertinent request mechanism, supported by the aims of our study as a justification, we sent a request to the INE to provide the microdata files corresponding to the SBDs that include multiple causes of death for the 2016-2017 period. All the mortalities residing in the CM were extracted. The deceased persons with the C45 code in any of the multiple causes of death (basic cause and immediate, intermediate and fundamental causes and of other processes), were extracted separately. Due to the data protection law, deceased the residents of municipalities with less than 10,000 inhabitants did not have their municipality code included in the received files. The microdata type files were provided by the INE in .txt format in chains of 230 characters with a detailed explanation of the design for their proper analysis. The codes for the CM municipalities were obtained from the INE website19.
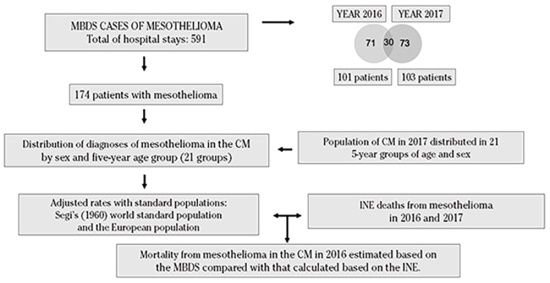
In both series (MBDS cases and INE deaths) the selection criteria was the presence of code C45, specific for mesothelioma in the ICD-10. We checked the five different sites: 45.0 = mesothelioma of pleura, C45.1 = mesothelioma of peritoneum, C45.2 = meso-thelioma of pericardium, C45.7 = mesothelioma of other specified sites, and C45.9 = mesothelioma of unspecified sites. This control had two objectives: to calculate the proportion of pleural mesotheliomas with respect to other known sites and to evaluate the quality of the coding for each of the sources (MBDS and INE), assuming that the exclusive use of codes C45.7 and C45.9, in the absence of other more precise site codes, represented a marker of poor quality coding. To this end, two files with 174 cases and 149 deaths, respectively, were developed in which, in addition to the case/death identification code, five fields that control the C45 code type were incorporated. Figure 1 shows a clarifying algorithm of MBDS and INE data exploitation.
CM: Community of Madrid.
Figure 1. Algorithm of Minimum Basic Data Set (MBDS) and the Spanish National Statistics Institute (INE) data exploitation.
The study variables in both series, MBDS (cases) and INE (deaths), were as follows: 1- distribution of diagnoses of mesothelioma in the CM by sex and five-year age group (21 groups); 2- prevalence of mesothelioma in the CM in 2016 and 2017; 3- incidence of mesothelioma in the CM in 2017; 4- mortality from mesothelioma in the CM in 2016; 5- geographic distribution of cases and deaths according to the municipality of residence codes of the patients; and 6- correlation of the municipal geographic pattern of risk obtained with the MBDS cases with that obtained with the INE deaths for the same period of 2016-2017. Mortality rate (and confidence interval at 95%, 95%CI) from mesothelioma in the CM was estimated as per the MBDS and compared with the one calculated according to the INE. Comparison was only possible for 2016, assuming that those patients registered in the MBDS in 2016 who were not listed in 2017 had died.
To determine the geographical pattern of risk of mesothelioma within the CM, the eight municipalities with more than 10,000 inhabitants with at least five registered cases of mesothelioma in the MBDS were selected. These data were compared with the deaths in the same period (2016-2017) of patients residing in those eight municipalities. A scatter-plot was developed, the Pearson correlation coefficient (r) was calculated, and a risk map was prepared with the Standardized Mortality Ratio (SMR) from INE data.
Data from both files, MBDS (cases) and INE (deaths), were analyzed using the R statistical package and programming language. The Microsoft Excel office suite was also used. This study was evaluated and approved by the Research Ethics and Animal Experimentation Committee of University of Alcala under code CEIG/HU/2018/35 on October 11, 2018.
Results
The MBDS series included 174 unique patients as data was provided with a unique identifying number that eliminates duplicate patients (120 men and 54 women), of whom 113 men and 53 women lived in municipalities with more than 10,000 inhabitants. In 2016, there were 258 hospital admissions and stays of 101 patients. In 2017, there were 333 hospital admissions and stays of 103 patients. Thirty patients were present in both years, and the number of patient incidents in 2017 was 103 - 30 patients = 73 patients. It was assumed that the 71 patients admitted in 2016 who were not admitted in 2017 died.
In this series, 3,539 ICD-10 codes were detected, of which 588 were unique codes. There were five different assigned C45 codes and the distribution of cases by these mesothelioma sites were: for pleural (C45.0), 125 patients, unspecified (C45.9), 43 patients, other unspecified sites (C45.7), 16 patients, peritoneum (C45.1), 16 patients and pericardium (C45.2), 3 patients. Assuming that in 32 patients a specific site of mesothelioma was not provided, the percentage of MBDS records with poor mesothelioma coding quality was 18.39%. Of the 143 patients with a known mesothelioma site, 125 had pleural mesothelioma; thus, this site represents 87.41% of the coded sites.
Table 1. Prevalence rates of mesothelioma in 2016 and 2017, and incidence rates of mesothelioma in the Community of Madrid according to the Minimum Basic Data Set data in 2017
| Prevalence | Incidence | ||||||
|---|---|---|---|---|---|---|---|
| 2016 | 2017 | 2017 | |||||
| Men | Women | Men | Women | Men | Women | ||
| Cases/100,000 inhabitants | 68 | 33 | 77 | 26 | 52 | 21 | |
| Adjusted rate (SE) | |||||||
| - to world SP | 1.09 (0.15) | 0.53 (0.11) | 1.16 (0.14) | 0.32 (0.07) | 0.77 (0.12) | 0.24 (0.06) | |
| - to European SP | 1.69 (0.21) | 0.70 (0.13) | 1.84 (0.22) | 0.47 (0.10) | 1.23 (0.18) | 0.36 (0.09) | |
| % Accumulated risk (0-74) | 0.12 | 0.07 | 0.13 | 0.05 | 0.09 | 0.03 | |
| Adjusted truncated rate (35-64) | 0.13 | 0.14 | 0.07 | 0.00 | 0.03 | 0.00 | |
| Life years potentially lost | 117.50 | 95.00 | 160.00 | 19.00 | 104.00 | 15.50 | |
| Mean age of patients | 70.99 | 67.48 | 71.57 | 72.46 | 71.58 | 73.52 | |
| Range | 5-90 | 19-97 | 39-89 | 56-86 | 39-89 | 56-86 | |
| Standard deviation | 12.37 | 14.16 | 10.39 | 8.19 | 10.44 | 8.59 | |
| Male population (2017) | 3.115.522 | ||||||
| Female population (2017) | 3.391.662 | ||||||
SE: standard error; SP: standard population.
The mean age of patients was 70.78 years old (SD: 11.94; range: 5-97). In men (n=120), the mean age was 71.24 years old (SD: 11.58; range: 5-90) whereas in women (n=54) the mean age was 69.83 years old (SD: 12.65; range: 19-97).
From the INE data series, only 137 out of the 149 deaths from mesothelioma had C45 as the basic cause of death. In 12 deaths, the C45 code was found in the intermediate or fundamental causes, but not as a basic cause. The distribution of deaths for the different C45 codes sites was as follows: C45.0 (mesothelioma of pleura), 72 deaths (52.55%); C45.9 (meso-thelioma of unspecified site), 62 deaths (45.26%); C45.7 (mesothelioma of other specified sites), 9 deaths (6.57%); and C45.1 (mesothelioma of peritoneum), 6 deaths (4.38%). In 97.44% of the deaths with a known site, this was pleural.
A specific site of mesothelioma was not found in 71 deceased patients. The percentage of INE records with poor mesothelioma coding quality was 47.65%, while that for the MBDS series was 18.39%.
Table 1 shown prevalence rates in 2016 and 2017, and incidence rates of mesothelioma in the CM according to MBDS data in 2017.
Mortality rates (95%CI) of mesothelioma in the CM according to MBDS data in 2016 were showed in Table 2. From the INE microdata file, the mortality crude rate per 100.000 population, the adjusted rate (with SE) for world and European SP, accumulated risk (0-74), life years potentially lost, mean age at death, standard deviation (SD), and range of patients who died in 2016 was calculated. In 2016, the mortality rate in men adjusted to European Standard Population was 1.25 and the mortality rate in women was 0.32.
The comparison between the MBDS estimated mortality and the INE calculated mortality for 2016 is shown also in Table 2. Mortality rates for men between the two sources were very similar (MBDS and INE), but mortality rates for women were overestimated from MBDS data in relation the calculated rates from INE. In any case, the overestimation was not statistically significant (95%CI European adjusted rates MBDS: 0.34-0.82, and INE: 0.48-0.16).
Table 2. Mortality rates (95%CI) of mesothelioma in the Community of Madrid in 2016
| Mortality (95%CI) 2016 | |||||
|---|---|---|---|---|---|
| MBDS | INE | ||||
| Men | Women | Men | Women | ||
| Deaths/100,000 inhabitants | 43 | 28 | 56 | 19 | |
| Adjusted rate (SE) | |||||
| - to world SP | 0.70 (0.12) | 0.44 (0.10) | 0.74 (0.08) | 0.22 (0.03) | |
| - to European SP | 1.06 (0.17) | 0.58 (0.12) | 1.25 (0.17) | 0.32 (0.08) | |
| % Accumulated risk (0-74) | 0.08 | 0.05 | 0.08 | 0.07 | |
| Life years potentially lost | 61.50 | 88 | 76 | 54 | |
| Mean age of patients | 70.65 | 67.39 | 75.02 | 74.32 | |
| Range | 5-90 | 19-97 | 24-95 | 40-87 | |
| Standard deviation | 13.43 | 15.29 | 12.29 | 12.08 | |
| Male population (2017) | 3.115.522 | ||||
| Female population (2017) | 3.391.662 | ||||
MBDS: Minimum Basic Data Set; INE: Spanish National Statistics Institute; SE: standard error; SP: standard population.
The geographical pattern of risk of mesothelioma within the CM highlighted eight municipalities that registered at least five cases of mesothelioma in the MBDS. The Table 3 shows the crude rates of mesothelioma cases / 100,000 inhabitants according to the MBDS and of the number of cases and deaths / 100,000 inhabitants according to the INE, for the full period 2016 - 2017. These data are shown for each of the municipalities with more than 10,000 inhabitants and with at least five registered cases of mesothelioma in the MBDS.
Table 3. Rate of mesothelioma cases and deaths in 2016 - 2017 for eight municipalities of the Community of Madrid
| MBDS | INE | |
|---|---|---|
| Municipality | Cases / 100,000 inhabitants | Deaths / 100,000 inhabitants |
| Aranjuez | 5 (8.59) | 6 (10.31) |
| Getafe | 13 (7.29) | 16 (8.97) |
| Leganés | 9 (4.79) | 6 (3.20) |
| Madrid | 83 (2.61) | 66 (2.07) |
| Fuenlabrada | 7 (3.60) | 4 (2.05) |
| Coslada | 5 (6.02) | 1 (1.20) |
| Alcorcón | 6 (3.57) | 2 (1.19) |
| Alcalá de Henares | 5 (2.57) | 2 (1.03) |
MBDS: Minimum Basic Data Set; INE: Spanish National Statistics Institute.
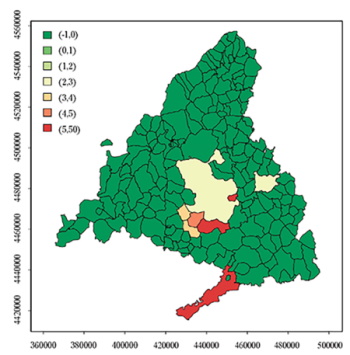
A scatter-plot was obtained from the crude rates of each of the eight locations (Figura 2); the Pearson correlation coefficient (r) was calculated to be 0.857. A risk map with the SMR according to the INE data was drawn up (Figura 3); municipalities with five or more detected cases are represented in colors other than green. Most mesothelioma cases showed an aggregation in municipalities located in the south, especially Getafe and Aranjuez.
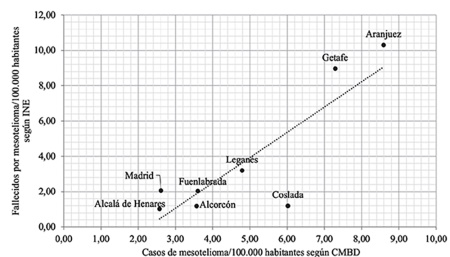
Figure 2. Municipalities of the Community of Madrid with five or more cases detected in the MBDS. Correlation between crude rates for cases and deaths per 100,000 inhabitants in the whole period (2016-2017). Pearson correlation coefficient (r) = 0.857.
Discussion
The lack of information on geographic areas not covered by PBCRs translates into an underreporting of cancer20. In Spain the CM is still one of these areas, and the MBDS could help to estimate the incidence of certain cancers, including mesothelioma. With the exception of incidence estimations from local series of cases14, no incidence of mesothelioma rates were known in Spain until now, with only mortality data being available13. Previous studies have validated the MBDS as a tool for epidemiological surveillance of cancer in general21 improving the information in PBCRs22. The MBDS has been used previously in the study of mesothelioma incidence in Italy but not in Spain, and thus, our study is a pioneering one in this regard. Italy has a National Registry of Mesotheliomas, and therefore the MBDS has been validated properly there18. In the CM, the only alternative to validating the MBDS as an instrument of epidemiological surveillance of mesothelioma is the SBDs recorded by the INE. The unit of the MBDS is the hospital discharge report, while the unit of the mortality registry of the INE is death. A patient can only die once but can be admitted to a hospital several times. It is therefore essential for a unique and anonymous patient identifier code number to be assigned during the processing of the MBDS data by the management unit. This process of anonymization is complex when large hospital discharge figures are used and is not exempt from errors.
The 2017 population censuses for the CM available on the INE website19 were used to calculate the adjusted rates. Following the direct standardization method23, calculations were carried out for two standard populations: Segi's (1960) world standard population and the European population. Segi's world population was chosen instead of the WHO (2000) world population23 because a large number of studies continue to use Segi's world population and because we could not compare our rates with rates published before 2000 if we had chosen the 2000 WHO standard world population. In turn, since the 2000 WHO standard population has an age structure that is intermediate between Segi's world and the European populations, having both rates allowed us to compare our rates to a certain extent with rates published exclusively with the 2000 WHO world standard population.
Rincón et al. studied mesothelioma deaths in the CM corresponding to the 1991-1999 period and calculated adjusted mortality rates (European standard population) of 0.97 for men and 0.3 for women24. These rates are significantly lower than those calculated in our series for 2016. The finding is in line with the predictions made in a 2013 study by López-Abente et al., according to which mesothelioma mortality rates in Spain would continue to increase at least until 2020 due to the birth cohort effect, given that the occupational exposure to asbestos was significantly lower only in men born after 196025. In the INE series of 149 deaths analyzed for the present study, 91.95% were born before 1960; therefore, it is expected that mortality will continue to increase due to the high weight that the birth cohort effect continues to have. These results reflect that exposure to asbestos has and will continue to have consequences in the coming years until the complete disappearance of the cohort of exposed individuals, at least until 2040 (in that year, those born in 1960 will be 80 years old). According to the study25, the prediction of the magnitude of mortality from mesothelioma in Spain is lower than that in other European countries. At the same time, another study warned of possible underdiagnosis of mesothelioma in Spain26. In our study, the percentage of the deceased (INE) lacking an specific site code for mesothelioma duplicate the percentage of the MBDS cases, which means that MBDS data can be of better quality than the SBDs of the INE. This would be one more reason to use the MBDS as an instrument for the epidemiological surveillance of mesothelioma.
The study of Rincón et al.24 on mortality from mesothelioma in the CM detected a large risk difference among municipalities and districts in the 1991-1999 period. The study identified a higher concentration of deaths in the more industrialized southeast zone24. In our study, we did not have access to postal codes, but the hospitals in southern Madrid in our MBDS series are those with the greatest activity related to the diagnosis and treatment of mesothelioma. Additionally, the largest aggregation of cases is concentrated in the municipalities of the southern zone. A study conducted by Odgerel et al. covers the period from 1994 to 2014, with data from more than 230 countries showing that the incidence of and mortality rates for mesothelioma have increased over time9. In Spain, López-Abente et al. studied the distribution of mortality from mesothelioma (pleural cancer) at the municipal level throughout the Spanish territory during the 1989-1998 period based on the basic cause of death in the SBDs27. The representation of the smoothed relative risk of mortality by mesothelioma on the map of Spain highlights the areas where there is greater exposure to asbestos. The preparation of maps based on municipal analysis appears to be a useful tool for the epidemiological surveillance of mesothelioma and a method for estimating asbestos exposure27. In this context, in our study Aranjuez was the municipality with the highest cases and deaths per 100.000 inhabitants along the 2016-2017 period and this could be related with factories using asbestos in the past placed in that area. A study by Crosignani et al. for the 1990-1998 period in Italy found that the MBDS not only aids in determining the cases of occupational cancer detected by PBCRs but also helps to identify the industries associated with such occupational exposure28.
Some limitations in our study should be mentioned. The change from CIE-9 and CIE-O-3 classification to CIE-10 could mean that codifying might not be sufficiently accurate as codifying personnel need to bear in mind the new codes for mesothelioma. Also, the number of cases of mesothelioma in municipalities under 10.000 habitants (only 5% of the total of municipalities) entail a loss of information about risk in those small geographic areas, so more studies should be conducted in order to resolve this limitation. Also, some strengths of our study are the good quality of structure and design of microdata provided by INE, from which we can also know the total amount of deaths in the period of our study. MBDS provides excellent and more complete data about diagnosis and particularly about location of mesothelioma, and this data can be easily exploded.
The CM does not have a PBCR, so the MBDS and SBDs of the INE are practically the only available sources. Validity of the combined use of the MBDS / INE as an epidemiological surveillance tool for mesothelioma is based on the homogeneity of data between the two sources. In this study, the first approximation was performed by comparing the mortality estimated by the MBDS and that calculated by the INE for 2016 (see Table 2). The second approximation was based on a high correlation between the crude rates of mesothelioma/100,000 inhabitants according to the MBDS and deaths from the INE, for the full period 2016 - 2017 in the eight municipalities with at least five cases of mesothelioma in the MBDS (see Table 3 and figure 2). The only study that used the MBDS for the detection of mesothelioma cases collected data from an Italian region that has a PBCR. In Italy, the MBDS has been successfully validated in the epidemiological surveillance of mesothelioma using the PBCR as a reference20. In the CM, this was not possible, but the similar mortality rates detected for 2016 with both sources (MBDS and INE) and the high correlation in the aggregation of cases and deaths obtained between the two sources (r = 0.86) suggest that in Spain, the MBDS is an important resource to exploit for epidemiological studies of mesothelioma risk.
In conclusion, mortality rates from mesothelioma in the CM in 2016 were higher than the ones previously registered. The geographical risk pattern continues to be focused on the more industrialized municipalities of the south. The aggregation of deaths in some of the municipalities in the southern zone was already known. The maximum aggregation of cases and deaths during 2016 and 2017 in Aranjuez (hitherto undetected municipality in the southern zone) is a new finding that requires further investigation. The MBDS is an effective instrument for monitoring cancer risk in general and mesothelioma in particular, providing good estimators of incidence and prevalence. The SBDs of the INE are also crucial for determining the mortality rates and increasingly incorporate more information of interest associated with death. Both sources, the MBDS and INE mortality, are complementary in the epidemiological surveillance of mesothelioma. The current use of the ICD-10 in both sources and the fact that their data are available with approximately one year of lag, should encourage future research in this field.