INTRODUCTION
Diabetic mellitus is the condition arising due to abnormal metabolism of carbohydrate, proteins and fats. It is caused by insulin deficiency, often combined with insulin resistance1.
As the number of the people with diabetes multiplies worldwide, the disease has taken an ever-increasing share of national and international health care budgets2. It is projected to become of the world’s main disablers and killers within the next 25 years3) (4.
Currently, the treatments of diabetes, in addition to insulin supplement mainly include many oral hypoglycemic agents like sulfonylureas, biguanides, thiazolidines, D-phenylalanine derivatives, meglitinides and ?-glucosidase inhibitors along with an appropriate diet and exercise. However, none can be considered to be the ideal one, due to their toxic side effects and sometimes diminution in response after prolonged use5) (6. Therefore, continuous efforts are being made to develop new compounds or combinations especially of herbal origin. Plants represent a vast source of potentially useful dietary supplements for improving blood glucose control and preventing long-term complications in type 2 diabetes mellitus7) (8. Traditional plant medicines are used throughout the world for a range of diabetic presentation. Numerous plants have been documented as beneficial in the treatment of diabetes8. However, the majority of traditional antidiabetic plants still await a proper scientific and medical evaluation of their ability to improve blood glucose control and or to prevent the diabetic complications9. Thus, there is a vital need to undertake a systematic study so as to explore the possible mechanism(s) of action of the traditional anti-diabetic plants.
Murraya koenigii, belonging to the family Rutaceae, commonly known as curry-leaf tree, is a native of India, Sri Lanka and other south Asian countries10. It is found almost everywhere in the Indian subcontinent. It shares the aromatic nature, more or less deciduous shrub or tree up to 6 m in height and 15-40 cm in diameter with short trunk, thin, smooth gray or brown bark and dense shady crown11.The plant has been reported to exhibit a wide array of activities including antidiabetic, hypocholesterolemic and in vivo hypoglycemic activity, etc.12 .13 .14-(19. The green leaves of the plant are used to treat piles, inflammation, itching, fresh cuts, dysentery, vomiting and dropsy15) (16.
Catharanthus roseus L., which belongs to family Apocynaceae, is commonly known as ‘periwinkle’. It is an important source of indole alkaloids, which are present in all plant parts17. It has been used for the treatment of diabetes, fever, malaria, throat infections, and chest complaints. It is also used for the regulation of menstrual cycles, and as a euphoriant18. The physiologically important and antineoplastic alkaloids namely Vincristine and Vinblastine are mainly present in the leaves whereas antihypertensive alkaloids such as ajmalicine, serpentine, and reserpine are reported to be present in the roots17) (19. Vincristine and Vinblastine alkaloids are used in the treatment of various types of lymphoma and leukemia17) (20 .21 22.
The present study was therefore, undertaken to verify the hypoglycemic potential of the leaves of M. koenigii and C. roseus by using various in-vitro techniques to explore their probable mechanism(s) of action.
MATERIALS AND METHODS
Chemicals and reagents
Glucose oxidase peroxidase kit was purchased from Pathozyme Diagnostics, Kagal, Maharashtra, India. Dialysis bags (12,000 MW cutoff; Himedia laboratories, India) were used in the study. All the chemicals used in the present study were of extra pure analytical grade.
Plant material
The leaves of M. koenigii and C. roseus were collected from the local areas of Kasegaon, District Sangli, (MS), India. The plant material was further identified and authenticated by the Department of Botany, Yashwantrao Chavan College of Science, Karad. The leaves of M. koenigii and C. roseus were cleaned thoroughly, dried in a hot air oven (50 °C), powdered, passed through 60 mesh sieve (BS) and stored in an airtight container at 4 °C till further use.
Preparation of plant extracts
Aqueous extracts were prepared by extracting the powders of leaves of M. koenigii and C. roseus with hot water (70 °C) in a mechanical shaker (24 h), filtered and freeze dried.
Evaluation of hypoglycemic activity of plant extracts using various in vitro methods
Determination of glucose adsorption capacity
The samples of plant extracts (1%) were added to 25 ml of glucose solution of increasing concentration (5, 10, 20, 50 and 100 mM) 9. The mixture was stirred well, incubated in a shaker water bath at 37 °C for 6 h, centrifuged at 4,000×g for 20 min and the glucose content in the supernatant was determined9) (23) (24. The concentration of bound glucose was calculated using the following Formula 1 25.
Effect of plant extracts on in-vitro glucose diffusion
25 ml of glucose solution (20 mM) and the samples of plant extracts (1%) were dialyzed in dialysis bags against 200 ml of distilled water at 37 °C in a shaker water bath9) (23) (26. The glucose content in the dialysate was determined at 30, 60, 120 and 180 min using glucose oxidase peroxidase diagnostic kit9) (23) (26. A control test was carried out without sample. Glucose dialysis retardation index (GDRI) was calculated by using the following Formula 2 23) (24.
Effect of plant extracts on in-vitro amylolysis kinetics
40 grams of potato starch was added to ≈900 ml of 0.05 M phosphate buffer (pH 6.5)9) (27. The solution after stirring at 65 °C for 30 min was made up to a final volume of 1000 ml to give a 4% (w/v) starch solution. 25 ml of the above starch solution, ?-amylase (0.4%), and the plant extracts (1%) were dialyzed in a dialysis bags against 200 ml of distilled water at 37 °C (pH 7.0) in a shaker water bath9) (23) (26. The glucose content in the dialysate was determined at 30, 60, 120 and 180 min. A control test was carried out without sample9) (23) (25) (26. Glucose dialysis retardation index (GDRI) was determined by using the formula mentioned in the method of in-vitro glucose diffusion.
Glucose uptake by yeast cells
Commercial baker’s yeast was washed by repeated centrifugation (3,000×g; 5 min) in distilled water until the supernatant fluids were clear and a 10% (v/v) suspension was prepared in distilled water9) (26. Various concentrations of extracts (1-5 mg) were added to 1 ml of glucose solution (5-25 mM) and incubated together for 10 min at 37 °C. The reaction was started by adding 100 μl of yeast suspension, vortexed and further incubated at 37 °C for 60 min9) (23) (26. After 60 min, the tubes were centrifuged (2,500 × g, 5 min) and glucose was estimated in the supernatant9) (26. The percent increase in glucose uptake by yeast cells was calculated using the following Formula 3 24) (28.
Where, Abs control is the absorbance of the control reaction (containing all reagents except the test sample), and Abs sample is the absorbance of the test sample.
RESULTS AND DISCUSSION
The results of the glucose adsorption capacity exhibited by the selected plant extracts are represented in Figure 1. The results of the studies on glucose adsorption capacity showed that the extracts of M. koenigii and C. roseus could bind glucose effectively. The glucose adsorption capacity shown by the extracts was found to be directly proportional to the glucose concentration. It was also revealed that both the plant extracts were effective in adsorbing glucose at both low and higher concentrations namely 5 and 100 mmol L-1 glucose used in the study. The glucose adsorption capacity of the selected plant extracts was observed to be directly proportional to the molar concentration of glucose9) (23) (26. A relatively higher amount of glucose was found to be bound with an increased glucose concentration. No significant (p≤0.05) differences were marked between the adsorption capacities of M. koenigii and C. roseus. It was also indicated from the study that the extracts of M. koenigii and C. roseus could effectively bind glucose even at lower concentrations of glucose (5 mM) thereby decreasing the amount of glucose and retarding its transport across the intestinal lumen. Thus, it contributes in blunting the postprandial hyperglycemia. The present findings are in accordance to the observations reported for insoluble fiber-rich fractions isolated from Averrhoa carambola and aqueous extract of bark of Albizzia lebbeck7) (9) (23) (29.
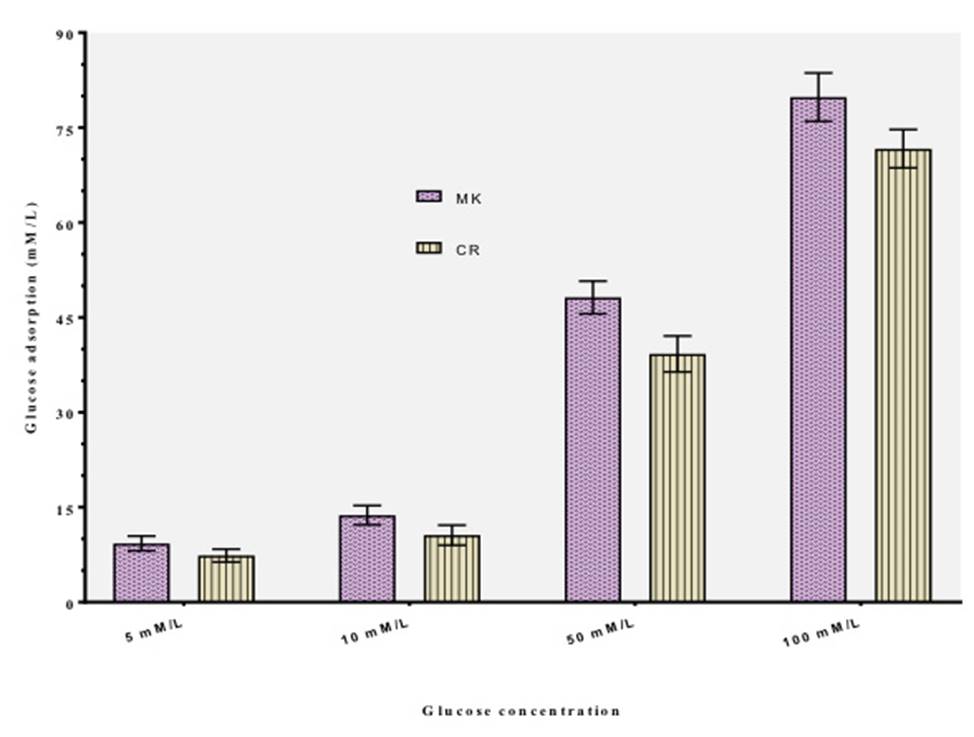
Figure 1: Glucose binding capacity of M. koenigii and C. roseus at different concentrations of glucose. Values are mean + SD of triplicate determinations
Table 1 highlights the results of the effect of the extracts of M. koenigii and C. roseus on in-vitro glucose diffusion. The movement of glucose diffusion across the dialysis membrane was monitored once in 30 min till 180 min9) (23) (26.The rate of diffusion of glucose across the dialysis membrane was found to increase with time from 30 to 180 min9) (26. Both the samples of plant extracts exhibited significant inhibitory effects on movement of glucose into the external solution across the dialysis membrane as compared to9) (23) (26. GDRI was determined on the basis of the retardation of glucose diffusion. It was observed that the retardation of glucose diffusion by the extract of M. koenigii was significantly higher (p≤0.05) than C. roseus. The aforesaid effect of the extract of M. koenigii was reflected with its higher glucose dialysis retardation index (GDRI) value than those observed for the extract of C. roseus.
Table 1: Effect of selected samples on glucose diffusion and glucose dialysis retardation index
| Sample | Glucose content in dialysate (mM) | |||
|---|---|---|---|---|
| 30min | 60min | 120min | 180min | |
| Control | 0.81c+0.01 | 1.39c+0.01 | 1.60c+0.01 | 1.93c+0.01 |
| M. koenigii | 0.43a+0.01(46.92) | 1.05a+0.01 (24.47) | 1.39a+0.01(13.13) | 1.73a+0.01(10.37) |
| C. roseus | 0.48b+0.01(40.75) | 1.11b+0.01 (20.15) | 1.44b+0.01(10.0) | 1.85b+0.01(4.15) |
| Values in parenthesis indicate glucose dialysis retardation index (GDRI). Mean values (n=3) with different superscript letters in columns differ significantly from each other (p≤0.05) | ||||
Table 2 illustrates the effects of extracts of M. koenigii and C. roseus on the amylolysis kinetics model. Glucose dialysis retardation index (GDRI) which is determined on the basis of the retardation of diffusion of glucose diffusion, is considered to be an important in -vitro index to assess the effect of a fiber on the delay in glucose absorption in the gastrointestinal tract9) (23) (26)30(33. A relatively higher GDRI indicates a higher retardation index of glucose by the sample. The GDRI was observed to be 44.0% and 29.0% for M. koenigii and C. roseus respectively at 60 min which gradually got reduced to 27.28% and 18.19% respectively at 120 min. Several possible factors have been mentioned that may be responsible for the inhibition of alpha amylase enzyme; namely fiber concentration, the presence of inhibitors on fibers, encapsulation of starch and enzyme by the fibers present in the sample, thereby reducing accessibility of starch to the enzyme, and direct adsorption of the enzyme on fibers, leading to decreased amylase activity9) (23) (25) (26.
Table 2: Effect of selected samples on starch digestibility and glucose dialysis retardation index
| Sample | Glucose content in dialysate(mM) | |||
|---|---|---|---|---|
| 30 min | 60 min | 120 min | 180 min | |
| Control | 0.0 | 0.25c+0.01 | 0.33c+0.01 | 0.43c+0.01 |
| M. koenigii | 0.0 (100) | 0.14a+0.01 (44.0) | 0.24a+0.01(27.28) | 0.37a+0.01(13.96) |
| C. roseus | 0.0 (100) | 0.18b+0.01 (28.0) | 0.27b+0.01(18.19) | 0.40b+0.01(6.98) |
| Values in parenthesis indicate glucose dialysis retardation index (GDRI). Mean values (n=3) with different superscript letters in columns differ significantly from each other (p≤0.05) | ||||
Our observations greatly emphasize that inhibition of the alpha amylase enzyme may be one of the probable mechanisms through which the extracts of M. koenigii and C. roseus exert their hypoglycemic effect. The inhibitors of carbohydrate hydrolyzing enzymes promotes a delay in the digestion of carbohydrate thereby prolonging the overall carbohydrate digestion time to cause a reduction in the rate of absorption of glucose and consequently blunting the postprandial plasma glucose rise23) (26) (34) (35. Several inhibitors of alpha amylase enzyme have been recently developed from natural sources and some of them in clinical use are Acarbose, Miglitol and Voglibose35.

Figure 2: Effect of M. koenigii and C. roseus extract on the uptake of glucose by yeast cells. Values are mean ± SD of triplicate determinations
Figure 2 highlight the rate of glucose transport across cell membrane in yeast cells system for the extracts of M. koenigii and C. roseus. The mechanism of transport of glucose across the yeast cell membrane have received attention and has been considered as an important technique for in-vitro screening of hypoglycemic activity of various compounds/ medicinal plants9) (23) (26. The results of the study revealed that both the extracts under study promoted transport of glucose across the yeast cells. The amount of glucose which remains in the medium after a specific time interval acts as a measure of the glucose uptake by the yeast cells9) (23) (26. The rate of uptake of glucose into the yeast cells was found to be linear in all the 5 glucose concentrations used in this study. It was observed that the extracts of M. koenigii and C. roseus has improved glucose uptake by Saccharomyces cerevisiae yeast cells in a dose-dependent manner, with improved glucose uptake increasing proportionally as the concentration of glucose in the medium increases9) (23) (26. However, the extract of M. koenigii exhibited significantly higher (p≤0.05) activity than the extract of C. roseus at all concentrations used in the study. The percent increase in the uptake of glucose by the yeast cells was found to be inversely proportional to the concentration of glucose and got decreased with an increase in the molar concentration of the glucose solution. Previous studies involving the transport of non metabolizable sugars, metabolizable glycosides have suggested that the transport of sugar across the yeast cell membrane is mediated by stereo specific membrane carriers and usually takes place by the process of facilitated diffusion23) (31) (32) (36.
CONCLUSION
In conclusion, the results of the present investigation highlighted the hypoglycemic activity of M. koenigii and C. roseus as assessed by various in-vitro methods. Inspite of the fact that in-vitro screening is not a reliable predictor of hypoglycemic effect in vivo, the various model systems used in the present study would provide an insight on the possible mechanism by which the extracts of M. koenigii and C. roseus may contribute in lowering the postprandial glucose levels. The hypoglycemic effect exhibited by the extracts of M. koenigii and C. roseus is observed to be mediated by increasing glucose adsorption, decreasing glucose diffusion rate and at the cellular level by promoting the transport of glucose across the cell membrane as highlighted by employing simple in-vitro model of yeast cells. These observed effects further, need to be confirmed by employing different in vivo models and clinical trials which may contribute for their effective utilization as an adjunct in effective management of diabetes mellitus.

















