Meu SciELO
Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em
SciELO
Similares em
SciELO -
 Similares em Google
Similares em Google
Compartilhar
Medicina Oral, Patología Oral y Cirugía Bucal (Ed. impresa)
versão impressa ISSN 1698-4447
Med. oral patol. oral cir. bucal (Ed.impr.) vol.10 no.5 Nov./Dez. 2005
Epidermoid carcinoma of the lip: An immunohistochemical study
Estudio inmunohistoquímico del carcinoma epidermoide de labio
Belén García-Montesinos Perea (1), José Fernando Val Bernal (2), Ramón Saiz Bustillo (3)
(1) Médico Adjunto de Cirugía Maxilofacial. Hospital Universitario Marqués de Valdecilla (Santander).
Doctora en Medicina y Cirugía
(2) Jefe de Departamento de Anatomía Patológica. Hospital Universitario Marqués de Valdecilla (Santander).
Catedrático de Anatomía Patológica. Facultad de Medicina. Universidad de Cantabria
(3) Jefe de Servicio de Cirugía Maxilofacial. Hospital Universitario Marqués de Valdecilla (Santander).
Doctor en
Medicina y Cirugía. Profesor asociado de Patología Quirúrgica, Facultad de Medicina, Universidad de
Cantabria.
Address:
Dra. Belén García-Montesinos Perea
C/ Castelar nº 5, 2º derecha.
39004. Santander. Cantabria
E-mail: gmontesinos@humv.es
Received: 16-05-2004 Accepted:22-12-2004
|
García-Montesinos-Perea B, Val-Bernal JF, Saiz-Bustillo R. Epidermoid carcinoma of the lip: An immunohistochemical study. Med Oral Patol Oral Cir Bucal 2005;10:454-61. |
|
ABSTRACT
Objectives: To determine the expression of the c-erb-B2, p53, bcl-2, Ki67 and CD44varV6 proteins, and to establish their prognostic value in epidermoid carcinoma of the lip. Key words: Immunohistochemistry, cancer of the lip, epidermoid carcinoma, c-erb-B2, p53, bcl-2, Ki67, CD44.
|
RESUMEN Objetivos: Determinar la sobreexpresión de las proteínas c-erb-B2, p53, bcl-2, Ki67 y CD44varV6 y establecer su valor pronóstico en el carcinoma epidermoide de labio. Palabras clave: Inmunohistoquímica, cáncer de labio, carcinoma epidermoide, c-erb-B2, p53, bcl-2, ki67, CD44. |
INTRODUCTION
Epidermoid carcinoma of the lip is the most frequent tumor of the oral cavity, accounting for between 25 and 30% of tumors located at this level, and representing 0.6 – 1.0% of all malignant tumors in humans. Immunohistochemical techniques quantify the cellular kinetics, preserving both the tissular architecture and the inter-tissular reactions (1). These techniques operate at the molecular level of the cancer (secondary to the activation of proto-oncogenes such as c-erb-B2, to the inactivation of suppressor genes such as p53, local genetic alterations in those genes that regulate apoptosis such as bcl-2, and the oncogenic viruses) and on the biology of tumor growth (dependent on tumoral kinetics such as the Ki67 protein and the tumoral environment such as the adhesion molecule CD44varV6).
The c-erb-B2 protein is located in the tumoral cellular membrane, above all in the microvilli, and is activated by its fusion with a molecule similar to Tumor Growth Factor alpha (TGF) at the superficial cellular level. Together they codify the overexpression of the normal forms of growth factor receptors, in such a way that they become extremely sensitive to a very small quantity of themselves.
The p53 gene codifies a phosphoprotein which regulates DNA replication, cellular proliferation and cell death (2). In those cells exposed to mutagenic agents, although an isolated mutation is not able to transform cells, the fact that the normal p53 protein does not exist predisposes the cells to additional mutations, and finally, to malignant transformation. In addition to mutation, the normal function of the p53 gene can be altered by fusion with viral onco-proteins or with the individual’s own proteins (3-5).
Protein bcl-2 presents a punctiform cytoplasmic distribution, of intramitochondrial location. The mechanism by which this gene avoids apoptosis is not clear. It is thought that its overexpression confers on it an increased growth pattern, at the same time as the occurrence of mutations which affect the proto-oncogenes and suppressor genes.
Ki67 protein is present in all active phases of the cell cycle, in such a way that it identifies proliferating cells, tumors with a high Ki67 index being those with the worst prognosis (6).
The CD44 and the CD44varV6 adhesion molecules are present in normal epithelium of the head, neck and larynx. Their function seems to be to maintain the integrity of the epithelium, so that their loss is an indicator of malignant transformation of squamous epithelium and is related to the progression of carcinomas of the head and neck (7-9).
The aim of this study is to determine the prognostic value of the following: overexpression of the c-erb-b2, p53 and bcl-2 proteins, the grade of tumor proliferation through the modification of the nuclear protein Ki67, and of the presence or absence of the adhesion molecule CD44varV6 in epidermoid carcinoma of the lip.
MATERIAL AND METHODS
A study of immunohistochemical variables in epidermoid carcinoma of the lip was carried out, taking as a reference population, patients diagnosed and treated for this tumor from 1975 to 1995 in the Cantabrian Community, Spain.
The criteria for inclusion in the study were: the anatomicopathological diagnosis of epidermoid carcinoma of the lip in the surgical specimens; the availability of histological preparations, surgical samples and clinical histories; and the existence of at least five years clinical follow-up for the patients.
Information was obtained from the clinical records of the Maxillofacial Surgery Department, Hospital Universitario Marqués de Valdecilla, Santander, and the corresponding reports from the Pathology Department of the same Hospital.
An experienced pathologist selected one of the preparations from each case, in such a way that there was a sufficient amount of tumor material against the normal cellular population. The paraffin-block samples were located, checking that they corresponded to the previously selected preparation. Within each of these, an area of tumor population of at least 20% against the non-tumoral reference population was marked, avoiding areas of necrosis and hemorrhage. Following this initial selection procedure, the number of samples for immunohistochemical study was reduced to 79 tumors, of which a certain number were excluded as the different analyses were made.
The immunohistochemical study was carried out using the EnVision+system in an automatic staining machine (Dako ®) (table I). In order to quantify the parameters studied in all the cases, other known positive tumors were used as positive control and adjacent non-tumoral tissue was used as negative control. The evaluation was carried out by the same pathologist in accordance with the following criteria:
- Protein expression of c-erb-B2 gene (74 cases):
(-): No tumor cell staining.
(+): Cytoplasmic and/or membranous tumor cell staining.
- Protein expression of p53 suppressor gene (71 cases):
(-): No tumor cell staining.
(+): Between 10 and 50 % of tumor cells stained.
(++): More than 50 % of tumor cells stained.
- Protein expression of bcl-2 apoptosis regulator gene (76 cases):
(-): No tumor cell staining.
(+): Tumor cell staining. Staining also of the lymphocytic infiltrate and the keratin of the parakeratic globules.
- Ki67 tumor proliferation marker (69 cases):
The Simpson method was used to quantify the number of tumor cells in four consecutive high magnification fields (x 40). An estimate was made of the number of Ki67 positive tumor cells per 100 tumor cells.
- CD44varV6 adhesion molecule (79 cases) (Figure 1):
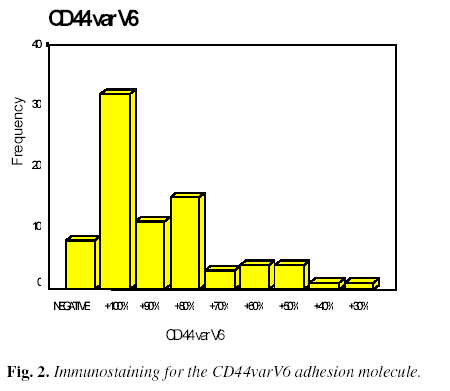
(-): Either no tumor cell staining, or less than 30 % tumor cell staining.
(+): Between 30 and 100% of tumor cells in the invasion area stained. The results were divided into groups of ten.
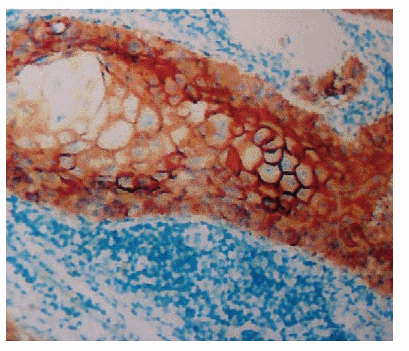
Fig. 1. Immunohistochemical localization of CD44varV6
in the epidermoid carcinoma of the lip.
The statistical analysis was carried out using the SPSS 8.0 program. On studying all the variables there were some cases that could not be evaluated, either because the sample was of insufficient quality following the immunohistochemical study, or because not enough tumoral tissue was available within the preparation for a correct evaluation. Both descriptive and inferential statistics were calculated, and the five-year survival tables were determined from all the variables using the Kaplan-Meier non-parametric model. The statistical significance was evaluated using the Log Rank test. Those variables having individual prognostic significance were studied together using the Cox multiple regression analysis in order to determine those variables with independent prognostic capacity.
RESULTS
Immunostaining for the c-erb-B2 protein was positive in 56 cases, all of these located in the cytoplasm, representing 75% of the tumor samples studied, and negative in the 18 remaining surgical samples.
With respect to the p53 protein, the staining was positive in between 10 and 50% of the tumor cells in 31 cases, representing 43.8% of the total sample; positive in more than 50% of the tumor cells in 19 cases (26.8%), and considered negative in 21 cases (29.6%).
For the bcl-2 protein, of the 76 cases analyzed, 73 were considered negative (96.2%) and only 3 were positive (3.8%).
The values for the Ki67 protein expression varied between a minimum of 0% and a maximum of 6.29%, with an average of 1.64% and a standard deviation of 1.34.
Regarding the study of the CD44varV6 adhesion molecule (Figure 2), positive staining of the invasive area was found in 100% of the tumor cells in 32 cases (40.5%), between 51 and 99% of tumor cells in 33 cases (41.8%), between 0 and 50% in six cases (7.6%), and was negative in only eight cases (10.1%).
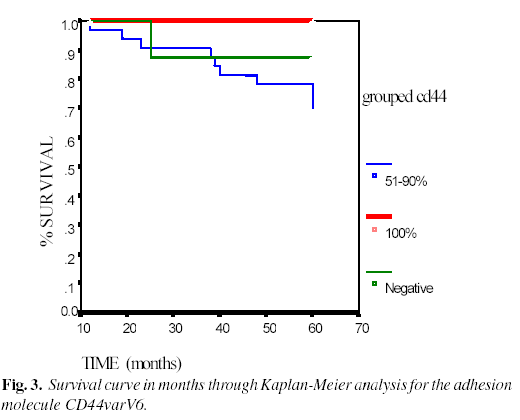
In the survival study using the Kaplan-Meier analysis, and analyzing the results of the different variables studied, a statistical significance was found only in the case of the adhesion molecule CD44varV6, in such a way that as the positive staining for this molecule decreased (Figure 3), the survival of the patients was reduced (p=0.0237). On applying the Cox multiple regression analysis, no independent prognostic significance was demonstrated.
DISCUSSION
Few publications exist on studies of the immunohistochemical variables in epidermoid carcinoma of the lip. In our study, which with 79 cases represents one of the most extensive series to date, we found no significant association between the c-erb-B2 protein and survival, which is in accordance with the majority of series studied on cancer of the head and neck where no important prognostic significance has been demonstrated (10, 11).
There are studies on carcinoma of the larynx in which a possible predictive value for lymph node metastasis has been indicated (12, 13); and in breast-cancer the c-erb-B2 protein has been indicated as a marker of worse prognosis and as a possible predictor for response to treatment (14).
In human cancer, molecular alterations of the p53 gene are the most common genetic finding. In this series no prognostic importance has been demonstrated, and results published in other studies are inconsistent.
Likewise, in a study on tumors of the head and neck it was shown that this factor has no prognostic value (13), and in another series of cutaneous epidermoid carcinomas it was concluded that the protein expression of the p53 suppressor gene is not an independent prognostic factor (15). On the other hand, in a study of carcinoma of the larynx an association between p53 and poor prognosis was found in late stages of the disease, independently of the presence, or otherwise, of cervical metastasis (7); and in other studies the overexpression of p53 was related with a better prognosis in cancer of the head and neck (16).
The relationship between the expression of the p53 suppressor gene and the pathogenesis of cancer of the lip has also been referred. In the 71 epidermoid carcinomas of the lip studied, 70.6% expressed the p53 protein, while in a series of 26 epidermoid carcinoma of the lip (17), this protein was detected in the cellular nucleus in 88% of the cases, in the normal adjacent epithelium in 64% and in the atypical adjacent epithelium in all the cases, demonstrating that ultraviolet radiation can induce mutations in the p53 gene, with a resulting neoplastic transformation. In a multi-centric study on epidermoid carcinoma of the lip, an increase in the expression of this gene was found in tumor tissue, and in areas of chronically photo-exposed skin (18). In another study on benign, premalignant, and malignant lesions of the lip, it was found that p53 expression may be related with the pathogenesis of cancer of the lip and exposure to the sun. For these authors, those patients with benign lesions of the lip with aberrant p53 expression should be given periodic reviews (19).
Regarding the protein expression of the bcl-2 apoptosis regulator gene, 96.2% of the 76 epidermoid carcinoma of the lips studied in our series did not present expression of this protein and no significant relationship was found between this variable and patient survival. In a series of 48 tumors of the head and neck, no prognostic value was found for this factor or p53, nor any relation with the presence of metastasis (20). However, in non-Hodgkin’s lymphomas of B cells, prostate adenocarcinomas, lung carcinomas and nasopharyngeal carcinomas, high levels of this protein and/or aberrant bcl-2 have been found, but with no clear prognostic significance (21).
In many tumors, such as those of the head and neck, it has been shown that proliferative activity correlates with tumor prognosis and progression (22), and that a strong correlation exists between a high and low index of Ki67 and a high or low grade of tumor differentiation (23). In tumors of the head and neck the results of studies on the relationship between different parameters that measure proliferation are controversial.
Likewise, in the results of our study the Ki67 tumor proliferation factor did not correlate significantly with survival, unlike others (7) who, in a study on carcinoma of the larynx, found that Ki67 proliferation correlated negatively with survival, but without achieving statistical significance. They also demonstrated a negative correlation between Ki67 and CD44varV6.
In recent years the Ki67 tumor proliferation index has been investigated as a predictor of response to radiotherapy in tumors of the head and neck, in such a way that the quantification of the Ki67 antigen, before and after radiotherapy, can be a useful parameter in the prediction of the clinical evolution of patients affected by these types of tumors (24). In this regard, Raybaud-Diogene et al. (25) have referred to a poor response to radiotherapy in those patients with tumors of the head and neck with p53 over expression and a fraction of Ki67 tumor proliferation <20%, and finding, furthermore, that these markers have a better predictive value than tumor stage.
Finally, the adhesion molecule CD44varV6, the absence of which is related with tumor progression, was the only immunohistochemical marker studied in which we have found a significant relationship between its presence and greater survival. There are studies of the oral cavity which demonstrate that this molecule is present in normal oral epithelium, maintaining the integrity of the squamous epithelium and tending to disappear in premalignant and malignant lesions. It lso associates significantly with the presence of regional etastases (26, 27). Likewise, in carcinoma of the larynx (7), a ignificant relationship between the presence of CD44varV6 and a etter prognosis was found, as was a relation between the decrease in D44 expression and an increase in the prevalence of metastasis, but ithout significance in this instance. However, not all the published studies have confirmed all of these findings (28).
In conclusion, as can be appreciated from the analysis of our data, and in accordance with the majority of the published series, where the majority of results are usually inconsistent, we can confirm that an immunohistochemical study has no relevant prognostic value in this type of tumor of the lip. In this regard, the immunohistochemical variables analyzed in our series should not generally be used as prognostic indicators, nor should they be used routinely in the anatomicopathological study of these tumors, since the classic and more cost effective clinical and histopathological studies continue to have a better and more significant prognostic value.
REFERENCES
1. Garcia R, Coltrera MB, Gown AM. Analysis of proliferative grade with anti PCNA7 cyclin monoclonal antibodies in fixed embedded tissues. Am J Pathol 1989; 134: 733-9. [ Links ]
2. Harris C, Hollstein M. Clinical implications of the p53 tumor supressor gene. N Eng J Med 1993; 329: 1318-27. [ Links ]
3. Harris C. Chemical and physical carcinogenesis: advances and perspectives for the 1990s. Cancer Res 1991; 51: 5023-44. [ Links ]
4. Werness B, Levine A, Howley P. Association of human papillomavirus types 16 and 18 E6 proteins with p53. Sciencie 1990; 248: 76-9. [ Links ]
5. Oliner JD, Kinzler KW, Meltzer PS, George DL, Vogelstein B. Amplification of a gene encoding a p53 associates protein in human sarcoma. Nature 1992; 358: 80-3. [ Links ]
6. Gerdes J, Li L, Schlueter C, Duchrow M, Wohlenberg C, Gerlach C et al. Inmunobiochemical and molecular biologic characterization of cell proliferation-associated nuclear antigen. That is defined by monoclonal antibody Ki 67. Am J Pathol 1991; 138: 867-73. [ Links ]
7. Hudson D, Speight P, Watt F. Altered expression of CD44 isoforms in squamous cell carcinomas and cell lines derived from these. Int J Cancer 1996; 66: 457-63. [ Links ]
8. Herold-Mende C, Seiter S, Born AI, Patzelt E, Schupp M, Zoller J et al. Expression of CD44 splice variants in squamous epithelia and squamous cell carcinoma of the head and neck. J Pathol 1996; 179: 66-73. [ Links ]
9. Soukka T, Salmi M, Joensuu H, Hakkinen L, Sointu P, Koulu L et al. Regulation of CD44 var v6 containing isoforms during proliferation of normal and malignant epithelial cells. Cancer Res 1997; 57: 2281-9. [ Links ]
10. Craven JM, Pavelic ZP, Stambrook PJ, Pavelic L, Gapany M, Kelley DJ et al. Expression of c-erb-2 gene in human head and neck carcinoma. Anticancer Res 1992; 12: 2273-6. [ Links ]
11. Field J, Spandidos DA, Yiagnisis M, Gosney JR, Papadimitriou K, Stell PM. C-erb-b2 expression in squamous cell carcinoma of the head and neck. Anticancer Res 1992; 12: 613-20. [ Links ]
12. Takes RP, Baatenburg de Joung RJ, Schuuring E, Hermans J, Vis AA, Litvinov SV et al. Marker for assesment of nodal metastasis in laryngeal carcinoma. Arch Otolaryngol Head Neck Surg 1997; 123: 412-9. [ Links ]
13. Weinstein G, Nuamah I, Tucker J, Montone K. Evaluation of HER-2/neu (cerb-B2) oncogene expression in whole organ sections of supraglottic squamous cell carcinoma. Ann Otol Rhinol Laryngol 1996; 105: 275-9. [ Links ]
14. Hynes N. Amplification and overexpression of the cerb-b2 gene in human tumors : its involvement in tumor development, significance as a prognostic factor, and potential as a target for a cancer therapy. Semin Cancer Biol 1993; 4: 19-26. [ Links ]
15. Mansoor A, Mc Kee PH, Simpson JA, McGuire B, Hobbs C. Prognostic significance of ki67 and p53 inmunorreactivity in cutaneous squamous cell carcinomas. Am J Dermatopathol 1996; 18: 351-7. [ Links ]
16. Santer R, Ridge J. p53 overexpression correlates with increased survival in patients with squamous carcinoma of the tongue base. Am J Surg 1992; 164: 651-3. [ Links ]
17. Berner A, Holm R, Naess A , Hjortdal O. p53 protein expression in squamocellular carcinomas of the lip. Anticancer Res 1993; 13: 2421-4. [ Links ]
18. Fabbrocini G, Russo N, Pagliuca MC, Delfino M, Staibano S, Molea G et al. p53, cyclin-D1, PCNA, AgNOR expression in squamous cell cancer of the lip: a multicenter study. Photodermatol Photoimmunol Photomed 2000; 16: 172-7. [ Links ]
19. Crosthwaite N, Teale D, Franklin C, Foster GA, Stringer BM. p53 protein expression in malignant, pre-malignant and non-malignant lesions of the lip. J Clin Pathol 1996; 49: 648-53. [ Links ]
20. Li Ma MD, Adam Ronai PD. Expression of p53 and Bcl-2 in squamous cell carcinoma of the Head and Neck : An inmunohistochemical study. Appl Inmunohistochemi 1998; 6: 77-83. [ Links ]
21. Miyashita T, Krajewski S, Krajewska M, Wang HG, Lin HK, Liebermann DA et al. Tumor supressor P53 is a regulator of bcl-2 and box gene expression in vitro and in vivo. Oncogene 1994; 9: 1799-1805. [ Links ]
22. Tubiana M, Conroli A. Proliferation kinetics in human solid tumors : relation to probability of metastatic dissemination and long term survival. Radiother Oncol 1989; 15: 1-18. [ Links ]
23. Lorz M, Meyer-Breiting E. Determination of cell proliferation using monoclonal antibodies studies of 21 squamous cell carcinoma of the head and neck area. Laryngol Rhinol Otol 1988; 67: 539-42. [ Links ]
24. Ogawa Y, Inomata T. Changes in Ki67 labeling rates of head and neck squamous cell carcinomas during preoperative radiation therapy. Oncology 1992; 49: 450-3. [ Links ]
25. Raybaud-Diogene H, Fortin A, Morency R, Roy J, Monteil RA, Tetu B. Markers of radioresistence in squamous cell carcinomas of the head and neck : A clinicopathologic an inmunohistochemical study. J Clin Oncology 1997; 15: 1030-8. [ Links ]
26. Bahar R, Kunishi M, Kayada Y, Yoshiga K. CD44varV6 expression as a progression marker in benign, premalignant and malignant oral epithelial tissues. Int J Oral Maxillofac 1997; 26: 443-6. [ Links ]
27. Kunishi M, Kayada Y, Yoshiga K. Down regulated expression of CD44varV6 in oral squamous cell carcinomas and its relationship to regional lymph node metastasis. Int J Oral Maxillofac 1997; 26: 280-3. [ Links ]
28. Piffko J, Bankfalvi A, Klauke K, Dreier R, Joos U, Bocker W et al. Unaltered strong inmunohistochemical expression of CD44v6 and CD44v5 isoforms during development and progression of oral squamous cell carcinomas. J Oral Pathol Med 1996; 25: 502-6. [ Links ]











 texto em
texto em