My SciELO
Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Enfermería Global
On-line version ISSN 1695-6141
Enferm. glob. vol.18 n.53 Murcia Jan. 2019 Epub Oct 14, 2019
https://dx.doi.org/10.6018/eglobal.18.1.294561
Articles
Quality of life and self-care in patients with Parkinson in a regional hospital: descriptive study
1Nurse, Vinaroz District Hospital, Vinaroz, Castellón de la Plana. Spain.
2Department of Nursing, Jaume I University, Castellón de la Plana. Spain.
3Department of Nursing, University of Alicante, Investén-isciii. CIBER of Frailty and Healthy Ageing, Alicante. Spain.
Objectives:
To determine the level of self-care and quality of life in a sample of patients in the early stages of Parkinson’s disease and identify the most highly affected dimensions of self-care and quality of life. To study the relationship between non-motor symptoms and these outcome measures.
Method:
A cross-sectional study was conducted in a sample of 21 patients with stage I or II Parkinson’s disease as defined by the Hoenh Yarh Scale. We studied the level of self-care, quality of life, non-motor symptoms and perceived health status. Descriptive, inferential and correlation analyses were performed using SPSS v.20.
Results:
Mean sample age was 71.86 (SD(8.93) years old, and 52.4% were males. The mean score for self-care was 1.14 (SD(0.35), and the most highly affected dimension was medication use, with 61.9% of the sample presenting complete autonomy, while for quality of life it was 17.28 (SD(7.75), and the most highly affected dimension was bodily discomfort (m=35.03; SD(19.61). We observed a correlation between non-motor symptoms and quality of life (r=0.246; p=0.022), but not between non-motor symptoms and self-care (r=0.010; p=0.662).
Conclusions:
Our sample presented an accepTable level of self-care and quality of life. The most highly affected quality of life dimensions were bodily discomfort followed by cognitive impairment, and non-motor symptoms were related to the level of quality of life. It is important to consider these aspects when designing therapeutic education interventions targeting patients with Parkinson’s disease.
Key words: Parkinson’s disease; quality of lif; self-care; non-motor symptoms
INTRODUCTION
Parkinson’s disease (PD) is the second only to Alzheimer’s as the most prevalent neurodegenerative disorder in Spain1, affecting between 87,833 and 396,976 people2. Furthermore, prevalence is related to population ageing and management of the disorder is complex, rendering PD a major health and social problem3. Onset usually occurs between 55 and 60 years of age, with a higher incidence among men. Although the average life expectancy of a patient with PD is generally the same as for the general population, complications may arise in the latter stages of the disease that can lead to death4.
The cardinal symptoms of PD are tremor, rigidity, slowness of movement and postural instability. For many years, PD was only thought to affect the motor system. However, it has since been found to affect other neurons over time, giving rise to non-motor symptoms that disrupt and damage the autonomic, limbic and somatosensory systems and cause severe alterations in behaviour and mood. Despite advances in neurology, the aetiology of PD remains unclear; nonetheless, genetic and environmental factors, such as early exposure to agrochemicals, are now known to play a role in its pathophysiology5.
Studies have demonstrated that patients with PD present impaired quality of life compared with the general population, as a result of motor and non-motor symptoms that affect their ability to function and contribute to their deterioration6. Some authors have emphasised the prevalence of non-motor symptoms, their rate of underdiagnosis and their impact on quality of life, since these are the main cause of morbidity and the primary reason for institutionalisation and hospitalisation7, entailing high health costs.
More than 60% of patients with PD are in the early stages8. Consequently, therapeutic education interventions with these patients may help prevent or delay worsening of symptoms and improve their quality of life through self-care. Further research is required to determine the areas of quality of life that are most affected in the early stages of PD and to measure these patients’ needs and perceptions of their own well-being, in order to design therapeutic education interventions that target their self-care needs. Promoting self-care will enable these patients to exercise greater control over their health and microenvironment and achieve better overall well-being and a higher quality of life9.
The primary aim of this study was to determine the level of self-care and quality of life in a sample of patients in the early stages of Parkinson’s disease and identify the most highly affected dimensions of self-care and quality of life. A further goal was to study the relationship between non-motor symptoms and self-care and quality of life.
METHOD
Design and setting
We conducted a cross-sectional study between July 2014 and June 2015 to determine the quality of life and self-care in patients with Parkinson’s disease attending the neurology service of a district hospital (HUCV) in the province of Castellón. We used the STROBE guide to design and conduct the study10.
Population and sample
The target population consisted of patients diagnosed with PD (ICD-9-332) and attending the HUCV neurology service (N=162). Using purposive sampling, we included patients aged 50-85 years old in the early stages of PD (stages I or II on the Hoehn Yahr Scale11), who obtained a score greater than 60 on the Barthel index12and greater than or equal to 70% on the Schwab & England Activities of Daily Living Scale13. We excluded subjects who did not want to participate and those who presented intellectual disability (Pfeiffer Scale)14.
In line with the results of the GRANMO programme, a sample of 27 subjects was considered sufficient, establishing a 95% confidence interval, an accuracy of 20%, a percentage of the population of patients in the early stages of PD of 50% and a 20% replacement rate.
Variables and instruments
Sociodemographic variables included age, sex, educational level (no formal education, basic education, post-compulsory secondary education, university education), marital status (married, single, separated/divorced, widowed) and occupational status (active, unemployed, retired).
Self-care and quality of life variables
- Self-care was measured using theEscala de Valoración de la Autonomía para el Autocuidado(Self-Care Autonomy Scale, Spanish initials: EVAA). This rates autonomy on a scale of 1 to 10 where the higher the score, the lower the degree of autonomy15.
- Quality of life was measured using the Parkinson’s Disease Questionnaire (PDQ-39). This yields a score between 0 and 100 where the higher the score, the better the individual’s perceived quality of life16.
- Non-motor symptoms were measured using the Non-Motor Symptoms Scale (NMSS) for Parkinson’s disease. This yields a score between 0 and 360 where a value close to 0 indicates fewer non-motor symptoms17.
- Perceived health status was measured using the EuroQoL-5D (EQ-5D) instrument, where scores range between 1 (better health status) and 0 (death)18.
Data collection
Subjects were recruited and data collected between January and March 2015. Patients with PD were identified from the neurology service database, and medical records were consulted to select subjects who met the inclusion criteria.
Data were collected during patients’ appointments at the neurology service. Patients who did not have an appointment already scheduled during the data collection period were contacted by telephone. Patients received a telephone call to remind them of their appointment.
Two nurses collected data during interviews using anad hocdata collection spreadsheet. A pilot study was conducted with 5 subjects to confirm that the procedure functioned correctly. Average interview duration was 30 minutes.
Ethical considerations
This study was approved by the HUCV and the Ethics Committee at Jaume I University, and observed the ethical principles of the Declaration of Helsinki. All subjects received prior information and were asked to give their informed consent for data processing. Personal data were anonymised by assigning a specific code, and confidentiality was maintained in line with Articles 11.1.e) and 11.6 of Organic Law 15/1999 on the Protection of Personal Data.
Statistical analysis
A descriptive analysis was performed of all variables according to their nature. A comparison of means or medians was performed using the Mann Whitney U-test and the Kruskal-Wallis test. The relationship between mean scores for non-motor symptoms and self-care and quality of life was analysed using Pearson’s correlation coefficient. The level of statistical significance was p=0.05. Statistical analysis was performed using SPSS v.20.
RESULTS
A total of 27 subjects met the inclusion criteria. One was excluded as it was not possible to establish contact, four refused to participate and one was lost during the study, leaving a final sample of 21 subjects.Table 1gives a summary of the sociodemographic variables.
The EVAA scale yielded a mean score for self-care of 1.14 (SD(0.35), with 85.7% (n=18) of the sample obtaining a functional autonomy level of 1 and 2 (greater degree of autonomy). The most highly affected dimension was medication use, whereby 61.9% (n=13) showed complete autonomy, 23.8% (n=5) needed stimulus/supervision, 9.5% (n=2) required help and 4.8% (n=1) presented total dependence. For mobility, 76.3% (n=16) presented complete autonomy, 14.3% (n=3) needed stimulus/supervision and 9.5% (n=2) required help. We found significant differences between educational level and the items nutrition-food (p=0.008), nutrition-hydration (p=0.001) and grooming-hygiene (p=0.008) (Table 2).
Table 2 Description of EVAA and its items and relationship with sociodemographic variables

EVAA: Escala de Valoración de la Autonomía para el Autocuidado (Self-Care Autonomy Scale)
The mean score for quality of life measured using the PDQ-39 scale was 17.28 (SD(7.75), and the most highly affected dimensions were bodily discomfort (m=35.03; SD(19.61) followed by cognitive impairment (m=26.78; SD(18.75). No significant differences were detected between the sociodemographic variables and mean scores for quality of life (Tables 3 and4 ).
Table 3. Description of PDQ-39 and its dimensions and relationship with sociodemographic variables

m: mean; SD: standard deviation; PDQ-39: Parkinson’s Disease Questionnaire; ADL: activities of daily living
Table 3 (continuation). Description of PDQ-39 and its dimensions and relationship with sociodemographic variables

m: mean; SD: standard deviation; PDQ-39: Parkinson’s Disease Questionnaire; ADL: activities of daily living
Table 4. Description of PDQ-39 and its dimensions and relationship with sociodemographic variables

m: mean; SD: standard deviation; PDQ-39: Parkinson’s Disease Questionnaire
Table 4 (continuation). Description of PDQ-39 and its dimensions and relationship with sociodemographic variables
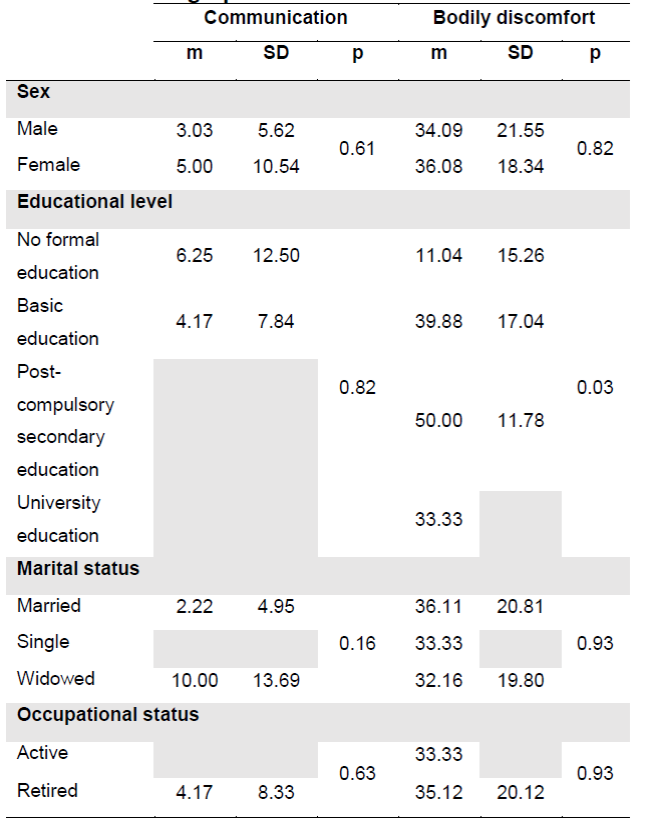
m: mean; SD: standard deviation; PDQ-39: Parkinson’s Disease Questionnaire
The mean score for perceived health status, measured using the EQ-5D scale, was 0.67 (SD(0.13) and the most highly affected dimensions were anxiety/depression, with a mean of 1.90 (SD(0.53), followed by mobility with a mean of 1.48 (SD(0.51). The EQ-5D index presented a mean of 75.48 (SD(15.24). No significant differences were detected between the sociodemographic variables and perceived health status (p<0.05) (Table 5).
Table 5. Description of EQ-5D and its dimensions and relationship with sociodemographic variables

m: mean; SD: standard deviation; EQ-5D: EuroQoL-5D Health Questionnaire
The mean score for non-motor symptoms, assessed using the NMSS scale, was 41.33 (SD(20.58). The domain of urinary function showed the highest mean, at 7.95 (SD(6.67), while the cardiovascular system domain presented the lowest mean, at 0.95 (SD(1.20). The correlation between non-motor symptoms and quality of life was low (r=0.246; p=0.022), while that between non-motor symptoms and self-care was negligible (r=0.010; p=0.662).
DISCUSSION
Spain is posed with the challenge of chronicity. More than 20% of the population rendered dependent by chronic disorders are affected by neurological diseases. Health care for the chronically ill must change to focus on preventing the complications that arise from these disorders, adopting a holistic, integrated and multidisciplinary approach19. Parkinson’s disease is a chronic, progressive disorder associated with a high burden of disability that escalates health care needs20. One important factor in the quality of life (QoL) of patients with Parkinson’s disease is their stage on the Hoenh Yarh Scale, since the step from stage II to III entails a significant decline in QoL and an increase in non-motor symptoms. This change affects patients with PD more than age itself21. Consequently, the design and implementation of educational interventions in the early stages, based on the most severely affected areas of quality of life and self-car, can help improve patient autonomy and QoL.
Quality of life and the capacity for self-care were close to good in our sample. The most highly affected dimensions of self-care in our study were medication use and mobility, whereas other studies have found that patients with PD require more help to carry out activities of daily living19. In addition, other studies have obtained a higher quality of life22; however, these included patients in all stages of PD, whereas we only included patients in the early stages, and they also analysed much larger samples using different scales.
The results of our study indicate that functional status in the initial stages of PD affects certain QoL dimensions through a self-care deficit, coinciding with Jones23, who suggested that the areas with greatest impact on QoL as a consequence of loss of functionality are those related to physical function, social support and cognitive status. In our study, the least affected QoL dimension was communication, while the most affected was cognitive status. It should be noted that we did not consider concomitant diseases such as arthritis, diabetes or circulatory problems in our study, although these exert a negative effect on the cognitive and functional status of patients with PD24.
No significant differences were observed in the level of QoL depending on the sociodemographic variables studied. Some authors25have suggested that a lower educational level may benefit QoL in subjects with PD due to greater ease in accepting the disease. However, these results do not coincide with our findings. Furthermore, the severity of PD may have a greater impact on QoL in terms of physical function, mobility and activities of daily living, but psychological adjustment to the disease also exerts a direct influence on QoL.
Our results for non-motor symptoms do not coincide with those reported by other authors20, since the correlation with quality of life was low and was not significant with self-care. It is probable that these results were due to our small sample size. We also observed a higher prevalence of urinary symptoms in our sample, whereas the literature indicates that sleep disorders/fatigue and mood/cognitive impairment are the most frequent symptoms23)(26; in our study, these occupied second and third place. Yang27has calculated that depression affects one in three people with PD, and has claimed that this is exacerbated by the presence of non-motor symptoms, which are very prevalent in the early stages of PD28and exert a major negative influence on QoL.
Interventions with chronically ill patients should be aimed at enabling them to become active patients who are aware of their illness and adopt a more proactive role. The overall objective of therapeutic education is to help users overcome a self-care deficit so that they can better respond to the demands of self-care entailed in the process of managing their chronic disease and thus enhance their quality of life. The capacity for self-care is promoted by actions that enable patients to recognise, treat and manage their own health problems autonomously, in the knowledge that the health care system is close at hand. It is necessary to implement various actions that promote the concept of self-care and foster the acquisition of this capacity among the population in general and patients in particular. People must be helped to manage their treatment plan (drugs, diet, etc.), to lead the most autonomous and satisfactory life as possible and to learn how to manage the emotional effects of their disease29. This would increase patient satisfaction, enhance daily QoL and improve disease management, besides reducing resource consumption (visits, emergencies, hospitalisation) due to decompensation or complications. To achieve this, it is essential to determine educational needs and identify those aspects of the disease that require most attention.
Our results should be interpreted with caution since the study focused on a specific service and analysed a small sample that was not recruited through random sampling. Consequently, it is difficult to generalise the results. Nevertheless, we did attain the number of subjects necessary according to the sample size calculation performed. It should also be noted that we focused on patients in the early stages of PD, since these are more amenable to educational interventions. In addition, this was a descriptive study and thus did not allow for the verification of hypotheses. Despite these limitations, our findings are useful because they facilitate the design of educational interventions aimed at improving self-care and quality of life based on the needs of this group of patients.
CONCLUSIONS
Our sample presented an accepTable level of self-care and QoL. The main self-care dimensions affected were medication use and mobility. Meanwhile, the most highly affected QoL dimensions were bodily discomfort followed by cognitive impairment. The correlation between non-motor symptoms and quality of life was low but significant, whereas we found no correlation between non-motor symptoms and level of self-care. An analysis of the dimensions affected in self-care, QoL and non-motor symptoms is important when designing educational interventions in patients with PD.
REFERENCIAS
1. Rodríguez Pupo Jorge Michel, Díaz Rojas Yuna Viviana, Rojas Rodríguez Yesenia, Ricardo Rodríguez Yuniel, Aguilera Rodríguez Raúl. Actualización en enfermedad de Parkinson idiopática. CCM. 2013 Jun; 17( 2 ): 163-177. [ Links ]
2. Grupo Andaluz de Trastornos del Movimiento. Recomendaciones de Práctica Clínica en la Enfermedad de Parkinson. Sociedad Andaluza de Parkinson. Sociedad Andaluza de Neurología. 2012. [ Links ]
3. García Ramos R, López Valdés E, Ballesteros L, De Jesús S, Mir P. Informe de la fundación del cerebro sobre el impacto social de la enfermedad de Parkinson en España. [monografía en internet]. Madrid: 2013 [acceso 12 de febrero de 2015]. Disponible en: http://www.fundacióndelcerebro.es/docs/informe_parkinson.pdf [ Links ]
4. Peñas Domingo E. El Libro Blanco del Párkinson en España. Aproximación, análisis y propuesta de futuro. Editor. Patronato sobre Discapacidad. Ministerio de Sanidad, Servicios Sociales e Igualdad. 2015. [ Links ]
5. Massano J, Bhatia KP. Enfoque clínico de la enfermedad de Parkinson: Características, diagnóstico y Principios de la gestión. Cold Spring Harbor Perspectivas en Medicina. 2012; 2(6). [ Links ]
6. Martínez Jurado E, Cervantes Arriaga A, Rodriguez Violante M. Calidad de vida en pacientes con enfermedad de Parkinson. Revista Mexicana de Neurociencia. 2010; 11(6): 480-486. [ Links ]
7. Argandoña-Palacios L, Perona-Moratalla AB, Hernández Fernández F, Díaz-Maroto I, García-Muñozguren S. Trastornos no motores de la enfermedad de Parkinson: introducción y generalidades. Rev Neurol 2010; 50(2):1-5. [ Links ]
8. Benito León J, Bermejo Pareja F, Morales Gonzalez JM, Porta Etessam J, Trincado R, Vega S, Louis ED. Incidence of Parkinson disease and parkinsonism in trhee elderly populations of central Spain. Neurology. 2004;64: 734-41. [ Links ]
9. Anzola PD, Galinsky D, Morales MF, Salas AM. La atención de los ancianos: un desafío para los años noventa. OPS/OMS. 1994; 546: 352-59. [ Links ]
10. Von Elm E, Altman D, Egger M, Pocock S Gotzsche P, Vandenbrouke J. Declaración de la Iniciativa STROBE (Strengthening the Reporting of Observational studies in Epidemiology): directrices para la comunicación de studios observacionales. Gac Sanit. 2008; 22(2): 144-50. [ Links ]
11. Hoehn MM, Yahr MD. Parkinsonismo: Aparición, progresión y mortalidad. Neurology; 1967; 17(5): 427-442. [ Links ]
12. Cid Ruzafa J, Damián Moreno J. Valoración de la discapacidad física: el Índice de Barthel. Rev Esp Salud Pública. 1997; 71: 127-137. [ Links ]
13. Schwab RS, England AC. En Third Symposium on Parkinson's disease. E And S Livingstone: Edinburgh; 152-157, 1969. [ Links ]
14. Hipocampo [sede web]. Cádiz: Ricardo de la Vega y Antonio Zambrano; [actualizado 28 de septiembre de 2014]. Escalas y test; [aproximadamente 1 pantalla]. Disponible en: http://www.hipocampo.org/pfeiffer.asp [ Links ]
15. Generalitat Valenciana. Escalas e Instrumentos para Valoración en Atención Domiciliaria. Valencia: Conselleria de Sanitat; 2006. [ Links ]
16. Martínez-Martín P., Frades Payo B. Quality of life in Parkinson´s disease: validation study of the PDQ-39 Spanish version. The Grupo Centro for Study of Movement Disoders. J Neurol 1998; 245(1): 34-8. [ Links ]
17. Internacional Parkinson's Disease Non-Motor Group. Escala de evaluación de síntomas no motores en la enfermedad de Parkinson. [Monografía en Internet]. Internacional Parkinson's Disease Non-Motor Group [19 de octubre de 2014]. Disponible en: http://www.getm.info/documentos/Escala_de_evaluacion_de_sintomas_no_motores_E_Parkinson.pdf [ Links ]
18. Gómez Picard P, Fuster Culebras J. Atención a la cronicidad: desafío estratégico, macrogestión y políticas de salud. Enfermería Clínica. 2014; 24(1): 12-17. [ Links ]
19. Terriff DL, Williams JVA, Patten SB, Lavorato DH, Bulloch AGM. Patterns of disability, care needs, and quality of life of people with Parkinson's disease in a general population sample. Pakinsonism Relat Disord. 2012; 18: 828-832. [ Links ]
20. Leonardi M, Raggi A, Pagani M, Carella F, Soliveri P, Albanese A, et al. Relationships between disability, quality of life and prevalence of nonmotor symptoms in Parkinson's disease. Parkinsonism Relat Disord. 2012;18:35-39. [ Links ]
21. Tamás G, Gulácsi L, Bereczki D, Baji P, Takáts A, Brodszky V, et al. Quality of Life and Costs in Parkinson's Disease: A Cross Sectional Study in Hungary. Plos One. 2014; 9(9). [ Links ]
22. Cano De la Cuerda R, Vela Desojo L, Miangolarra Page J, Macias Macias Y, Muños Hellin E. Calidad de vida relacionada con la salud en la enfermedad de parkinson. Medicina. 2010; 70: 503-507. [ Links ]
23. Jones JD, Malaty I, Price CC, Okun MS, Bowers D. Health comorbidities and cognition in 1948 with idiopathic Parkinson's disease. 2012; 18: 1073-1078. [ Links ]
24. Magalhaes Navarro-Peternella F, Silva Marcon S. Calidad de vida de las personas con enfermedad de Parkinson y su relación con la evolución en el tiempo y la gravedad de la enfermedad. Latino-Am Enfermagem. 2012; 20(2): 384-91. [ Links ]
25. Berganzo K, Tijero B, et.al. Síntomas no motores en la enfermedad de Parkinson y su relación con la calidad de vida y los distintos subgrupos clínicos. Neurología. 2014. [ Links ]
26. Munhoz R, Moro A, Silveira-Moriyama L, Teive HA. Non-motor signs in Parkinson's disease: a review. Arq Neuropsiquiatr. 2015: 1-9. [ Links ]
27. Yang S, Sajatovic M, Walter B. Psychosocial Interventions for Depression and Anxiety in Parkinson's Disease. Journal of Geriatric Psychiatry and Neurology. 2012; 25(2): 113-121. [ Links ]
28. Carter J, Stewart B, Lyons K, Archbold P. Do Motor and Non-motor Symptoms in PD Patients Predict Caregiver Strain and Depression?. Movement Disorders. 2008; 23(9): 1211-1216. [ Links ]
29. González Mestre A. La autonomía del paciente con enfermedades crónicas: De paciente pasivo a paciente activo. Enfermería Clínica. 2014;24(1): 67-73. [ Links ]
Received: May 22, 2017; Accepted: November 08, 2017











 text in
text in 



