My SciELO
Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista de Osteoporosis y Metabolismo Mineral
On-line version ISSN 2173-2345Print version ISSN 1889-836X
Rev Osteoporos Metab Miner vol.9 n.1 Madrid Jan./Mar. 2017
https://dx.doi.org/10.4321/s1889-836x2017000100003
Originales
Effects of the catalase antioxidant enzyme in vascular calcification and bone demineralization
1Servicio de Metabolismo Óseo y Mineral - Instituto Reina Sofía de Investigación Nefrológica - REDinREN del ISCIII - Hospital Universitario Central de Asturias - Oviedo (España)
2Servicio de Metabolismo Óseo y Mineral - Instituto Reina Sofía de Investigación Nefrológica - REDinREN del ISCIII - Hospital Universitario Central de Asturias - Oviedo (España)
3Servicio de Metabolismo Óseo y Mineral - Instituto Reina Sofía de Investigación Nefrológica - REDinREN del ISCIII - Hospital Universitario Central de Asturias - Oviedo (España)
4Servicio de Metabolismo Óseo y Mineral - Instituto Reina Sofía de Investigación Nefrológica - REDinREN del ISCIII - Hospital Universitario Central de Asturias - Oviedo (España)
5Universidad/Instituto Sanger - Cambridge (Reino Unido)
6Universidad/Instituto Sanger - Cambridge (Reino Unido)
7Departamento de Biología de Sistemas - Universidad de Alcalá de Henares - REDinREN del ISCIII - Alcalá de Henares - Madrid (España)
8Laboratorio de Medicina - Hospital Universitario Central de Asturias - Oviedo (España)
9Laboratorio de Medicina - Hospital Universitario Central de Asturias - Oviedo (España)
10Servicio de Metabolismo Óseo y Mineral - Instituto Reina Sofía de Investigación Nefrológica - REDinREN del ISCIII - Hospital Universitario Central de Asturias - Oviedo (España)
11Departamento de Biología de Sistemas - Universidad de Alcalá de Henares - REDinREN del ISCIII - Alcalá de Henares - Madrid (España)
12Servicio de Metabolismo Óseo y Mineral - Instituto Reina Sofía de Investigación Nefrológica - REDinREN del ISCIII - Hospital Universitario Central de Asturias - Oviedo (España)
14Departamento de Medicina - Universidad de Oviedo - Oviedo (España)
13Servicio de Metabolismo Óseo y Mineral - Instituto Reina Sofía de Investigación Nefrológica - REDinREN del ISCIII - Hospital Universitario Central de Asturias - Oviedo (España)
Introduction
Cardiovascular disease currently represents the leading cause of death in the developed world. It is expected to continue rising in the coming decades due to our aging population. One factor that contributes to cardiovascular risk is oxidative stress [1]. Different stimuli are associated with the development of cardiovascular disease including macrophage activation, hyperglycemia, LDL oxidation and even angiotensin II which exert their harmful effects, at least partially, through the local synthesis of reactive oxygen species [2,3,4,5].
On the other hand, there is evidence of a positive relationship between oxidative stress and vascular calcifications [6]. Vascular smooth muscle cells (VSMC) subjected to oxidative stress increase the activity of alkaline phosphatase and calcium deposition, indicating their transdifferentiation toward cells capable of mineralizing (osteoblast-like/chondrocyte) [7]. In VSMC in primary culture of mouse aorta, the oxidative stress induced by hydrogen peroxide or glucose oxidase promotes calcification and overexpression of Runx2 (Cbfa1), transcription factor related bone osteogenic differentiation, mediated AKT [8]. Interestingly, these same stimuli have the opposite effect on osteoblast precursor cells, demonstrating the importance of hydrogen peroxide or oxygenated water (H2O2) in the process of differentiation of mesenchymal cells lineage [9].
The main antioxidant enzymes involved in the catalytic removal of hydrogen peroxide is catalase, glutathione peroxidases and thyroiodin peroxidase. Of these, the catalase is the most efficient in removing the hydrogen peroxide enzyme.
One of the factors affecting aging is the progressive accumulation of oxidative damage. This damage may be due to exposure to normal intracellular oxidative stress or, in pathological situations, an increase in this stress due to inflammation or other causes can actually cause accelerated aging. Therefore, our objective was to evaluate the role played by overexpression of catalase on the process of vascular calcification associated with moderate kidney disease and its effect on bone mass [10]. For this purpose, a transgenic mouse model overexpressing catalase enzyme, subjected to chronic renal failure was used.
Material and methods
Experimental model in vivo
Establishment of chronic renal failure (CRF)
C57/BL6J wild (WT) and C57/BL6J transgenic (TG) overexpressing the antioxidant enzyme catalase were used. To induce CRF, mice underwent 3 months old to a first intervention, which involved the lateral opening of the animal on the right side where the two poles of the kidney is cauterized. Isoflurane anesthesia was used (1-2%) by inhalation. A week after the first operation, the animal underwent a second intervention, which consisted of opening the left side and complete removal of the kidney. After 16 weeks of the last operation, the animals were sacrificed by exsanguination, anesthetized with CO2. In slaughter animal serum he was obtained to analyze and general biochemical markers of bone metabolism: urea, calcium, phosphorus, Ca-P product, iPTH and FGF23. the remaining piece of kidney, aorta and tibias were also extracted.
The left tibia, which was preserved in ethanol 70º, was analyzed by computerized microtomography (µCT) with a SkyScan 1174, Bruker µCT (Kontich, Belgium) equipment. The 2D and 3D morphometric analysis was carried out using the CTAn software. The region of interest (ROI) was defined manually in each sample. For the trabecular region, 150 cuts were selected and threshold levels used in the gray scale between 78 and 250. The morphometric analyses were based on internal plug-ins CTAN in 2D and 3D. Morphometric parameters were measured trabecular bone volume (BV/TV,%), trabecular spacing (TbSp, µm), trabecular number (ToNb, mm-1) and trabecular porosity (TbPo, µm).
All studies were approved and authorized by the Committee on Animal Experimentation of the University of Oviedo.
In vitro experimental model
Primary cultures of aortic VSMCs from C57/BLJ6 WT and TG were used. To do this, the aortas of animals chopped and put explants to grow in culture dishes coated with fibronectin to promote adhesion.
Cells were cultured in DMEM medium supplemented with fetal bovine serum at 10%. When cells reached 60-70% confluency was replaced with DMEM-F12 supplemented medium with 0.1% bovine albumin (control medium) and calcifying medium supplemented medium consisting of control phosphorus and calcium at concentrations of 3 mM and 2 mM, respectively. Cells were incubated under these conditions for 8 days.
The basal activity of catalase was measured in VSMC from WT and TG mice using the commercial kit "catalase assay kit" (Cayman Chemical, 707002), following the protocol established by the manufacturer.
Levels of reactive oxygen species in VSMC cultured WT and TG mice with control medium and calcifying were measured with a fluorochrome specific mitochondria, dihydrorhodamine 123 (DHR 123).
Proteins of cell cultures were extracted with a buffer composition 50 mM Tris-HCl, 150 mM NaCl, 1% NP-40, sodium deoxycholate 0.5%, 1.0 mM EDTA and 0.1% SDS inhibitor proteases. the sample was sonicated in cold 10 minutes to prevent the breakdown of proteins and centrifuged at 10,000 rpm for 5 minutes at 4°C proteins of the supernatant was collected, quantified by the Bio-Rad DC method and stored at -80.
20 µg of protein were electrophoresed on acrylamide gels of 0.75 mm thickness under denaturing conditions (SDS-PAGE), for identification using known molecular weight markers (Molecular Weight Markers RainbowTM, GE Healthcare, United Kingdom). Proteins were transferred to a nitrocellulose membrane (Hybond AmershamTM 0.45 µm PVDF, GE Healthcare, UK). The transfer was carried out in the cold for one hour at 100 v. After the same, the membrane was blocked for one hour with 5% milk in phosphate buffered saline (PBS). Subsequently, membranes were kept overnight with the primary antibody (monoclonal antibody of catalase in 1: 5000 dilution in BSA, Runx2 1: 500 in BSA against glyceraldehyde 3-phosphate dehydrogenase (GAPDH) diluted 1: 5000 in BSA, all from Santa Cruz Biotechnology, Inc., Santa Cruz, California, USA). The next day, the membrane was washed three times at intervals of 10 minutes each with wash solution composed of PBS and Tween-20 (Sigma-Aldrich), and subsequently incubated with antibody or goat against rabbit. The membrane was washed with the washing solution described above and the protein was detected by ECL Western ClarityTM Substrate (BIO-RAD, USA) commercial kit. The relative quantification of the intensity of the bands obtained in the Western blotting was performed with Image LabTM Software and Molecular Imager scanner ChemiDocTM XRS+ (both from BIO-RAD, USA).
Common techniques employed
qRT-PCR
The analysis by qRT-PCR was performed with the cDNA obtained from different experimental conditions using the High Capacity Reverse Transcription Kit (Applied Biosystems) and phenol RNA extraction. For normalization of results constitutive expression of genes (rRNA Runx2 and GAPDH) and ΔΔCT relative quantification method was used.
Results
a) In vivo studies:
Biochemical markers
The CRF in WT and TG mice showed increased levels of urea, Ca-P product, and iPTH FGF23. In the case of FGF23 increase it was much higher (10 times) in WT mice with CRF regarding their Sham control.
Moderate CRF effect on vascular calcification and bone level changes
Although the C57/BL6J mouse strain used for the calcification studies did not allow us to obtain vascular calcification in the aortas (Table 1), a significant increase in gene expression of Runx2, osteoblast differentiation marker, was observed only in the WT group with CRF (Figure 1B. This increase was not observed in the TG animals with CRF. The effect of CRF on the calcification process was studied in other soft tissue such as the kidney, with an increase of calcium content in CRF mice with a higher increase in WT than in TG mice (Table 1).
Table 1 Values of biochemical WT and TG mice with and without CRF

tfn2WT: wild mouse; TG: transgenic mouse; CRF: chronic renal failure. *p<0.05 with respect to their respective Sham.

Figure 1 Levels of gene expression of Runx2 (relative units) in aorta in different groups.WT: wild mouse; TG: transgenic mouse; CRF: chronic renal failure
Vascular level changes were also observed at bone level. In WT animals with CKD deterioration of bone structure it was about its Sham control group with a statistically significant decrease in trabecular bone volume (Figure 2A) and the number of trabeculae (Figure 2C), and increased trabecular separation (Figure 2B) and trabecular porosity (Figure 2D). The group TG mice with CRF did not differ with respect to their Sham control group at trabecular level.
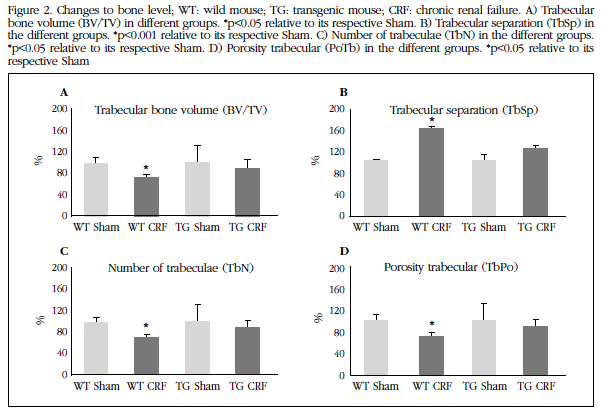
Figure 2 Changes to bone level; WT: wild mouse;TG: transgenic mouse; CRF: chronic renal failure. A) Trabecular bone volume (BV/TV) in different groups. *p<0.05 relative to its respective Sham. B) Trabecular separation (TbSp) in the different groups. *p<0.001 relative to its respective Sham. C) Number of trabeculae (TbN) in the different groups. *p<0.05 relative to its respective Sham. D) Porosity trabecular (PoTb) in the different groups. *p<0.05 relative to its respective Sham
b) In vitro studies:
Effect of overexpression of catalase in the hydrogen peroxide purification and Runx2 expression in VSMCs
The VSMCs from the TG mice had increased activity and protein expression of the catalase enzyme at baseline compared with those of WT mice (Figure 3 A and B).

Figure 3 A) Basal Activity of catalase enzyme in WT and TG VSMC mice.B) Protein expression in relative units of catalase enzyme in VSMC WT and TG mice by Western blotting; WT: wild mouse; TG: transgenic mouse. *p<0.001 compared to WT group
When calcifying medium was added to VSMC WT mice there was increased calcium content. This effect was not observed in VSMC TG mice (Figure 4A). In parallel, the protein expression of Runx2 was studied in VSMC of WT and TG mice with calcifying medium for 8 days and an increase in Runx2 expression in VSMC of WT mice was observed which was not observed in TG (Figure 4B).
Study of the levels of markers of oxidative stress and protein levels of catalase
An increased level of fluorescence of the probe DHR123 was observed in the VSMC of WT mice cultured with calcifying medium, indicating an increase in mitochondrial oxygen reactive species. This effect was not observed in VSMC of TG mice, but there was a decrease of reactive oxygen species (Figure 5). Lower protein expression was also observed of the enzyme catalase by culturing the VSMC of WT mice for 8 days with calcifying medium relative to those from TG mice (Figure 6).

Figure 5 Effect of addition of calcifying medium (Ca and P) on the levels of reactive oxygen species in mitochondrial VSMC WT and TG mice.UR: relative units DHR123 fluorescence probe. WT: wild mouse; TG: transgenic mouse. *p<0.05 compared to control
Discussion
In this work we have been able to confirm that overexpression of catalase antioxidant enzyme in a mouse model protects the process of vascular calcification and bone deterioration. In the VSMC of these same mice, a decrease was observed in the levels of reactive oxygen species, but also osteogenic proteins such as Runx2.
At the biochemical level, increased FGF23 was particularly noticeable in WT animals with CRF regarding the Sham group (10 times). This effect was less marked in the TG with CRF whose increase was twice that of the control group (Sham). Some authors have postulated the role of FGF23 as a calcification inducer [11,12]. It has even been associated with high levels of renal mortality in patients [13]. On the other hand, it inhibits FGF23 expression of CYP27B1 gene encoding the alpha 1-hydroxylase suppressing renal calcitriol synthesis from its precursor 25-hydroxyvitamin D3 [14]. Furthermore, FGF23 activates CYP24 gene expression that encodes 24 hydroxylase, the enzyme that hydrolyzes and inactivates calcitriol [15]. This contributes to the decline in the synthesis of calcitriol, but also to degradation which leads to decreased levels of vitamin D, a factor that could induce vascular calcification as has been shown in epidemiological studies [16,17].
The calcium content in the aorta of TG mice with CRF was similar to WT mice with CRF. While this may seem paradoxical and contradictory to other observed results, Giachelli et al. have confirmed the absence of calcification in the aorta in the same strain of mice used for transgenic generation [18]. However, the protective effect of the overexpression of catalase preventing calcium accumulation was observed in other soft tissue such as the kidney.
Moreover, high levels of FGF23 have been associated with suppression of osteoblast differentiation and mineralization of bone matrix in vitro [19]. This could perhaps explain the negative effect of CRF on bone deterioration in WT mice and to a much lesser extent in the TG mice. Recent studies by our group have shown bone loss in a rat model with CRF and high levels of phosphorus, PTH and FGF23. In the same study, gene silencing studies have confirmed that FGF23 only has a direct effect activating Dkk1 [20], inhibitor of Wnt pathway which is involved in pathway inactivation. Thus, high levels of FGF23, as observed in our experimental model in WT mice with CRF, could have contributed to the decrease in bone mineral density through inactivation of the Wnt pathway.
The increase in reactive oxygen species contributes to increased osteogenic protein, being a stimulus for the start of the calcification process [7,8]. This effect has been observed in VSMC of WT mice subjected to calcifying stimuli. However, in VSMC of TG mice subjected to a calcifying stimulus, not only did the expression of reactive oxygen species not increase but it decreased, as with the protein expression of Runx2, osteogenic protein and early marker process calcification. In fact, the importance of hydrogen peroxide has noted as a second messenger involved in intracellular signaling [21] regulated by oxidative stress. A drop of hydrogen peroxide by catalase overexpression contributes to lower oxidative stress and decreased vascular calcification process through inhibition of Runx2.
In view of the results obtained, studies are needed to ascertain the mechanisms by which decreased oxidative stress confers an advantage at both vascular and bone level. The reduction of the inflammatory process, maintaining protein levels of renal Klotho, the main molecule involved in aging, or regulation of the Wnt pathway may help explain the different behavior, so further research into the field is required.
In summary, the overexpression of catalase enzyme reduced the calcification process both in vivo and in vitro, showing in vivo that this decline was accompanied by an improvement in bone parameters studied.
Referencias
1 Stocker R, Keaney JJ. Role of oxidative modifications in atherosclerosis. Physiol Rev. 2004;84:1381-478. [ Links ]
2 Rajagopalan S, Meng XP, Ramasamy S, Harrison DG, Galis ZS. Reactive oxygen species produced by macrophage-derived foam cells regulate the activity of vascular matrix metalloproteinases in vitro. Implications for atherosclerotic plaque stability. J Clin Invest. 1996;98:2572-9. [ Links ]
3 Ushio-Fukai M, Alexander RW, Akers M, Yin Q, Fujio Y, Walsh K, et al. Reactive oxygen species mediate the activation of Akt/Protein kinase B by angiotensin II in vascular smooth muscle cells. J Biol Chem. 1999;274:22699-704. [ Links ]
4 Uemura S, Matsushita H, Li W, Glassford AJ, Asagami T, Lee KH, et al. Diabetes mellitus enhances vascular matrix metalloproteinase activity: role of oxidative stress. Circ Res. 2001;88:1291-8. [ Links ]
5 Zmijewski JW, Moellering DR, Le Goffe C, Landar A, Ramachandran A, Darley-Usmar VM. Oxidized LDL induces mitochondrially associated reactive oxygen/nitrogen species formation in endothelial cells. Am Physiol Heart Circ Physiol. 2005;2:852-61. [ Links ]
6 Sutra T, Morena M, Bargnoux AS, Caporiccio B, Canaud B, Cristol JP. Superoxide production: a procalcifying cell signalling event in osteoblastic differentiation of vascular smooth muscle cells exposed to calcification media. Free Radic Res. 2008;42:789-97. [ Links ]
7 Mody N, Parhami F, Sarafian TA, Demer LL. Oxidative stress modulates osteoblastic differentiation of vascular and bone cells. Free Radic Biol Med. 2001;31:509-19. [ Links ]
8 Byon CH, Javed A, Dai Q, Kappes JC, Clemens TL, Darley-Usmar VM, et al. Oxidative stress induces vascular calcification through modulation of the osteogenic transcription factor Runx2 by AKT signalling. J Biol Chem. 2008;283:15319-27. [ Links ]
9 Wang L, Zhang YG, Wang XM, Ma LF, Zhang YM. Narigin products human adipose-derived mesenchymal stem cells against hydrogen peroxide-induced inhibition of osteogenic differentiation. Chem Biol Interact. 2015;242:255-61. [ Links ]
10 Roman-Garcia P, Carrillo-Lopez N, Fernandez-Martin JL, Naves-Diaz M, Ruiz-Torres MP, Cannata-Andia JB. High phosphorus diet induces vascular calcification, a related decrease in bone mass and changes in the aortic gene expression. Bone. 2010;46:121-8. [ Links ]
11 Fang Y, Ginsberg C, Sugatani T, Monier-Faugere MC, Malluche H, Hruska KA. Early chronic kidney disease-mineral bone disorder stimulates vascular calcification. Kidney Int. 2014;85:142-50. [ Links ]
12 Jimbo R, Kawakami-Mori F, Mu S, Hirohama D, Majtan B, Shimizu Y, et al. Fibroblast growth factor 23 accelerates phosphate-induced vascular calcification in the absence of Klotho deficiency. Kidney Int. 2014;85:1103-11. [ Links ]
13 Gutiérrez OM, Mannstadt M, Isakova T, Rauh-Hain JA, Tamez H, Shah A, et al. Fibroblast growth factor 23 and mortality among patients undergoing hemodialysis. N Engl J Med. 2008;359:584-92. [ Links ]
14 Shimada T, Hasegawa H, Yamazaki Y, Muto T, Hino R, Takeuchi Y, et al. FGF-23 is a potent regulator of vitamin D metabolism and phosphate homeostasis. J Bone Miner Res. 2004;19:429-35. [ Links ]
15 Liu S, Tang W, Zhou J, Stubbs JR, Luo Q, Pi M, et al. Fibroblast growth factor 23 is a counter-regulatory phosphaturic hormone for vitamin D. J Am Soc Nephrol. 2006;17:1305-15. [ Links ]
16 Dobnig H, Pilz S, Scharnagl H, Renner W, Seelhorst U, Wellnitz B, et al. Independent association of low serum 25-hydroxyvitamin d and 1, 25-dihydroxyvitamin d levels with all-cause and cardiovascular mortality. Arch Intern Med. 2008;168:1340–9. [ Links ]
17 Naves-Díaz M, Cabezas-Rodríguez I, Barrio-Vázquez S, Fernández E, Díaz-López JB, Cannata-Andía JB. Low calcidiol levels and risk of progression of aortic calcification. Osteoporos Int. 2012;23:1177-82. [ Links ]
18 Lau WL, Linnes M, Chu EY, Foster BL, Bartley BA, Somerman MJ, et al. High phosphate feeding promotes mineral and bone abnormalities in mice with chronic kidney disease. Nephrol Dial Transplant. 2013;28:62-9. [ Links ]
19 Wang H, Yoshiko Y, Yamamoto R, Minamizaki T, Kozai K, Tanne K, et al. Overexpression of fibroblast growth factor 23 suppresses osteoblast differentiation and matrix mineralization in vitro. J Bone Miner Res. 2008;23:939-48. [ Links ]
20 Carrillo-López N, Panizo S, Alonso-Montes C, Román-García P, Rodríguez I, Martínez-Salgado C, et al. Direct inhibition of osteoblastic Wnt pathway by fibroblast growth factor 23 contributes to bone loss in chronic kidney disease. Kidney Int. 2016;90:77-89. [ Links ]
21 Cozzolino M, Brenna I, Ciceri P, Volpi E, Cusi D, Brancaccio D. Vascular calcification in chronic kidney disease: a changing scenario. J Nephrol. 2011;24(Suppl 18):S3-S10. [ Links ]











 text in
text in 




