Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Revista Española de Enfermedades Digestivas
versión impresa ISSN 1130-0108
Rev. esp. enferm. dig. vol.109 no.12 Madrid dic. 2017
PICTURES IN DIGESTIVE PATHOLOGY
Immune mediated colitis caused by lung cancer treatment with atezolizumab
Santiago González-Vázquez1, Susana de-la-Riva-Onandía1, José Ignacio Echeveste2 and Miguel Muñoz-Navas1
Departments of 1Gastroenterology and 2Pathology. Clínica Universidad de Navarra. Pamplona, Spain
Introduction
Atezolizumab is an IgG1 isotype monoclonal antibody against the protein programmed cell death-ligand 1 (PD-L1). PD-L1 may be highly expressed in some tumors and is believed to inhibit immune cells that recognize and attack tumor cells. Inhibition of PD-L1 can remove its inhibitory effect and provoke an anti-tumor response.
In October 2016, the Food and Drugs Administration (FDA) approved atezolizumab for the treatment of patients with metastatic non-small cell lung cancer after disease progression during or following platinum based chemotherapy.
Case report
We present the case of a 43-year-old male with stage IV lung adenocarcinoma in progression, despite standard chemotherapy. He was participating in a clinical randomized trial with four cycles of chemotherapy as follows: cisplatin, pemetrexed and atezolizumab. He showed a good tolerance.
After the last cycle of chemotherapy, he had bloody diarrhea and fever. Stool cultures were negative. A colonoscopy was performed and a pancolitis with a multiple fibrin-coated ulcer was identified. Numerous glandular crypts with cryptic micro-abscesses and a chronic inflammatory lymphoplasmocytic infiltrate were found on biopsy of the colon. These histological findings are similar to those described in ulcerative colitis.
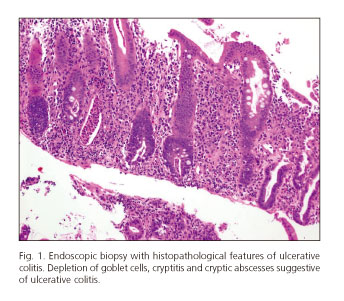
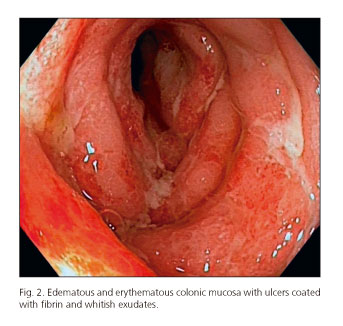
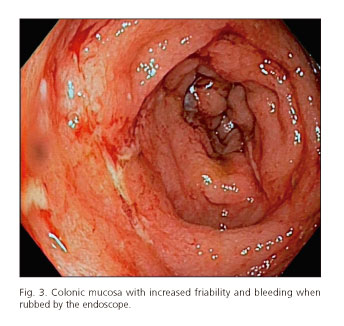
Conclusion
Immune mediated colitis is a possible adverse event that is poorly described. It occurs in 19.7% of all patients receiving atezolizumab and requires the administration of corticosteroids for its resolution.
References
1. Abdel-Rahman O, ElHalawani H, Fouad M. Risk of gastrointestinal complications in cancer patients treated with immune checkpoint inhibitors: A meta-analysis. Immunotherapy 2015;7(11):1213-27. DOI: 10.2217/imt.15.87. [ Links ]
2. De Velasco G, Je Y, Bossé D, et al. Comprehensive meta-analysis of key immune-related adverse events from CTLA-4 and PD-1/PD-L1 inhibitors in cancer patients. Cancer Immunol Res 2017. pii: canimm.0237.2016. DOI: 10.1158/2326-6066.CIR-16-0237. [ Links ]
3. Nishijima TF, Shachar SS, Nyrop KA, et al. Safety and tolerability of PD-1/PD-L1 inhibitors compared with chemotherapy in patients with advanced cancer: A meta-analysis. Oncologist 2017. pii: theoncologist.2016-0419. DOI: 10.1634/theoncologist.2016-0419. [ Links ]













