Introduction
Prostate cancer (PC) is the most common urogenital malignancy in older men and the second leading cause of cancer death in men in Europe. This is an important health problem, especially in developed countries, with a higher proportion of elderly men in the general population. In Spain, the estimated incidence and mortality of prostate cancer according to Globocan (year 2012) is 27,853 / 100,000 inhabitants (122,1 new cases / 100,000 / year) and 5,481 deaths per year (24 deaths / 100,000 / year)2.
Current therapeutic practice considers hormone therapy as first line treatment, with androgen suppression by luteinizing hormone-releasing hormone (LHRH) agonists or orchiectomy, or complete androgen blockade (LHRH agonist combined with an anti-androgen), both in high-risk clinically localized PC as in localized prostate advanced or metastatic cases. Combination therapy with radiotherapy was consistently associated with a specific improvement in the disease and Overall Survival (OS), compared with single-mode treatment, in Phase III randomized studies3.
LHRH analogues (leuprorelin, goserelin and triptorelin) act by reducing the amount of testosterone produced by testes. Treatment with these drugs is also called chemical castration or medical castration. In order to avoid the initial flare effect when these are first administered, antiandrogen therapy with flutamide or bicalutamide should preceded or co administered with LHRH agonist, for at least 7 days.
LHRH antagonists, such as degarelix, rapidly and directly inhibit the release of androgens, unlike LHRH agonists without causing exacerbation. Therefore, they do not require coadministration with an antiandrogen. This therapy is only used in the case of patients with short-term complications.
The combination of radiotherapy with brachytherapy, with or without ADT, during two or three years, is another primary treatment option for high-risk or locally advanced patients. However, the optimal duration of ADT in this setting remains unclear. Radical prostatectomy plus pelvic lymph node dissection (PLND is remains as an option in high-risk patients who may benefit from surgery, and for selected patients with locally advanced disease, with no fixation to adjacent organs.
If the first hormone line fails, a second hormone treatment may sometimes be attempted: if androgen suppression loses efficacy, an antiandrogen may be added to the therapy, or if complete androgen blockade fails, the antiandrogen will be removed.
If this second hormonal line also fails, it is said that the disease is progressing to a condition known as Castration-Resistant Prostate Carcinoma (CRPC). The definition of CRPC4 includes castrate serum testosterone < 50 ng/dL and biochemical or radiological progression:
- Biochemical progression: Increased levels of Prostate Specific Antigen (PSA) in 3 measurements with at least one week between them, with 2 elevations > 50% regarding the nadir and PSA > 2 ng/mL;
- Radiological progression: either two or more new bone lesions on bone scan or a soft tissue lesion using RECIST cirteria 1.1.
New evidence has been recently published about the use of docetaxel, a drug whose patent has expired; these seem to show benefits in the setting of hormone-sensitive PC patients. Docetaxel has not been approved for use in this indication5. Docetaxel in combination with prednisone or prednisolone is indicated for the treatment of patients with hormone-refractory metastatic PC. Therefore, the use of docetaxel in this new scenario would be off-label, according to Royal Decree 1015/2009 on use of drugs in special situations. It requires an Informed Consent of the patient, which must be recorded in the medical history6.
Just like the introduction of docetaxel represented a before-and-after in CRPC treatment the introduction into the sensitive hormone patient should be considered as an important therapeutic advance in CP. For this reason, from the GENESIS-SEFH group, an evaluation report was carried out according to the MADRE methodology, which analyzed the current situation.
Efficacy
The studies analyzed for this evaluation were the CHAARTED7 and STAMPEDE8 clinical trials, because these provide the most mature data with the highest number of patients; as well as a meta-analysis9, including patients with M0 and M1 stages.
CHAARTED is a multicenter, open label, phase III trial, in which 790 patients recently diagnosed with metastatic PC were randomized to receive either 6 cycles of docetaxel plus ADT or ADT alone. The primary objective of the study was to determine whether docetaxel therapy at the beginning of ADT for metastatic hormone-sensitive prostate cancer would result in longer overall survival (OS) than that with ADT alone, and some of the secondary endpoints determined were: the time until castration-resistant disease, time to clinical progression, time to serological progression, PSA response measured at 6 and 12 months, and quality of life measured through the FACT-P Questionnaire (Functional Assessment of Cancer Therapy-Prostate).
Metastatic disease was defined as high-volume if the patient presented visceral metastasis or ≥ 4 bone lesions with ≥ 1 beyond the vertebral bodies and pelvis.
STAMPEDE is a multicenter, phase III, open clinical trial, randomized 2:1:1:1, where patients received, respectively, ADT (control arm), ADT + zoledronic acid, ADT + docetaxel, or ADT + docetaxel + zoledronic acid. The study included 2,962 patients recently diagnosed with PC, either metastatic, positive node (N +) or high-risk locally advanced; the latter should meet at least two of the following criteria: T3/4 stage, Gleason pattern 8-10, or PSA levels ≥ 40 ng/ml. The primary endpoint of the study was to determine OS and PFS (Progression Free Survival). The secondary endpoints determined were skeleton-related events, and quality of life measured through the questionnaire by the EORTC QLQ-C30 (European Organization for Research and Treatment of Cancer, quality life questionnaire).
Table 1 shows the results of the CHAARTED and STAMPEDE clinical trials.
Table 1 Outcomes of the CHAARTED and STAMPEDE clinical trials
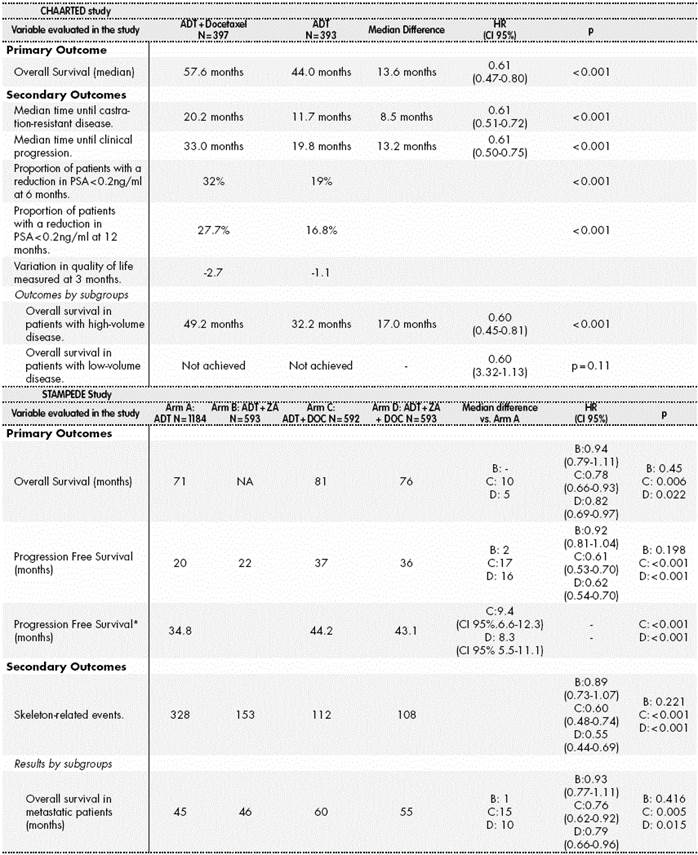
ADT: Androgen Deprivation Therapy; ZA: zoledronic acid; DOC: docetaxel.
*A model was used to calculate the trimmed mean adjusted to the first 84 months of Progression Free Survival, because important evidence was observed regarding lack of proportionality along the curve with the use of Hazard Ratio.
A meta-analysis was also included in the evaluation; its objective was to see the effect of adding on docetaxel and biphosphonates to ADT in terms of OS and PFS. Seven controlled and randomized clinical trials were included, with high-risk localized or metastatic hormone-sensitive PC patients, where docetaxel + standard care therapy (hormone therapy + /-radiotherapy) was compared with standard therapy; 3 of the studies included patients in M1 stage (STAMPEDE, GETUG-15 and CHAARTED) and 4 studies were conducted in patients in M0 stage (STAMPEDE, GETUG-12, TAX 3501 and RTOG 0521). In total, 3,206 patients in Stage M1 and 3,978 patients in M0 stage were included.
Table 2 shows the main results of the meta-analysis.
Table 2 Outcomes of the Meta-Analysis
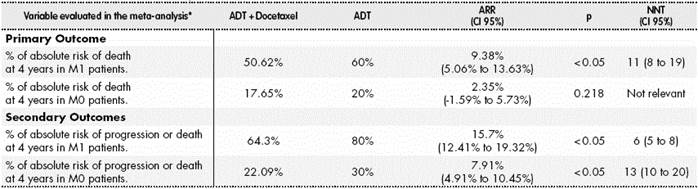
ADT: Androgen Deprivation Therapy; ARR: Absolute Risk Reduction; NNT: Necessary numbers of patients to treat in order to obtain an additional efficacy unit.
*Because no differences between medians can be obtained, a 40% survival at 4 years is assumed in the group treated with ADT monotherapy. In M0 patients, an 80% is assumed. The same assumption is made for Progression Free Survival: 20% in M1 patients and 70% in M0 patients.
Safety
Adverse events related to docetaxel are widely known and described in literature. The most common adverse events are: neutropenia, anemia, thrombocytopenia, peripheral sensory neuropathy, motor peripheral neuropathy, febrile neutropenia, gastrointestinal reactions (fatigue, diarrhoea, nausea and vomiting), alopecia, and skin reactions. Among the most severe: Grade 4 hypersensitivity reactions, febrile neutropenia, and liver toxicity.
In the CHAARTED study, the proportion of Grade 3-5 events in the experimental arm was 29% vs. 3% in the control arm. The most frequent events were: neutropenia (12%), febrile neutropenia (6%), fatigue (4%), leukopenia (5%), lymphopenia (2%), allergic reaction (2%), anaemia (1%) and diarrhoea (1%). The protocol description specifies that adverse events among the patients in the control arm must not be routinely documented, and this might explain such a low proportion.
In the STAMPEDE study, the proportion of Grade 3-5 events in the experimental arm treated with ADT + docetaxel was 52% vs. 32% in the control arm. The most frequent events were: febrile neutropenia (15%), neutropenia (12%), endocrine alterations (10%), gastrointestinal alterations including diarrhoea, abdominal pain, constipation and vomiting (8%), general alterations such as asthenia and fever (7%) and musculoskeletal alterations (6%).
There was one death in the CHAARTED study and eight deaths in the STAMPEDE study (one due to neutropenic sepsis in the ADT + docetaxel arm, and seven in the ADT + docetaxel + zoledronic acid arm: 4 of them due to neutropenic sepsis, 1 due to pneumocystis pneumonia, 1 due to interstitial pneumonitis, and 1 due to pneumonia); these could have been associated with docetaxel.
Approximately 86% of patients in the CHAARTED study intervention group (N = 397) received the complete treatment. Of this proportion, approximately 74% received the established courses of treatment without any dose reduction. In the STAMPEDE study, 77% in the ADT + docetaxel arm N = 592) and 71% in the ADT + docetaxel + zoledronic acid (N = 593) completed the 6-course administration.
The differences in safety outcomes between both studies could be caused by the differences in the population included in each one10. There was no recommendation about how to use the colony-stimulating factors in either of the protocols for the two studies.
It must be taken into account that treatment with docetaxel could involve a reduction in patients’ quality of life; this aspect is a major key element to be considered at the time of deciding on treatment approach. According to the data in the FACT-P Quality of Life questionnaire, collected in the CHAARTED study, docetaxel leads to a higher worsening in quality of life, measured at 3 months since treatment initiation. Even though the measurement was conducted at 12 months, it showed better outcomes than the control arm, and patients treated with docetaxel could even return to their baseline status11.
Economic area
For the economic analysis, only those costs derived of the price of docetaxel were taken into account, because treatment with docetaxel is also added to standard treatment (hormonal therapy). The associated indirect costs were not considered for the economic evaluation, such as those derived from adverse events occurred, or analytical and/or radiological tests for chemotherapy follow-up. For the calculation of cost per day in those medications with dose adjusted by weight, a standard patient of 70 kg was taken as reference. For those medications with dose adjusted per body surface, the reference was a standard patient with 1.7 m2. The incremental cost compared to the reference therapy would be € 3,196.98.
According to the data from the CHAARTED and STAMPEDE studies and the cost of treatment, the estimated additional cost per year of life gained (LYG) is compatible with a estimated incremental cost effectiveness ratio (ICER) between € 2,267.36 and € 3,851.78. Therefore, this would be an efficient therapy (See Table 3).
Table 3 Incremental Cost-Effectiveness
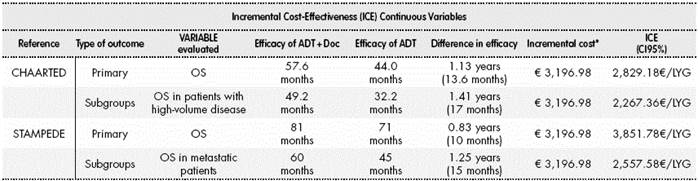
ADT: Androgen Deprivation Therapy; OS: Overall Survival. LYG: year of life gained.
*Indirect associated costs were not taken into account, such as those derived of adverse events occurred or lab and/or radiological tests for chemotherapy follow-up.
According to the National Statistics Institute, the Spanish male population (from 18 to 110-year old) was of 22,813.635 inhabitants by July, 2016.
The incidence of PC in Spain is of 122 cases per 100,000 inhabitants. If we consider that approximately 5% of de novo diagnoses are metastatic, the number of patients adequate for receiving docetaxel at national level, according to the CHAARTED criteria, could be estimated in 1,392 patients. Taking into account the incremental cost of € 3,196.98 vs. ADT in monotherapy, the impact in Spain would be of € 4,449,018.82.
Additional considerations
The comparator is adequate, and the primary endpoint in both studies is the variable with maximum clinical relevance: OS. The difference in OS between both studies could be caused by the proportion of non-metastatic patients included in the STAMPEDE study, who therefore had better disease prognosis. In both studies, there are statistically significant differences in the secondary variables with higher interest, such as PFS or Time until Clinical Progression. This is shown even though patients included would have access to docetaxel subsequently, which indicates the clear benefit of bringing forward the use of the drug to this first line with ADT.
There is doubt regarding patients without metastasis. The CHAARTED study does not include them, unlike the STAMPEDE study, where they represent approximately 39% of the total patients included. However, outcomes are not confirmed in these patients, although it is a fact that there is no significant interaction either.
Regarding the analysis by sub-groups, the CHAARTED study presents consistency in all subgroups; however, there is a dubious interaction in PS (ECOG). But with a dubious interaction “p”, even though it cannot be ruled out, in a pre-specified analysis, with biological plausibility but inconsistency in the outcomes obtained by the STAMPEDE study, evidence does not seem to be enough in order to consider that patients with PS (ECOG) = 0 will benefit less than those with PS (ECOG) = 1-2.
On the other hand, the STAMPEDE study presents consistency in all sub-groups, except in one of the analyses, in patients with recurrent disease in the arm treated with ADT + docetaxel + zoledronic acid, with a significant “p” for interaction. Given that CI95% is extremely wide due to the low number of patients, and that in the arm treated with ADT + docetaxel there is consistency between recurrent or non-recurrent disease, we cannot confirm in general that patients with recurrent disease will benefit of the therapy.
It is interesting to note that the score in the Gleason Scale does not show interaction in any of the studies. That is to say, the increase in survival would appear both in patients with a Gleason Score ≥ 8 and in patients with a lower score.
It is also worth highlighting in the CHAARTED study that the 95% Confidence Intervals between subgroups with high or low volume disease are completely overlapping, and we can reach the conclusion that there is no evidence about any different performance. We could say that no interaction was found, and therefore, the value assigned to the low-volume subgroup cannot be other than the overall value of the study, because there is no evidence otherwise12.
Meta-analysis confirms the overall survival benefit in metastatic patients. In non-metastatic patients, we do not have evidence to suggest that the result is different, since there is no interaction in the STAMPEDE subgroup analysis, and the meta-analysis does not obtain a sufficient sample to affirm or discard the benefit.
Conclusion: Therapeutic Positioning and Conditions for Use
In view of the efficacy and safety results, the proposed positioning is considered Category D-1: It is included in the Pharmacy Formulary with specific recommendations: to advance the use of docetaxel added to androgen deprivation therapy to first-line metastatic hormone-sensitive prostate cancer, regardless of metastatic volume, in those patients who meet the CHAARTED study criteria:
PC diagnosis by Pathology, with radiological evidence of metastasis, in the defined setting either for high or low volume disease*.
Patients with ECOG 0, 1 or 2 with an adequate organic function to receive chemotherapy.
No peripheral neuropathy > grade 1 or history of hypersensitivity to docetaxel.
No history of heart disease such as myocardial infarction in the last 6 months, active angina, or congestive heart failure.
• Patients previously treated with ADT during ≤ 24 months, with progression 12 months after treatment completion may benefit from docetaxel therapy.
Patients on treatment with ADT for metastatic disease, who have initiated treatment during the last four months, and without evidence of progression, could also benefit of the addition of docetaxel.
*To deprive patients without high volume of the important survival benefit does not seem compatible with a correct interpretation of the subgroup analysis from the CHAARTED study.
It would be an off-label use, because docetaxel is indicated in metastatic “hormone refractory” PC. Therefore, informed consent will be mandatory, and this must be entered in the clinical record, according to article 13.1 of Royal Decree 1015/2009 of June, 19th, which regulates the availability of drugs in special situations.











 texto em
texto em 


