Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO  Similares en Google
Similares en Google
Compartir
Revista Española de Enfermedades Digestivas
versión impresa ISSN 1130-0108
Rev. esp. enferm. dig. vol.96 no.11 Madrid nov. 2004
| ORIGINAL PAPERS |
Postoperative radiotherapy-induced morbidity in rectal cancer
M. Garay Burdeos, M. García-Botella, V. Viciano Pascual, M. D. Torregrosa Macías1, J. Aguiló Lucia, M. Ata,
M. C. Muñoz Alonso and J. García del Caño
Department of General and Digestive Surgery. 1Oncology Section (Department of Internal Medicine). Lluís Alcanyís
Hospital. Xàtiva, Valencia. Spain
ABSTRACT
Objectives: we analyzed long-term morbidity and bowel function alteration after postoperative radiotherapy for rectal cancer following resection with anastomosis.
Patients and methods: thirty-seven patients who underwent surgery with intention to cure and a minimal follow-up period of 3 years were included. These patients were divided into two groups: in the first group, 14 patients received postoperative chemo-radiotherapy, 5-fluorouracil plus folinic acid, and 45 Gy plus 5 Gy boost. In the second group, there were 23 patients regarded as controls. We designed a questionnaire about their bowel function and analyzed the morbidity detected in their follow-up.
Results: the group that was treated with postoperative chemo-radiotherapy had more daily bowel movements (p = 0.03) and night-time movements (p = 0.04); incontinence (69.2 versus 17.4% in the control group; p = 0.002), and perianal skin irritation (p = 0.04) versus the control group. Although without meaningful differences, the group under treatment wore a pad more often, had more defecatory urgency, could distinguish worse gas from stool, and needed more frequently antidiarrheal measures. Major complications were present in 28.6% of the under-treatment group: three intestinal resections were performed due to actinic stenosis; one patient had a residual stercoral fistula; another had several occlusive crises solved with medical treatment.
Conclusions: postoperative chemo-radiotherapy had a high morbidity rate, which determined a significant alteration in quality of life. Accuracy in indication is therefore necessary, as well as a consideration of other alternatives for treatment, such as preoperative chemo-radiotherapy and total mesorectal excision.
Key words: Rectal cancer. Chemo-radiotherapy. Morbidity.
Garay Burdeos M, García-Botella M, Viciano Pascual V, Torregrosa Macías MD, Aguiloacute; Lucia J, Ata M, Muñoz Alonso MC, García del Caño J. Postoperative radiotherapy-induced morbidity in rectal cancer. Rev Esp Enferm Dig 2004; 96: 765-772.
Recibido: 12-11-03.
Aceptado: 09-03-04.
Correspondencia: Miguel Garay Burdeos. Servicio de Cirugía Oral y Digestiva. Hospital Lluis Alcanyís. Carretera Xàtiva-Silla, km. 2. 46800 Xàtiva. Valencia. Tel.: 962 289 563. e-mail: miguelgaray59@hotmail.com
INTRODUCTION
The surgical treatment of rectal cancer is burdened with a high percentage of local recurrence (1). It varies from 20 to 70% according to tumor stage. In the last few years a great effort has been made in order to decrease disease progression, and the management of rectal cancer has substantially changed due to advances in surgical techniques, changes in radiotherapy technology, and the emerging role of some chemotherapeutic agents such as oxaliplatin. All this has meant continuous changes in standard therapeutic protocols (2).
Several studies have proved that chemo-radiotherapy, either pre- or post-operative, improves survival time and decreases local recurrence (2-6). Therefore, radiotherapy (RT) is recommended as a part of rectal cancer treatment since 1990 (7-11). At the same time, surgical modifications, such as total mesorectal excision (TME), have succeeded in decreasing local recurrence below 10% even in high-risk patients (12,13).
Nevertheless, radiotherapy is not free from side effects, which may have a great impact on the patient's quality of life (14). Short-term morbidity and mortality have been analyzed in several studies, but studies about long-term morbidity are rare (1,14-16).
Difficulties for such a post-radiotherapy morbidity study may include various factors: concern regarding metastatic disease or local recurrence, which may lead to underestimate local alterations associated with radiotherapy; various chemotherapy protocols (combined or not with radiotherapy), and the different radiation techniques used-dose fractionation and limited-field radiation (14,17).
The objective of this study is to assess the long-term side effects of postoperative radiotherapy and its impact on anorectal function.
PATIENTS AND METHODS
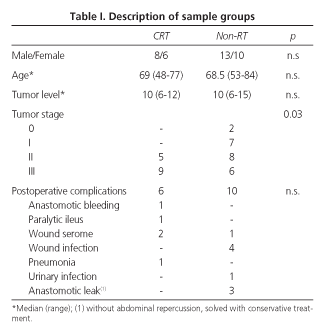
Clinical records of patients who underwent surgery for rectal cancer in the Department of Surgery at the Hospital "Lluís Alcanyís" during the period 1993-1998 were retrospectively reviewed. There was a group of 68 patients who fulfilled the following criteria: tumor location was below 15 cm from the anal margin, there was no invasion of adjacent organs, and all of them had been operated using anterior resection of the rectum with primary anastomosis and no diverting stoma. Nine patients of this group were excluded due to metastatic disease at the time of surgery; 13 died because of disease progression; 2 of them needed a temporary diversion due to an anastomotic leak, as this complication itself could alter bowel function; 1 patient happened to have a non-resectable gastric cancer, and 6 patients could not be contacted. The remaining 37 patients were included in this study and were divided up in two groups: the first one included those 14 subjects who had been treated with postoperative chemo-radiotherapy (CRT); the other group, which was considered the control group, included the remaining 23 who did not receive radiotherapy. In the control group there were two patients whose stage was 0, as they suffered from big, degenerated villous tumors (Tis) that required an anterior resection.
The treatment schedule of those 14 patients in the CRT group was as follows: two chemotherapy cycles with 5-fluorouracil (425 mg/m2) plus folinic acid (20 mg/m2), followed by radiotherapy plus 5-fluorouracil concurrently for the first and last three days, reaching a total dose of 45 Gy, and adding at the end a presacral boost of 5, 4 or 9 Gy in relation to their body volume. Treatment was completed with two more chemotherapy cycles with 5-fluorouracil and folinic acid.
Radiotherapy was delivered through a high-energy linear accelerator. Radiation technique was isocentral with
4 fields into the pelvis and lateral fields in the presacral boost, a fractionation of 180 cGy per each session until the full dose was reached. Radiation in the prone position and astringent diet were used as protective measures to minimize the toxicity of radiotherapy.
Along the year 2002, all patients included in this study were interviewed in order to assess their bowel function, and all their clinical records were revised to detect all possible major complications suffered by these patients after discharge from hospital. In this way we assured a minimal follow-up of 3 years after completing the treatment of rectal cancer. The interview gathered: number of daily bowel movements and how many at night time, consistency of stool (liquid, semisolid, or solid), incontinence and its frequency (occasional le; once per week; frequent gt; once per week), soiling, need to wear a pad, ability to distinguish between flatus and stool, urgency (if they were able to defer defecation for more than 15 minutes), tenesmus, perianal skin irritation, bleeding, mucorrhea, and need for antidiarrheal measures (diet, medication). Quality of life questionnaires specifically made for colorectal cancer, as the QLQ-CR 38 developed by EORTC, were not used since at the time of this study they had not been fully adapted and validated regarding Spanish cultural features (18).
Follow-up and anorectal function complications detected were analyzed according to the answers in the written interview. Chi-square testing was used for percentage comparisons between both groups.
RESULTS
We did not find any significant differences between both groups regarding sample characteristics -age, sex, and tumor level- except for tumor stage (Table I). We also could not find any noteworthy differences in the postoperative complications of both groups. Colorectal anastomosis bleeding in the CRT group was solved by endoscopic coagulation. Three minimal anastomotic leaks in the control group (which had poor clinical manifestation and which were exteriorized by a drain) had a good outcome in a few days with conservative treatment (parenteral nutrition and antibiotics).
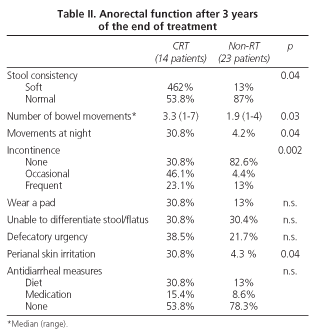
Survey results showed (Table II) that the CRT group had a mean (3.3) of bowel movements per day that was significantly higher (p = 0.03) than the control group (1.9). A higher percentage of CRT group patients, 30.8%, had bowel movements at night-time versus 4.2% in the control group (p = 0.04). There were also significant differences detected in stool consistency: 46.2% of patients who had received radiotherapy had softer stools, versus 13% in the control group.
In all, 69.2% of patients in the CRT group suffered from incontinence (most of them occasionally), though often episodes were also present in a high percentage. However, in the control group, incontinence was only present in 17.4% of patients and that was statistically significant (p = 0.002). We noticed, however without significant differences, that a high percentage of patients in the CRT group needed to wear a pad, while only 13% in the control group needed it. We did not find differences between both groups regarding their capacity to discriminate between flatus and stool.
Defecation urgency was relatively frequent in both groups, and we did not find significant differences between both groups. However, a high percentage (38.5%) of patients in the CRT group had problems to delay discharge more than 15 minutes, while only 21.7% had such urgency in the control group. Perianal irritation was more common in patients within the CRT group (p = 0.04).
Although an important number of patients had some type of anorectal dysfunction, only 31% of them needed some dietetic or pharmacologic measures, loperamide being the most widely used drug.
During follow-up, four patients (28.6%) in the CRT group suffered from major complications with recurrent obstructive episodes at the end of radiotherapy. Three of them needed surgery. An ileal resection was performed in two of them due to stricture caused by bowel radiation. They also needed dilatation of colorectal anastomotic strictures produced by radiotherapy. In one patient, a Hartmannacute;s procedure had to be performed because of an anastomotic fistula in a stiff and thickened rectum four months after radiotherapy completion. This patient still suffers from persistent intestinal fistula with sporadic fecal soilage from the rectal stump. None of the other patients in the CRT group and the control group has ever presented any other major complication during follow up.
DISCUSSION
Radiotherapy is regarded as another component of the therapeutical armamentarium for rectal cancer, its essential aim being to decrease local recurrence (2-11); it is even more effective when associated with chemotherapy. Chemo-radiotherapy has shown a remarkable decrease in recurrence and a better survival rate (6). However, several publications (1,3,5,15-17,19) have stated the adverse effects of radiotherapy, both pre- and post-operative.
Acute morbidity affects most patients in varying degrees, and manifests as gastrointestinal, hematological, and dermatologic disorders, as well as lumbosacral plexopathies (1,7,14,19). Although we had no mortality in our study, as in other studies (5), the literature describes (1,3,7) a small percentage of patients that suffer from severe complications due to sepsis and gastrointestinal problems. That places mortality rate secondary to radiotherapy in a level between 0.3 and 18%. Several authors attribute this considerable morbi-mortality to patient-related factors (age, diabetes and atherosclerotic disease), type of drugs used, and radiation technique.
However, radiation induces several disorders that produce late adverse effects (1,14-16), as intestinal obstruction, actinic enteritis, and proctitis, with figures ranging between 4 and 15%. This percentage was greater in our study (28%), probably because of sample size. Out of four patients with symptomatic actinic disorders, three had to be operated upon; however, they were not fully free from symptoms afterwards, because of adhesions and permanent rectal actinic changes. One patient even had a permanent rectal fistula to the abdominal wall in spite of going through a Hartman's operation that excluded intestinal transit.
Functional changes of the pelvic floor have not been properly documented (1,14-16,20), and there are few reports that analyze long-term functional changes. Most studies are related to survival, local relapse, and changes following surgical technique (14).
In accordance with other similar reports (1,15,16), we confirmed the toxicity of postoperative chemo-radiotherapy. It causes an important detrimental effect on bowel function, with softer stools, increased number of daily bowel movements (daytime and at night), incontinence, soiling, and perianal skin irritation. Other changes described (1,15), as the need to wear a pad, defecation urgency, and the use of antidiarrheal measures showed non-significant differences, although they were more frequent in the CRT group, probably also because of sample size (1,14,15,20). The causes of these functional changes are related to reduced rectal capacity and compliance, as well as to disorders in anorectal pressures.
The damage to anorectal function and the sequels of actinic lesions, together with the frequent need of medical or surgical treatments to improve these symptoms, may significantly modify the quality of life for such patients (1,14).
In the 1990s, a number of clinical trials were carried out with preoperative radiotherapy, and it was suggested that radiotherapy was better administered before surgical procedures (4,6,10), as it would lead to less side-effects and complications, as well as to a lower chance of small bowel damage.
However, since total mesorectal excission in rectal cancer has become standard, controversy has arisen on the need of radiotherapy (9,12,13,21,22), due to its low local relapse rate, similar to the rate obtained with preoperative chemo-radiotherapy. Heald et al. (12) revealed that TME diminishes local recurrence rates to 2% in 10 years" time after curative resections, which suggests that total mesorectal excission is the main determinant for outcome. Survival is 80 at 10 years in curative resections. Nevertheless, it has considerable postoperative complications, though not greater than those of other techniques, and as many as 17% anastomotic leaks. This is why a temporary ileostomy is recommended when the anastomosis is placed at less than 6 cm from the anal margin (12,23,24). Vascular infiltration, Dukes" stage, and circumferential margin involvement are the only significant predictors of local recurrence in Heald's experience.
Therefore, having postoperative RT morbidity and local recurrence rates with both TEM and CRT in mind, the pending question is whether the present-day treatment for rectal cancer is TME associated with preoperative chemo-radiotherapy or not (4,24,25). The answer to this question is still expected from the results of trials currently in progress. Furthermore, we believe that greater selectivity and stress are needed when defining those cases -individually analyzed and based on prognostic factors- that may benefit from neoadjuvant treatment (6,24,25).
Rising overall and disease-free survival, decreasing local recurrence rates, and having fewer postoperative complications should not be the only goal in the treatment of rectal cancer. We must provide our patients with the best quality of life. So, we should be stricter at the time of prescribing postoperative chemo-radiotherapy, we should have in mind the loss in quality of life that it involves, and maybe we should keep it only for a few patients with a high risk of progression.
REFERENCES
1. Ooi BS, Tjandra JJ, Green MD. Morbidities of adjuvant chemother-apy and radiotherapy for resectable rectal cancer. Dis Colon Rectum 1999; 42: 403-18. [ Links ]
2. Gastrointestinal Tumor Study Group. Prolongation of the disease-free interval in surgically treated rectal carcinoma. N Engl J Med 1985; 312: 1465-72. [ Links ]
3. Colorectal Cancer Collaborative Group. Adjuvant radiotherapy for rectal cancer: a systematic overview of 8507 patients from 22 randomised trials. Lancet 2001; 358: 1291-304. [ Links ]
4. Bosset JF, Mantion G, Lorchel F, Magnin V, Pelissier EP, Gerard JP, et al. Adjuvant and neoadjuvant radiation Therapy for rectal cancer. Sem Oncol 2000; 27: 60-5. [ Links ]
5. Martín de Vidales C, Cerezo L, Vazquez de la Torre ML, Zapatero A, Del Cerro E, Pinar B, et al. Estudio comparativo en el adenocarcinoma de recto: radioterapia preoperatoria o postoperatoria. Rev Esp Enferm Dig 1995; 87: 199-204. [ Links ]
6. Sauer R. Adjuvant and neoadjuvant radiotherapy and concurrent radiochemotherapy for rectal cancer. Pathol Oncol Res 2002; 8: 7-17. [ Links ]
7. Marjinen CAM, Glimelius B. The role of radiotherapy in rectal cancer. Eur J Cancer 2002; 38: 943-52. [ Links ]
8. Sociedad Valenciana de Cirugía. Guía de práctica clínica en el cáncer colorrectal. Generalitat Valenciana. Consellería de Sanitat. Valencia, 2002. [ Links ]
9. Tratamiento adyuvante del cáncer colorrectal. En: Lledoacute; S, ed. Cirugía Colorrectal. Asociacioacute;n Española de Cirujanos. Madrid: Arán Ediciones. 2000. [ Links ]
10. Glimelius B. Radiotherapy in rectal cancer. Br Med Bull 2002; 64: 141-57. [ Links ]
11. Minsky BD, Coia L, Haller DG, Hoffman J, John M, Landry J, et al. Radiation therapy for rectosigmoid and rectal cancer: results of the 1992-1994 patterns of care process survey. J Clin Onc 1998; 16: 2542-7. [ Links ]
12. Heald RJ, Moran BJ, Ryall RDH, Sexton R, MacFarlane JK, et al. Rectal cancer. The Basingstoke experience of total mesorectal excision, 1978-1997. Arch Surg 1998; 133: 894-9. [ Links ]
13. Buuml;low S, Christensen IJ, Harling H, Kronborg O, Fenger C. Recurrence and survival after mesorectal excision for rectal cancer. Br J Surg 2003; 90: 974-80. [ Links ]
14. Reis ED, Vine AJ, Heimann T. Radiation damage to the rectum and anus: pathophysiology, clinical features and surgical implications. Colorectal Dis 2002; 4: 2-12. [ Links ]
15. Kollmorgen CF, Meagher AP, Wolff BG, Pemberton JH, Martenson JA, Illstrup DM. The long-term effect of adjuvant postoperative chemoradiotherapy for rectal carcinoma on bowel function. Ann Surg 1994; 220: 676-82. [ Links ]
16. Nathanson DR, Espat NJ, Nash GM, D"Alessio M, Thaler H, Minsky BD, et al. Evaluation of preoperative and postoperative radiotherapy on long-term functional results of straight coloanal anastomosis. Dis Colon Rectum 2003; 46: 888-94. [ Links ]
17. Coia LR, Gunderson LL, Haller D, Hoffman J, Mohiuddin M, Tepper JE, et al. Outcomes of patients receiving radiation for carcinoma of the rectum. Results of the 1988-1989 patterns of care study. Cancer 1999; 86: 1952-8. [ Links ]
18. Arraras JI, Vera R, Manterola A, Martínez M, Arias F, Martínez E, et al. El cuestionario de calidad de vida para cáncer colorrectal. EORTC QLQ-CR38. Estudio de validacioacute;n para nuestro país. Oncología 2003; 26 (9): 285-92. [ Links ]
19. Marijnen CAM, Kapiteijn E, Van de Velde CJH, Martijn H, Steup WH, Wiggers T, et al. Acute side effects and complications after short-term preoperative radiotherapy combined with total mesorectal excision in primary rectal cancer: report of a multicenter randomized trial. J Clin Oncol 2002; 20: 817-25. [ Links ]
20. Ammann K, Kirchmayr W, Klaus A, Muuml;hlmann G, Kafka R, Oberwalder M, et al. Impact of neoadjuvant chemoradiation on anal sphincter function in patients with carcinoma of the midrectum and low rectum. Arch Surg 2003; 138: 257-61. [ Links ]
21. Dahlberg M, Glimelius B, Pahlman L. Changing strategy for rectal cancer is associated with improve outcome. Br J Surg 1999; 86: 379-84. [ Links ]
22. Wiggers T, Van de Velde CHJ. Reduction by half. The need for standarised surgical technique in studies of radiotherapy for rectal cancer. Eur J Surg 1999; 165: 407-9. [ Links ]
23. MacFarlane JK, Ryall RDH, Heald RJ. Mesorectal excision for rectal cancer. Lancet 1993; 341: 457-60. [ Links ]
24. Kapiteijn E, Kranenbarg EK, Steup WH, et al. Total mesorectal excision (TME) with or without preoperative radiotherapy in the treatment of primary rectal cancer. Eur J Surg 1999; 165: 410-20. [ Links ]
25. Simunovic M, Sexton R, Rempel E, Moran BJ, Heald RJ. Optimal preoperative assessment and surgery for rectal cancer may greatly limit the need for radiotherapy. Br J Surg 2003; 90: 999-1003. [ Links ]











 texto en
texto en