INTRODUCTION
Magnetic resonance imaging, endoluminal ultrasound and computed tomography are imaging tools commonly used to evaluate rectal tumor staging 1. Rectal endoluminal ultrasonography (US) with rigid probes or endoscopic ultrasonography (EUS) is the preferential method for local staging of rectal carcinoma (RC) and decisively influences the therapeutic approach of patients 2. Nevertheless, its accuracy varies significantly, ranging from 63% to 95% for T staging and from 64% to 80% for N staging 1,3,4,5. It is well known that operator expertise is an important factor in the accuracy of RC by endoluminal US 6. However, tumor anatomical characteristics (e.g., stenosis), lymph node location (e.g., pelvic lateral nodes) or criteria (e.g., size) may also influence the final result 4,7.
Conventional EUS (C-EUS) does not provide whole tumor assessment for all stenosing tumors. Miniprobe EUS (mp-EUS) may easily overcome these obstacles due to its reduced diameter and flexibility 8,9. mp-EUS may be the most adequate ultrasonographic tool for the differential diagnosis of tumors limited to the mucosa (T1m) or with involvement of the submucosa (T1sm) as it uses high frequencies and thus resolves the limitations of C-EUS 10,11.
C-EUS provides only two-dimensional images of the lesions and structures. Three- dimensional EUS (3D-EUS) may be able to provide simultaneous spatial information of different planes and even a multi-plane vision. It also allows the isolation of structures or lesions of interest and presents them in different perspectives, changing their texture and/or transparency 12,13,14,15. However, the application of 3D-EUS in this setting has rarely been reported 12,14,16,17. Therefore, we aimed to compare RC preoperative staging using C-EUS with 3D-EUS and mp-EUS.
METHODS
Selection of participants
The prospective study was approved by the Ethics Committee for Health of our hospital. Patients with RC (defined as located up to 15 cm from the anal verge) that underwent endoscopic ultrasonography staging of the disease gave their written informed consent prior to inclusion in the study. Patients less than 18 years of age, pregnant women or individuals unable to give informed consent were excluded.
Procedures
Two enemas were given one hour before the procedures. Three types of equipment were used in staging: a) a conventional echoendoscope with frequencies of 7.5 and 12 MHz (Olympus GF-UM20(r)); b) mp-EUS with 12 MHz (Olympus UM-2R(r)); and c) 3D-EUS with the Easy 3D Freescan software from Echotech(r). The assessment for T and N stages (defined in accordance with TNM staging 18)) was initiated with mp-EUS followed by C-EUS. 3D-EUS was the final technique performed and images were acquired via a conventional echoendoscope. The identification of lymph nodes in the perirectal space was conducted from the distal rectum to the iliac vessels. The differences in the ability of the three techniques to assess the entire lesion were also assessed.
Reference tests
Patients were divided in two groups according to the use of neoadjuvant therapy with radio/chemotherapy. Prior assessment with a conventional echoendoscopy was performed before neoadjuvant therapy and was considered as a reference in all cases. Pathological assessment after surgery was considered as the gold standard for those who did not receive preoperative therapy.
Statistical analysis
The PASW(r) version 21 software was used for the analysis. The kappa coefficient was used to estimate the agreement between techniques and accuracy; this was calculated as the proportion of true results versus the total number of patients. In addition, sensitivity and specificity were estimated as the proportion of true positive or negative cases, respectively.
The relationship between longitudinal or circumferential extent of the tumor and staging accuracy was evaluated for all three techniques using a Spearman's correlation.
RESULTS
Sixty patients aged between 34 and 89 years (mean value of 63.8 ± 11.8) were assessed, and 36 (60%) patients were male. All patients underwent surgery, 27 (45%) without preoperative adjuvant therapy.
Mp-EUS assessed the lesion in its entirety in 97% of cases, whereas this was achieved less frequently via C-EUS and 3D-EUS, in 85% of cases (p = 0.01). Staging was not possible using C-EUS in 15% (n = 9) of cases vs 3% (n = 2) of cases with mp-EUS, due to tumor stenosis. In these cases where it was not possible to evaluate the whole of the tumor, T and N stages results were based on the tumor extent that was observed.
T staging
Comparison of T staging by mp-EUS, 3D-EUS and C-EUS in all patients (n = 60) (Table 1)
Both the mp-EUS and 3D-EUS techniques were highly concordant with C-EUS for T staging, with Kappa coefficients of 0.81 and 0.87. Global accuracy of T staging by mp-EUS compared to C-EUS was 86.7% and 81.7% compared to 3D-EUS.
Comparison of T staging obtained using different EUS techniques and anatomopathological staging (APS) (n = 27) (Table 2)
T staging by C-EUS, mp-EUS and 3D-EUS was concordant with APS with Kappa coefficient values of 0.81, 0.76 and 0.95, respectively.
Simultaneous comparison of T staging using the three EUS techniques and T APS (n = 27) (Table 2)
There were no significant differences with regard to the simultaneous comparison of the three techniques (p = 0.75).
N staging
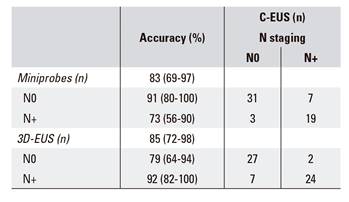
Comparison of N staging by mp-EUS, 3D-EUS and C-EUS staging in all patients (n = 60) (Table 3)
The mp-EUS and 3D-EUS analyses were concordant with C-EUS for N staging, with kappa coefficients of 0.65 and 0.79, respectively.
The sensitivity of C-EUS compared to mp-EUS and 3D-EUS for the identification of metastasized nodes was 73.1% and 92.3%, respectively. The corresponding values for specificity were 91.2% and 79.4%, positive predictive values were 86.4% and 77.4%, negative predictive values were 81.6% and 93.1% and global accuracy was 83.3% and 85%, respectively.
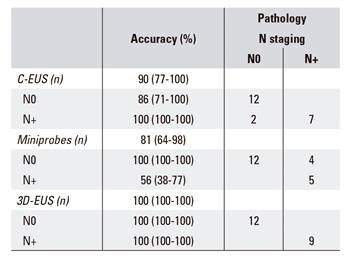
Comparison of N staging obtained via different EUS techniques and APS (n = 21) (Table 4)
Anatomopathological information with regard to the presence of metastasized nodes was available in 21 (77.8%) cases. Of the 27 patients that underwent surgery, it was not possible to obtain this information in six (22.2%) cases as a transanal resection was performed. N staging by C-EUS, mp-EUS and 3D-EUS and pathological analysis were concordant with respective kappa coefficients of 0.80, 0.59 and 1. The sensitivity of C-EUS, mp-EUS and 3D-EUS for the identification of metastasized nodes was 77.8%, 55.6% and 100%, respectively. The corresponding values for specificity and positive predictive values were 100% for the three techniques, whereas the negative predictive values were 85.7%, 75% and 100% and global accuracy was 90.5%, 81% and 100%, respectively.
Simultaneous comparison of N staging accuracy using the three EUS techniques and N APS (n = 21) (Table 4)
There were no significant differences between the three techniques (χ2 = 4.42; p = 0.11).
DISCUSSION
The continuous technological development has led to the application of new techniques associated with endoscopic ultrasonography 19. Good results were obtained in this study with mini-probes and three-dimensional endoscopic ultrasonography.
There was a significant agreement with regard to T staging between miniprobes, 3D and conventional EUS. These values range between 77 and 93% in previous reports 12,14,16,17. Our findings confirm the high accuracy of 3D-EUS, which is higher than that of conventional EUS. However, stenosing rectal cancer cannot be assessed either by C-EUS or 3D-EUS in up to 21.6% of cases 20, the rate was 15% in our study. Interestingly, miniprobe-EUS allowed the identification of most lesions in their entirety. Miniprobes can in fact transpose stenosing RC (Fig. 1), overcoming the rigid end and "large" diameter characteristics of C-EUS, which may inhibit RC staging 4,7.

Fig. 1 Stenosing RC. A. Observation by colonoscopy with a miniprobe inserted in the lumen. B. Ultrasonographic image showing the invasion by perirectal fat.
Both 3D-EUS and mp-EUS had a high accuracy (Fig. 2) for T1 staging, which is in agreement with previous reports 21,22. With regard to T2 staging, 3D was superior to mp-EUS, which tended to overstage tumors 12,16,17,22,23,24. A very high accuracy for T3 staging was reported for 3D or mp-EUS 12,16,17,23,24,25,26, whereas mp-EUS had the lowest accuracy in relation to the higher ultrasound frequency for T4 staging, which is in line with current evidence 17,24,24,25,26,27. The use of 3D for T staging of rectal cancer had the best accuracy with mp and allowed the staging of stenosing tumors, although with some limitations for large masses (e.g., T4 staging).

Fig. 2 RC staging by 3D-EUS with invasion of the submucosa but without surpassing it, as confirmed by the surgical sample.
With regard to lymph node diagnosis with EUS, the results obtained with mp-EUS and 3D-EUS were also encouraging since they were in significant concordance with C-EUS, with a global accuracy of 90.5%. The highest sensitivity was obtained with 3D-EUS (100%), as well as a very high negative predictive value (100%). This not only represents the high capacity of 3D-EUS to identify lymph nodes but also reinforces our option for not imposing a cut-off value for node diameter in order to classify it as metastasized. However, there is no consensus with regard to this matter 28,29. In fact, this trend to improved accuracy was also observed in other studies 12,17,30,31.
In conclusion, Mp-EUS and 3D-EUS are valid techniques compared to C-EUS for RC staging. We suggest that Mp-EUS may be an alternative to C-EUS due to the possibility of staging a carcinoma during colonoscopy with interesting T-staging results, and it can usually assess the entire tumor mass. This is an advantage for stenosing masses. More importantly, even though the results were not statistically significant in the comparison of 3D-EUS with C-EUS, this technique may become the gold standard method in RC staging. This will ultimately improve the clinical decisions taken with regard to these patients, particularly for T2 vs T3 staging, which is highly relevant in patient management.