INTRODUCTION
Asthma is a common chronic inflammatory respiratory disease (1), with high morbidity (2) and mortality. Studies have shown (3) that the proportion of children aged 13-14 and children aged 6-7 suffering from asthma increases by 0.28 % and 0.18 % annually. Due to the high incidence of asthma and the great economic pressure to treat asthma, it has attracted more and more attention from all walks of life in the past (4). The etiology and pathogenesis of asthma have not been fully elucidated (5), which may be related to various factors such as genetics, bacteria, viruses, immunity, nutrition, and environment. Asthma is mainly treated by inhaled corticosteroids, long-acting β-receptor agonists, leukotriene antagonists, and other drugs (6). Recently, the efficacy of probiotics in allergic diseases has received special attention (7). Experiments have shown that probiotics have a clear effect on allergic diseases such as allergic rhinitis (8,9) and eczema (10). However, the current meta-analysis showed that Lactobacillus supplementation had a positive effect on asthma prevention (11), while other probiotics had no significant effect on asthma prevention (12) and treatment (13). This is inconsistent with the conclusions of some experiments (14,15). In this study, meta-analysis was used to study the efficacy of probiotics in the treatment of asthma and evaluate it, so as to provide a reference for the selection of treatment options for asthma patients.
METHOD
This study followed the Cochrane manual system evaluation and meta-analysis criteria, according to Prisma statement, clinical registration number: INP LA SY 202270076.
SEARCH STRATEGY
We searched PubMed, Embase, The Cochrane Library and Web of Science databases to collect randomized controlled trials that met the inclusion criteria until July 2022. References for the included studies were also searched to supplement access to relevant information.
STUDY SELECTION
Inclusion criteria were as follows: a) the study is a randomized controlled trial; b) the inclusion of subjects is not limited by age, gender, etiology, or ethnic group; c) asthma diagnosis is consistent with the Global Asthma Initiative (1); d) there was no significant difference in age, gender, course of disease among the groups, and they were comparable; e) the experimental group was treated with probiotics (unlimited strains, doses, and courses of treatment), and the control group was treated with placebo; e) the experiment uses one or more fractional exhaled nitric oxide (FeNO), forced expiratory volume in the first second (FEV1), FEV1/FVC (%), asthma symptom severity, Childhood Asthma Control Test (CACT), Asthma Control Test (ACT), and the number of exacerbations to evaluate the experimental results. Higher FeNO indicates more severe airway inflammation. FEV1, FEV1/FVC (%) correlated with lung function. CACT- ACT indicates the degree of asthma control in the form of a scale. In this study, the assessment of asthma severity using a rating scale was summarized as asthma symptom severity.
Exclusion criteria were: 1) diseases with liver, gastrointestinal, kidney, endocrine, neuronal, cardiovascular, or psychiatric disorders or malignant tumors that may affect the results of the active upper respiratory tract infection study; b) conference papers, reviews, case reports, summaries of experiences, and repeated literature; c) the information contained in the literature is incomplete and cannot be obtained through other information; and d) low quality of literature (Cochrane Handbook < 2).
ASSESSMENT OF RISK OF BIAS
Two commentators independently analyzed the included literature according to the Cochrane bias risk assessment criteria, and the inconsistencies were reached through discussion. The evaluation contents include: a) the generation of the random allocation scheme; b) the concealment of the allocation scheme; c) the implementation of the blind method; d) the integrity of result data; e) non-selective report of results; and f) other biases. “Low risk” means low risk of bias, “high risk” means high risk of bias, and “unclear risk” means that literature does not provide sufficient or certain information for bias assessment.
LITERATURE SCREENING AND DATA EXTRACTION
Two researchers independently screened literature, extracted data and cross-checked them. If there were differences, they were solved through discussion or consultation with a third party. In literature screening, we first read the topic, and after excluding the obviously irrelevant literature, we further read the summary and full text to determine whether it was included. The author of the original study was contacted by email or telephone, if necessary, to obtain undetermined but important information for this study. Data extraction included: research topics, first author, publication year, age, gender, course of disease, follow-up time, intervention measures, outcome indicators.
STATISTICAL ANALYSIS
Statistical analysis was performed using RevMan 5.4 software. For the enumeration data, relative risk (RR) and 95 % confidence interval (95 % CI) were used as efficacy analysis statistics. When there was statistical homogeneity among the studies (p > 0.1, I2 < 50 %), a fixed effect model was used for meta-analysis; if there was significant heterogeneity among the studies (p < 0.1, I2 > 50 %), the source of heterogeneity was further analyzed, and a subgroup analysis on factors that may lead to heterogeneity was performed. A random effects model was used for analysis. The funnel plot was used to judge whether there was publication bias in the included literature, and Egger's test could be used when there were at least ten studies. Inspection level was set at α = 0.05.
RESULTS
SELECTION OF STUDIES
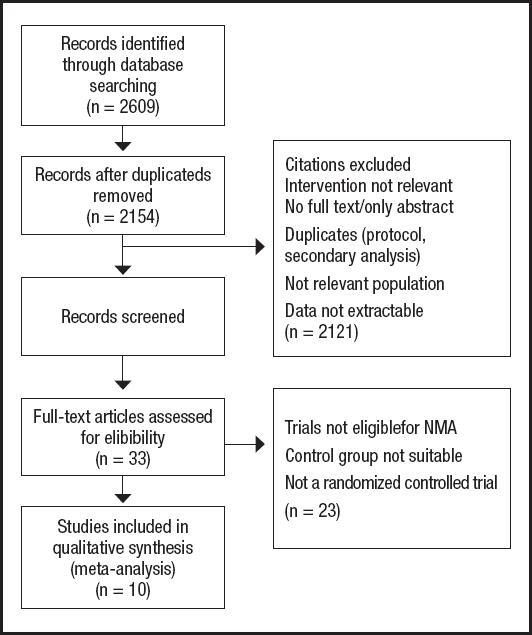
A total of 2,609 related pieces of literature were obtained from the literature screening process and preliminary examination of results, of which 455 were repetitive publications and 2,121 articles were excluded due to their irrelevant titles and abstracts. After layer-by-layer screening, 33 articles were selected for full-text review, 23 articles assessed as unqualified were excluded, and ten articles were finally included (16-18,21-26), including 1,101 patients. The search and selection steps are shown in figure 1.
The ten articles finally included were all randomized controlled studies from SCI journals. Among them, two articles studied the relationship between probiotics and FeNO, three articles studied the relationship between probiotics and FEV1, four articles studied the relationship between probiotics and FEV1/FVC (%), two articles studied the relationship between probiotics and asthma symptom severity, four articles studied the relationship between probiotics and CACT-ACT, and two articles studied the relationship between probiotics and the number of exacerbations. These ten studies were conducted in different countries, and the types and quantities of probiotics were also different. Table I summarizes the characteristics of each included study.
Table I. Characteristics of included studies.
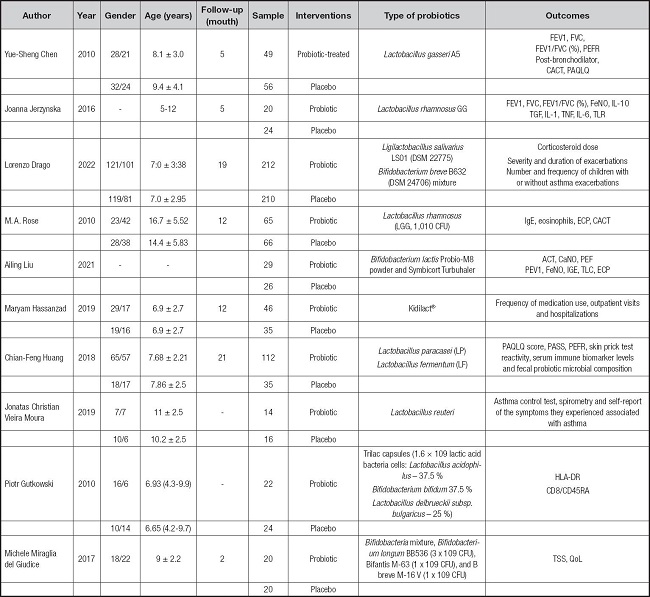
FEV1: forced expiratory volume in the first second; FVC: forced expiratory volume; PEFRs: peak expiratory flow rates; CACT: Childhood Asthma Control Test; FeNO: fractional exhaled nitric oxide; CACT: Childhood Asthma Control Test; PASS: Pediatric Asthma Severity Scores; PAQLQ: Pediatric Asthma Quality of Life Questionnaire; TSS: total symptom score; IL: interleukin; TGF: transforming growth factor; TNF: tumor necrosis factor; TLR: toll-like receptors; ECP: eosinophil cationic protein; HLA: human leukocyte antigen; QoL: quality of life; CFU: colony forming units.
ASSESSMENT OF RISK OF BIAS
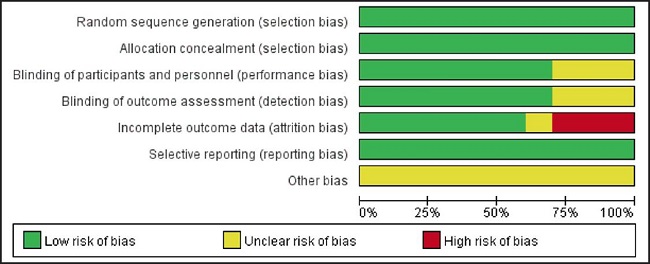
The bias risk of included studies was assessed according to the Cochrane manual. The results showed that the research quality of all included randomized controlled trials (RCTs) was high, and the risk bias was mainly due to the midway introduction of research by some subjects. The results of the bias risk assessment included in the study are shown in figures 2 and 3.
META-ANALYSIS
FeNO
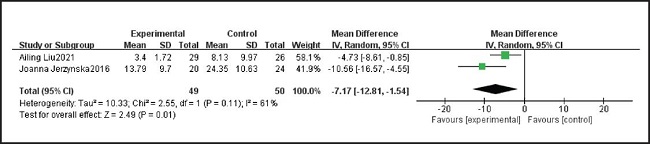
Two studies including 99 patients reported FeNO in patients taking probiotics and placebo. We tested the heterogeneity of the two studies, and the results showed p = 0.11 and I2 = 61 %, indicating that heterogeneity is high. Therefore, the random effect model was used. After summarizing the data, we found that probiotics were lower than placebo patients, and the difference was statistically significant (MD = -7.17, 95 % CI: -12.81, -1.54). The results are summarized in figure 4.
Asthma symptom severity
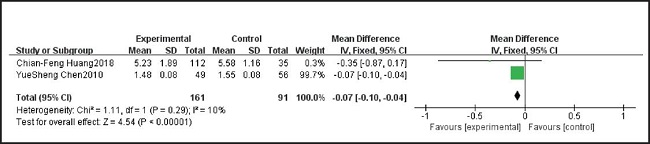
Two studies including 252 samples reported asthma symptom severity in patients taking probiotics and placebo. We tested for heterogeneity between two studies which showed p = 0.29 and I2 = 10 %, indicating very low heterogeneity; therefore, a fixed effects model was used. After pooling the data, we found asthma symptom severity in patients taking probiotics
It was lower than in the placebo patients, and the difference was statistically significant (MD = -0.07, 95 % CI: -0.10, -0.04). The results are summarized in figure 5.
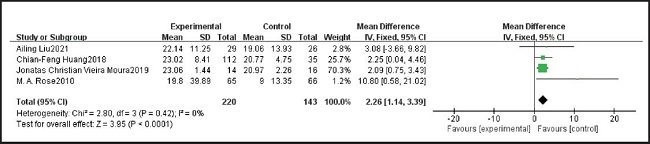
CACT-ACT
Four studies including 343 samples reported CACT-ACT of patients taking probiotics and placebo. We tested the heterogeneity of the four studies, and the results showed p = 0.42 and I2 = 0 %, indicating that heterogeneity was low. Therefore, we used the fixed effect model. After summarizing the data, we found that the CACT-ACT of patients taking probiotics was higher than that of patients taking placebo, and the difference was statistically significant (MD = 2.26, 95 % CI: 1.14, 3.39). The results are summarized in figure 6.
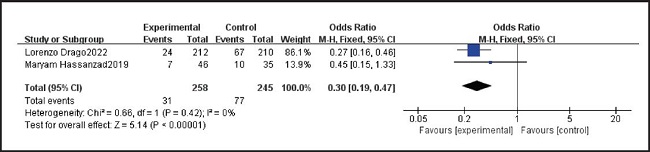
The number of acute episodes of asthma
Two studies, including 503 samples, reported the number of acute episodes in patients taking probiotics and placebos. We tested the heterogeneity of the two studies, and the results showed p = 0.42 and I2 = 0 %, indicating that heterogeneity was low. Therefore, the fixed effect model was used. After summarizing the data, we found that the number of acute episodes in patients with probiotics was lower than that in patients with placebo, and the difference was statistically significant (OR = 0.30, 95 % CI: 0.19, 0.47). The results are summarized in figure 7.
Lung function-related indicators
Three studies, including 179 patients, reported FEV1 in patients taking probiotics and placebos. We tested the heterogeneity of the three studies, and the results showed p = 0.05 and I2 = 66 %, indicating high heterogeneity. Therefore, the random effect model was used. After summarizing the data, we found that there was no statistically significant difference in FEV1 between probiotics and placebo (MD = 0.11, 95 % CI: -0.05, 0.26). The results are summarized in figure 8.
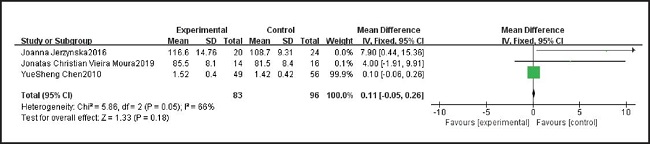
Figure 8. Comparison of forced expiratory volume in the first second (FEV1) results between probiotics and control group.
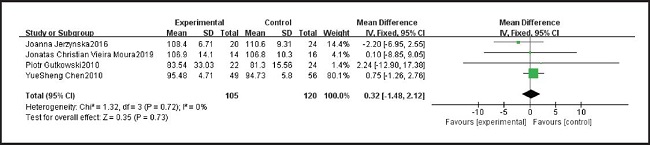
Four studies, including 125 patients, reported FEV1/FVC (%) in patients taking probiotics and placebos. We tested heterogeneity of the four studies, and the results showed p = 0.68 and I2 = 0 %, indicating that heterogeneity was low. Therefore, we used the fixed effect model. After summarizing the data, we found no significant difference in FEV1 between probiotics and placebo (MD = 0.32, 95 % CI: -1.48, 2.12). The results are summarized in figure 9.
TABLE BIAS AND SENSITIVITY ANALYSIS
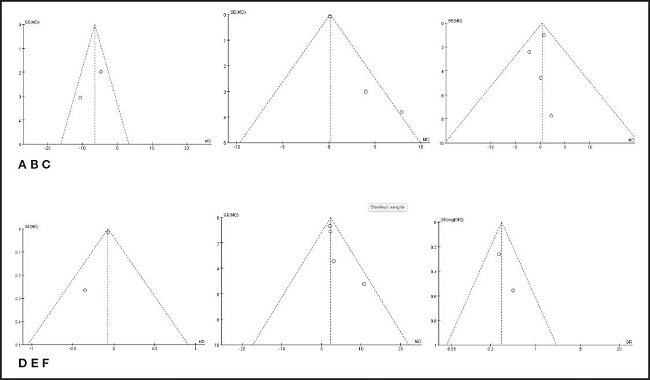
The funnel plot in figure 10 is basically symmetrical, indicating that there is no potential publication bias and the reliability of the research results is high.
DISCUSSION
In this study, a total of ten RCTs in SCI journals were included for systematic evaluation and meta-analysis. The results showed that probiotics can improve symptoms and airway inflammation in patients with asthma, reduce acute exacerbation of asthma, and have no significant improvement in lung function. This is different from the previous meta-analysis (13).
In our study, we found that the clinical symptoms of asthma patients improved after using probiotics (MD = -0.07, 95 % CI: -0.10, -0.04), and CACT-ACT score increased (MD = 2.26, 95 % CI: 1.14, 3.39). A study (22) showed no significant increase in IgE and IL-12 production in probiotics-treated subjects. This suggests that probiotics may not play a role through IgE, and it may control asthma through the intestinal-pulmonary axis, that is, probiotics enter the gastrointestinal tract to produce corresponding immune cells and cytokines, and lactic acid bacteria metabolites directly migrate from the intestine to the respiratory tract through circulation to produce corresponding effects. However, this is not completely consistent with the results of related experiments (27,28). Some experiments (2) have also proved that probiotics treat allergic asthma inflammation and pneumonia induced by OVA-LPS (ovalbumin-lipopolysaccharid) by regulating TLR4/NF-kB signaling pathways. There are some differences and contradictions in the existing research results. Therefore, the mechanism of probiotics affecting asthma needs further research and clarification (29,30).
FeNO reflects the level of airway inflammation in patients with asthma (31,32). In our study, FeNO in the experimental group was lower than that in the control group (MD = -7.17, 95 % CI: -12.81, -1.54). This indicates that the use of probiotics can control airway inflammation of asthma to a certain extent. This is consistent with the result of Kukkonen (19). At the same time, we found that FEV1 (MD = 0.11, 95 % CI: -0.05, 0.26) and FEV1/FVC (%) (MD = 0.32, 95 % CI: -1.48, 2.12) in patients with asthma using probiotics were not significantly different from those in the control group. It is worth noting that the two included studies (17,25) pointed out that probiotics could improve FEV1 in asthma patients, and Michele (20) found that taking probiotics and vitamin D3 simultaneously could also significantly reduce FeNO (p < 0.01). In another study (16), although there was a significant difference in FEV1 between the experimental group and the control group, the difference was statistically significant before and after the study (p = 0.035). When we did not incorporate the latter data into meta-analysis, the FEV1 results of the experimental group and the control group (MD = 5.50, 95 % CI: 0.87, 10.14) were statistically significant (p = 0.02). The number of studies is the main reason for this phenomenon. Although there was no statistical difference in the effect of probiotics on lung function of patients based on the existing data, this result may change with the increase of high-quality randomized controlled trials.
This study showed that the number of acute episodes in patients with asthma after using probiotics was significantly reduced (OR = 0.30, 95 % CI: 0.19, 0.47). Jonatas (25) pointed out that the improvement of asthma symptoms in patients treated with probiotics was mainly concentrated in Wheezing (p = 0.046), and there was no statistical difference in cough, tiredness, chest pain, nighttime symptoms, and absence from school. Lorenzo Drago (18) found that the frequency of acute exacerbations, severity and the number of times and doses needed to use drugs in patients with probiotics were lower than those in the control group, and there was statistical difference.
In this included literature, no major or minor adverse reactions occurred in all patients. The adverse reactions caused by probiotics are septicemia, bacteremia and gastrointestinal ischemia (33,34). In general, severe patients, severe infants, postoperative and hospitalized patients and patients with low immune function have more adverse reactions. Overall, however, the safety of probiotics in the treatment of asthma is worth ensuring (35).
Due to the limited number of included studies, no subgroup analysis was conducted. Therefore, the results may be affected by clinical heterogeneity. Studies have found that Lactobacillus have a certain preventive effect on asthma, while other probiotics have no effect (11). The duration of intervention, the standard of acute exacerbation of asthma, and patient age may affect the results. At the same time, relatively small sample size limits the accuracy of our analysis.