Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
International Microbiology
versión impresa ISSN 1139-6709
INT. MICROBIOL. vol.8 no.2 jun. 2005
| RESEARCH ARTICLE | |||
|
| |||
| Antibacterial activity of cerein 8A, a bacteriocin-like peptide produced by Bacillus cereus
Summary. The mode of action of cerein 8A, a bacteriocin produced by the soil bacterium Bacillus cereus 8A, was investigated. The effect of cerein 8A was tested against Listeria monocytogenes and a bactericidal effect at 400 arbitrary units (AU)/ml was observed. In addition, cerein 8A was bactericidal against Bacillus cereus at 200 AU/ml, and inhibited the growth of Escherichia coli and Salmonella Enteritidis. Stronger inhibition of these gram-negative bacteria was achieved when the chelating agent EDTA was added together with bacteriocin. The effect of cerein 8A on B. cereus and L. monocytogenes was also investigated by Fourier transform infrared spectroscopy (FTIR). Treated cells had an important frequency increase at 2920 cm-1 and a decrease at 1400 cm-1, corresponding to assignments of fatty acids. Transmission electron microscopy showed damaged cell walls and loss of protoplasmic material. These results suggest that the mode of action of cerein 8A is to interfere with cell membranes and the cell wall. [Int Microbiol 2005; 8(2):125-131] Key words: Bacillus cereus · Listeria monocytogenes · antimicrobial peptides · bacteriocins · infrared spectroscopy | ||
| |||
Actividad antibacteriana de la cereína 8A, un péptido de tipo bacteriocina producido por Bacillus cereus Resumen. Se investigó el modo de acción de la cereína 8A, una bacteriocina producida por la bacteria del suelo Bacillus cereus 8A. El efecto de la cereína 8A fue probado contra Listeria monocytogenes, obteniendo un efecto bactericida a concentraciones de 400 unidades arbitrarias (AU)/ml. La cereína 8A también tuvo un efecto bactericida contra Bacillus cereus a una concentración de 200 AU/ml. La bacteriocina inhibió el crecimiento de Escherichia coli y Salmonella Enteritidis. Mayor inhibición contra estas bacterias gram-negativas se consiguió cuando a la bacteriocina se le añadió el agente quelante EDTA. El efecto de la cereína 8A sobre B. cereus y L. monocytogenes también fue investigado por espectroscopía de infrarrojos de transformación de Fourier (FTIR). Las células tratadas mostraron un importante crecimiento en frecuencia de 2920 cm-1 y un decrecimiento de 1400 cm-1 de banda, correspondiéndose con la asignación de los ácidos grasos. La microscopía electrónica de transmisión mostró que las células habían padecido daños en la pared celular, con pérdida de material protoplásmico. Los resultados sugieren que el modo de acción de la cereína 8A se produce mediante su intervención en las membranas celulares y en la pared celular. [Int Microbiol 2005; 8(2):125-131] Palabras clave: Bacillus cereus · Listeria monocytogenes · péptidos antimicrobianos · bacteriocinas · espectroscopía de infrarrojos | Atividade antibacteriana da cereina 8A, um peptídeo tipo bacteriocina produzido por Bacillus cereus Resumo. O modo de ação da cereina 8A, uma bacteriocina produzida pela bactéria do solo Bacillus cereus 8A, foi investigado. O efeito da cereina 8A foi testado contra Listeria monocytogenes, resultando em um efeito bactericida com uma concentração de 400 unidades arbitrárias (UA)/ml. A cereina 8A também foi bactericida contra Bacillus cereus à 200 UA/ml. A bacteriocina inibiu o crescimento de Escherichia coli e Salmonella Enteritidis. Uma maior inibição contra estas bactérias gram-negativas foi alcançada quando o agente quelante EDTA foi adicionado junto com a bacteriocina. O efeito da cereina 8A sobre B. cereus e L. monocytogenes também foi investigado por espectroscopia de infravermelho com transformada de Fourier (FTIR). As células tratadas mostraram um importante aumento de frequência em 2920 cm-1 e uma diminuição na banda de 1400 cm-1, correspondendo a designações de ácidos graxos. A microscopia eletrônica de transmissão mostrou que as células apresentaram danos na parede celular com perda de protoplasma. Estes resultados sugerem que o modo de ação da cereina 8A é interferindo nas membranas celulares e parede celular. [Int Microbiol 2005; 8(2):125-131] Palavras chave: Bacillus cereus · Listeria monocytogenes · peptideo antimicrobiano · bacteriocinas · espectroscopia de infravermelho |
Introduction
Many bacterial species produce peptide antibiotics, called bacteriocins, that often have an antimicrobial effect on closely related organisms [17,26,35]. These compounds have been extensively studied because of their potential applications in the food industry as natural biopreservatives and in pharmaceuticals as antimicrobials [18,23]. Bacteriocins produced by lactic-acid bacteria have been the focus of many investigations because of their particular importance in the dairy industry [15,30]. Based on their chemical structures, stability, and mode of action, bacteriocins have been classified as: (i) lantibiotics; (ii) small heat-stable peptides; (iii) large heat-labile proteins; and (iv) complex proteins that require carbohydrate or lipid moieties for activity [17].
The mechanisms of action of peptide antibiotics are diverse, but the bacterial membrane is the target for most bacteriocins [17]. The type-A lantibiotics nisin, subtilin, Pep5, and epidermin have novel secondary modes of action in addition to their well-established lytic activity [34]. This dual antibiotic activity of a single molecule no doubt plays an important role in the antimicrobial mechanism of some bacteriocins [22]. Several bacteriocins or bacteriocin-like molecules have been described for Bacillus spp., including coagulin [14], cerein 7 [28], subtilosin [36], and thuricin 7 [8], and some of them have a broad spectrum of antibacterial activity. Bacillus cereus 8A, isolated from soils of native woodlands of southern Brazil, has antagonistic action against several pathogenic and food-spoilage microorganisms, such as Listeria monocytogenes, B. cereus, and pathogenic bacteria involved in bovine mastitis [4]. Treatment with proteases and trichloroacetic acid inhibits B. cereus 8A antimicrobial activity [4], suggesting that it produces a bacteriocin-like substance.
The aim of this work was to investigate the mode of action of cerein 8A and to explore its potential use as an antimicrobial to prevent the proliferation of L. monocytogenes and other pathogenic and food-spoilage microorganisms.
Materials and methods
Bacterial strains. Bacillus cereus 8A isolation and characterization have been described elsewhere [4]. The indicator strains Listeria monocytogenes ATCC 7644, Bacillus cereus ATCC 14579, Escherichia coli ATCC 25922 and Salmonella Enteritidis ATCC 13076 were stored in 20% glycerol at -21ºC, and propagated twice in BHI broth (Becton & Dickinson, Cockeysville, MD) before use.
Bacteriocin preparation and activity. B. cereus 8A was grown in 200 ml BHI broth at 30ºC in a shaker at 125 cycles/min for 32 h. Cells were harvested by centrifugation at 10,000 ×g for 15 min and the culture supernatant was filtered through a 0.22-µm membrane (Millipore, Bedford, MA). Bacteriocin activity was purified by precipitation with ammonium sulfate at 65% saturation. The pellet was resuspended in phosphate buffered saline (PBS) and extracted with 1-butanol (0.7 volumes). After butanol evaporation under reduced pressure, the bacteriocin was stored at 4ºC until its use for antimicrobial assays.
The antimicrobial activity of the purified bacteriocin was assessed by agar disc diffusion assay [25]. Aliquots (20 µl) of the bacteriocin preparation were applied onto 6-mm cellulose discs on agar plates previously inoculated with a 0.5 McFarland suspension of the indicator strain. Inhibition zones were measured after incubation at 37ºC for 24 h. The bacteriocin titer was calculated by the serial dilution method [25].
Growth determination. Viable cell counts, expressed as colony-forming units (cfu/ml), were determined as described previously [25]. Bacterial suspensions were diluted to 10-8 in 8.75 g NaCl/l; the samples were homogenized and then loaded (20 ml) in triplicate onto nutrient agar (Becton Dickinson) plates. The plates were incubated at 37ºC for 24 h, and 30-100 colonies were counted. In parallel, the optical density of the cultures at 600 nm (OD600)was measured.
Effect of cerein 8A against the indicator organisms. An overnight culture of each indicator organism was obtained by growing the respective bacteria in BHI at 37ºC. Samples (1 ml) of each culture were inoculated into Erlenmeyer flasks containing 99 ml BHI, which were then incubated at 37ºC. Growth was monitored at different intervals by determining the OD600 and by viable cell counts (cfu/ml). Bacteriocin, at a final concentration of 120 or 400 arbitrary units (AU)/ml, was added to cultures of L. monocytogenes and B. cereus, and its effects on turbidity and the number of viable cells were assessed at 2-h intervals. In addition, the effects of cerein 8A on gram-negative bacteria (E. coli and Salmonella Enteritidis) was evaluated at a concentration of 400 AU/ml. To evaluate the effect of a chelator on bacteriocin activity, 20 mM disodium EDTA (Riedel, Hannover) was added. Samples treated with EDTA alone served as controls.
Hemolysis and hemagglutination. The hemolytic and hemagglutination properties of the bacteriocin were verified using human and sheep erythrocytes. Tests were carried out essentially as described in [3]. Hemolysis was observed by visual inspection of sheep-blood agar plates. A clinical isolate of Aeromonas sp. with known hemolytic activity was used as positive control. For hemagglutination tests, 50 ml of the bacteriocin was mixed with the same volume of a 6% (v/v) washed erythrocyte suspension on a white porcelain tile. The negative control consisted of erythrocytes suspension and PBS. The reactions were visualized using a bright-field microscope, and considered positive if agglutination occurred within 10-15 min of incubation.
Hydrolytic activities. Murein hydrolytic activity was assayed on peptidoglycan agar plates [2]. Commercial lysozyme (Merck, Darmstadt, Germany) was used as positive control. To check whether the compound had phospholipase activity, the bacteriocin was spotted onto egg-yolk plates [13], using commercial phospholipase (Sigma, St. Louis, MO) as the control. Proteolytic activity was investigated using either skimmed-milk agar plates or the substrate azocasein [32], with pronase E (Sigma) as positive control. Bacteriocin dilutions were spotted onto the plates and the appearance of clear zones was monitored.
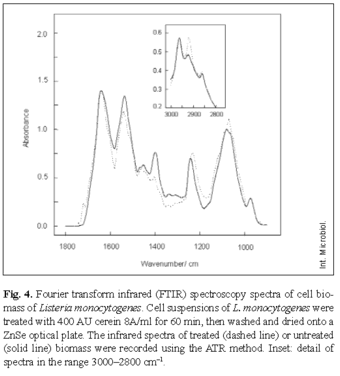
Fourier transform infrared (FTIR) spectroscopy. Bacteriocin was added (400 AU/ml) to cell suspensions of about 106 cfu/ml of either L. monocytogenes or B. cereus. After incubation for 60 min, both treated and control cells were washed three times with MilliQ water. Twenty microliters of each bacterial sample was evenly applied onto a ZnSe optical plate, dried for approximately 15 min under vacuum, and then analyzed by FTIR spectroscopy. The curves represent the average of three individual measurements of the same experiment. All IR spectra (4000-650 cm-1) were obtained by the attenuated total reflection (ATR) technique, using a Perkin-Elmer Spectrum One spectrometer (Perkin Elmer, Uberlingen, Germany) with a horizontal ATR device (Se, 45º). Fifty scans were taken with 4 cm-1 resolution. Reproducibility of the normalized spectra was ± 2%.
Transmission electron microscopy. Samples were taken from exponentially growing cultures (108 cfu/ml) of either L. monocytogenes or B. cereus treated or not with cerein 8A (320 AU/ml). Cells were harvested by centrifugation and washed twice with 0.1 M phosphate buffer (pH 7.3). The cells were fixed with 2.5% (v/v) glutaraldehyde, 2.0% (v/v) formaldehyde in 0.12 M phosphate buffer for 10 days and then postfixed in 2% (w/v) osmium tetroxide in the same buffer for 45 min. The samples were dehydrated in a graded acetone series (30-100%) and embedding in Araldite-Durcupan for 72 h at 60ºC. Thin sections (microtome UPC-20, Leica) were mounted on grids, covered with collodion film, and poststained with 2% uranyl acetate in Reynold's lead citrate. All preparations were observed with a JEOL JEM 1200ExII electron microscope (JEOL, Tokyo) operating at 120 kV.
Results
Effect on Listeria monocytogenes and Bacillus cereus. Figure 1 shows the effect of bacteriocin concentration on the survival of L. monocytogenes and B. cereus. The number of viable cells of both bacteria decreased as the bacteriocin concentration increased. Complete inhibition of the growth of L. monocytogenes was observed with 400 AU bacteriocin/ml, corresponding to an EC50 ("effective concentration", defined as the drug concentration producing 50% maximum effect) of approximately 120 AU/ml. In the case of B. cereus, growth was completely inhibited at a dose of 200 AU cerein 8A/ml, and the EC50 was approximately 40 AU/ml.
The kinetics of the bacteriocin effect on the growth of L. monocytogenes and B. cereus are shown in Fig. 2. Bacteriocin (120 AU/ml final concentration) added to a cell suspension of L. monocytogenes resulted in a difference in viable counts corresponding to 3 exponential-growth cycles compared to the control (Fig. 2A). The inhibition of L. monocytogenes growth was immediate and resulted in a decrease in OD600 during the incubation (Fig. 2A), suggesting that cell lysis is triggered in L. monocytogenes after cell death. At a concentration of 400 AU/ml, viable counts decreased drastically, reaching zero in 75 min (Fig. 2B).
The addition of bacteriocin (120 AU/ml final concentration) to a cell suspension of B. cereus caused a large decrease in the number of viable cells over a period of 10 h (Fig 2C). The OD600 of bacteriocin-treated B. cereus suspensions remained nearly constant during this period (Fig. 2C). At a higher dose of cerein 8A (400 AU/ ml), there was a rapid decrease in viable cell counts, which reached zero within 60 min, in parallel with a decrease in the OD600 (Fig. 2D).
Effect on gram-negative bacteria. Similarly, when a cell suspension of Salmonella Enteritidis was treated with 400 AU bacteriocin/ml (final concentration) the viable counts were much lower than those of the controls without bacteriocin (Fig. 3A). The addition of bacteriocin plus EDTA resulted in greater inhibition than obtained with bacteriocin alone. Similar results were obtained with E. coli (Fig. 3B).
Hemolysis, hemagglutination, and hydrolytic activities. The hemolytic and hemagglutination activities of cerein 8A were assayed and were negative against human and sheep erythrocytes. Negative reactions for muramidase, phospholipase, and protease were also observed (data not shown).
Infrared spectroscopy. The effect of cerein 8A on macromolecular structures of L. monocytogenes and B. cereus was analyzed by FTIR spectroscopy. Table 1 shows the possible assignments of the individual bands.
Treated cells of L. monocytogenes showed an important frequency increase at 2920 cm-1 and a decrease at 1400 cm-1 (Fig. 4). A displacement was observed at 1230 cm-1 (phospholipids). Smaller differences were observed in the range 1000-1100 cm-1 (deformation of carbohydrates). FTIR spectra of the cell biomass of B. cereus treated with cerein 8A showed very similar changes.
Electron microscopy. L. monocytogenes and B.cereus harvested from an early-stationary-phase culture (108 cfu/ml) were incubated with 320 AU cerein 8A/ml for 60 min. After incubation, the microorganisms were separated and prepared for transmission electron microscopy. Both L. monocytogenes and B. cereus cells showed vesiculization of the protoplasm, pore formation, and complete disintegration of the cells (Fig. 5A-C).
Discussion
Cerein 8A, an antimicrobial peptide produced by B. cereus 8A showed inhibitory activity against major food pathogens and spoilage bacteria [4]. Our results suggest that this substance has a bactericidal effect against L. monocytogenes and B. cereus, based on the observed decline in the number of living cells of either B. cereus or L. monocytogenes within 2 h after the addition of cerein 8A. A bactericidal effect for this compound is also supported by the decrease in OD600, which indicated that the cells of these indicator strains had lysed. However, the effect may depend on the specific assay conditions, such as the amount and purity of the bacteriocin, the indicator strain, and its cellular concentration [11]. Indeed, Boucabeille et al. [5] found linenscin OC2 to be bactericidal and bacteriolytic towards L. innocua, while at a low dose of linenscin OC2 bacteriostasis was observed. Similar results were reported for a bacteriocin produced by Brevibacterium linens ATCC 9175 [25].
Cerein 8A displayed similar kinetics against B. cereus and L. monocytogenes, killing the bacterial cells within 60 min. This bacteriocin, which inhibits several gram-positive bacteria, seems to be most active against Bacillus spp. [4]. Indeed, a higher dose was needed to kill L. monocytogenes than to kill B. cereus. This result agrees with reports that several bacteriocins are mostly active against closely related species [17,35]. Although bacteriocin cerein 8A appears not to kill E. coli and Salmonella Enteritidis cells, it greatly inhibited their growth. This inhibitory effect increased when the chelating agent EDTA was added, and was similar to the inhibitory effect reported for nisin [9]. In both cases, inhibition is most likely due to weakening of the outer plasma membrane following sequestration of magnesium ions by the chelating agents [24]. Antimicrobial activity has been associated with molecules frequently exported by bacteria, such as hemolysins or hydrolytic enzymes [19,31]. Some bacteriocins produced by bacteria isolated from food, such as linescin OC2, have hemolytic activity [5]. However, these activities are not associated with the antimicrobial activity.
FTIR spectroscopy of B. cereus and L. monocytogenes treated with cerein 8A revealed major changes in the assignments for symmetric C=O bonds (1400 cm-1), C-H antisymmetric stretching of CH2 bonds (2920 cm-1) and for P=O antisymmetric bonds, in the region corresponding to membrane fatty acids [12,16,33]. By affecting the bacterial membrane, cerein 8A may cause dissipation of the proton-motive force as well as leakage of intracellular contents. FTIR has been used to identify microrganisms [20,21], but few attempts have been made to use this technique to investigate antimicrobial mechanisms. The interaction of surfactin [7] and gramicidin S [1] with membrane lipids has been demonstrated by FITR studies. Since cerein 8A does not possess phospholipase activity, it could instead be a pore-forming peptide. This mechanism is supported by electron microscopy, which showed cell lysis after treatment with cerein 8A. Cell damage caused by cerein 8A resembles that observed with a crude bacteriocin produced by Lactobacillus salivarius [27]. Type-A lantibiotics, to which nisin, pediocin, and epidermin, and many other amphiphilic antimicrobial peptides belong, exert their activity by disrupting the functional barrier of microbial cytoplasmic membranes [10,22]. In addition to nisin and epidermin, lipid I and II binding has been observed for subtilin and mesarcidin produced by Bacillus spp. [6,22]. The hydrophobic bacteriocin cerein 7, produced by B. cereus Bc7, has also been characterized as a membrane-active compound [29].
In conclusion, our results suggest that cerein 8A has a bactericidal effect apparently by disrupting the membrane function of target organisms.
Acknowledgements. The authors thank Moema Queiroz from the Centro de Microscopia Eletronica (CME, UFRGS) for her technical assistance. This work was supported by CNPq (Brazil).
References
1. Akinoglu BG, Gheith M, Severcan F (2001) Thermodynamics study of gramicidin S and dipalmitoylphosphatidylcholine model membrane interactions based on the FITR spectroscopy. J Mol Struct 565-566:281-285 [ Links ]
2. Beukes M, Bierbaum G, Sahl HG, Hastings JW (2000) Purification and partial characterization of a murein hydrolase, millericin B, produced by Streptococcus milleri NMSCC 061. Appl Environ Microbiol 66:23-28 [ Links ]
3. Bizani D, Brandelli A (2001) Antimicrobial susceptibility, hemolysis, and hemagglutination among Aeromonas spp. isolated from water of a bovine abattoir. Braz J Microbiol 32:334-339 [ Links ]
4. Bizani D, Brandelli A (2002) Characterization of a bacteriocin produced by a newly isolated Bacillus sp. strain 8A. J Appl Microbiol 93:512-519 [ Links ]
5. Boucabeille C, Mengin-Lecreulx D, Henkes G, Simonet JM, van Heijenoort J (1997) Antibacterial and hemolytic activities of linescin OC2, a hydrophobic substance produced by Brevibacterium linens. FEMS Microbiol Lett 153:295-301 [ Links ]
6. Brötz H, Bierbaum G, Leopold K, Reynolds PE, Sahl HG (1998) The lantibiotic mesarcidin inhibits peptidoglycan synthesis by targeting lipid II. Antimicrob Agents Chemother 42:154-160 [ Links ]
7. Carrillo C, Teruel JA, Aranda FJ, Ortiz A (2003) Molecular mechanism of membrane permeabilization by the peptide antibiotic surfactin. Biochim Biophys Acta 1611:91-97 [ Links ]
8. Cherif A, Ouzari H, Daffonchio D, Cherif H, Ben Slama K, Hassen A, Jaoua S, Boudabous A (2001) Thuricin 7: a novel bacteriocin produced by Bacillus thuringiensis BMG1.7, a new strain isolated form soil. Lett Appl Microbiol 32:243-247 [ Links ]
9. Cutter CN, Siragusa GR (1995) Population reductions of gram-negative pathogens following treatments with nisin and chelators under various conditions. J Food Prot 58:977-983 [ Links ]
10. Dalmau M, Maier E, Mulet N, Viñas M, Benz R (2002) Bacterial membrane injuries induced by lactacin F and nisin. Int Microbiol 5:73-80 [ Links ]
11. De Vuyst L, Vandamme EJ (1994) Bacteriocins of lactic acid bacteria. Blackie Academic & Professional, London [ Links ]
12. Filip Z, Hermann S (2001) An attempt to differentiate Pseudomonas spp. and other soil bacteria by FT-IR spectroscopy. Eur J Soil Biol 37:137-143 [ Links ]
13. Habermann E, Hardt KL (1972) A sensitive and specific plate test for the quantitation of phospholipases. Anal Biochem 50:163-170 [ Links ]
14. Hyronimus B, Le Marrec C, Urdaci MC (1998) Coagulin, a bacteriocin-like inhibitory substance produced by Bacillus coagulans I4. J Appl Microbiol 85:42-50 [ Links ]
15. Jack RW, Tagg JR, Ray B (1995) Bacteriocins of gram-positive bacteria. FEMS Microbiol Rev 59:171-200 [ Links ]
16. Kamnev AA, Antonyuk LP, Matora LY, Serebrennikova OB, Sumaroka MV, Colina M, Renou-Gonnord MF, Ignatov VV (1999) Spectroscopic characterization of cell membranes and their constituents of the plant-associated soil bacterium Azospirillum brasilense. J Mol Struct 480-481:387-393 [ Links ]
17. Klaenhammer T (1993) Genetics of bacteriocins produce by lactic acid bacteria. FEMS Microbiol Rev 12:39-85 [ Links ]
18. Kolter R, Moreno F (1992) Genetics of ribosomally synthesized peptide antibiotics. Annu Rev Microbiol 46:141-163 [ Links ]
19. Lai ACY, Tran S, Simmonds SR (2002) Functional characterization of domains found within a lytic enzyme produced by Streptococcus equi subsp. zooepidemicus. FEMS Microbiol Lett 215:133-138 [ Links ]
20. Maquelin K, Kirschner C, Choo-Smith LP, van den Braak N, Endtz HP, Naumann D, Puppels GJ (2002) Identification of medically relevant microorganisms by vibrational spectroscopy. J Microbiol Methods 51:255-271 [ Links ]
21. Mariey L, Signolle JP, Amiel C, Travert J (2001) Discrimination, classification, identification of microorganisms using FTIR spectroscopy and chemometrics. Vib Spectrosc 26:151-159 [ Links ]
22. McCafferty DG, Cudic P, Yu MK, Behenna DC, Kruger R (1999) Synergy and duality in peptide antibiotic mechanisms. Curr Op Chem Biol 3:672-680 [ Links ]
23. Milles H, Lesser W, Sears P (1992) The economic implication of bioengineering mastitis control. J Dairy Sci 75:596-605 [ Links ]
24. Montville TJ, Bruno MEC (1994) Evidence that dissipation of proton motive force is a common mechanism of action for bacteriocins and other antimicrobial proteins. Int J Food Microbiol 24:53-74 [ Links ]
25. Motta AS, Brandelli A (2002) Characterization of an antibacterial peptide produced by Brevibacterium linens. J Appl Microbiol 92:63-70 [ Links ]
26. Naclerio G, Ricca E, Sacco M, De Felice M (1993) Antimicrobial activity of a newly identified bacteriocin of Bacillus cereus. Appl Environ Microbiol 59:4313-4316 [ Links ]
27. Ocaña VS, Holgado AAPR, Nader-Macías ME (1999) Characterization of a bacteriocin-like substance produced by a vaginal Lactobacillus salivarius strain. Appl Environ Microbiol 65:5631-5635 [ Links ]
28. Oscáriz JC, Lasa I, Pisabarro AG (1999) Detection and characterization of cerein 7, a new bacteriocin produced by Bacillus cereus with a broad spectrum of activity. FEMS Microbiol Lett 178:337-341 [ Links ]
29. Oscáriz JC, Pisabarro AG (2000) Characterization and mechanism of action of cerein 7, a bacteriocin produced by Bacillus cereus Bc7. J Appl Microbiol 89:361-369 [ Links ]
30. O'Sullivan L, Ross RP, Hill C (2002) Potential of bacteriocin-producing lactic acid bacteria for improvements in food safety and quality. Biochimie 84:593-604 [ Links ]
31. Parret AHA, De Mot R (2002) Bacteria killing their own kind: novel bacteriocins of Pseudomonas and other g-proteobacteria. Trends Microbiol 10:107-112 [ Links ]
32. Riffel A, Lucas F, Heeb P, Brandelli A (2003) Characterization of a new keratinolytic bacterium that completely degrades native feather keratin. Arch Microbiol 179: 258-265 [ Links ]
33. Rodriguez MPQ (2000) Fourier transform infrared (FITR) technology for the identification of organisms. Clin Microbiol Newsl 22:57-61 [ Links ]
34. Sahl HG, Bierbaum G (1998) Lantibiotics: biosynthesis and biological activities of uniquely modified peptides from Gram-positive bacteria. Annu Rev Microbiol 52:41-79 [ Links ]
35. Tagg JR, Dajani AS, Wannamaker LK (1976) Bacteriocins of gram-positive bacteria. Bacteriol Rev 40:722-756 [ Links ]
36. Zheng G, Slavik MF (1999) Isolation, partial purification and characterization of a bacteriocin produced by a newly isolated Bacillus subtilis strain. Lett Appl Microbiol 28:363-367 [ Links ]












 Curriculum ScienTI
Curriculum ScienTI