Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Revista Española de Enfermedades Digestivas
versión impresa ISSN 1130-0108
Rev. esp. enferm. dig. vol.110 no.5 Madrid may. 2018
https://dx.doi.org/10.17235/reed.2018.5408/2017
REVIEW
Quality indicators in colonoscopy. The colonoscopy procedure
1Servicio de Digestivo. Hospital San Juan de Dios. Santa Cruz de Tenerife, Spain
2Servicio de Calidad Asistencial. Hospital Clínico Universitario Virgen de la Arrixaca. Murcia, Spain
3Unidad de Endoscopias. Servicio de Medicina de Aparato Digestivo. Hospital Clínico Universitario Virgen de la Arrixaca. Murcia, Spain
4Gestión del Conocimiento. Sociedad Española de Patología Digestiva (SEPD). Madrid, Spain
5Servicio de Medicina de Aparato Digestivo. Hospital Clínico Universitario Virgen de la Arrixaca. Murcia, Spain
INTRODUCTION
When attempts are made to define quality within a health care setting, a reflective attitude is fitting on the huge number of potential, legitimate definitions that may be put forward according to the approach under consideration. A way of approaching this concept would be to define it as that property or set of properties inherent in a health care service that allows its being considered as comparable to, or better or worse than, other services of the same kind. Three relevant characteristics stem from this definition. First, the need to understand quality as a relative concept. Second, the fact that this concept involves comparison (comparable, better, worse). Third, the need for a standard, norm, or model to which comparisons may be made 1.
Thus, depending on the approach under consideration, a definition of health care quality may be diverse. Bearing this heterogeneity in mind, Avedis Donabedian systematized quality assessment in health care as a triad made up of structure, process, and outcome. Structural measurements or indicators assess characteristics in the overall health care setting, process indicators assess performance during service provision, and outcome indicators assess the effects of the health care services provided 2.
As was to be expected, and taking into consideration the relevance quality measurement has progressively acquired within health systems, endoscopic procedures have now been included in this systematic evaluation by means of tools such as the indicators posited by Donabedian. However, a major challenge gastroenterologists and endoscopists must confront on a daily basis is the maintenance of high quality standards in colonoscopy despite its increasing demand. It is therefore essential that ongoing quality improvement programs be implemented using evidenced-based, valid, reliable indicators. In accordance with this need, the Sociedad Española de Patología Digestiva (SEPD) has developed a project dealing with digestive endoscopy indicators. In continuation of this enterprise, the present study deals with quality procedures and indicators as recommended for the colonoscopy setting. It is relevant to refer to the initial study that started this project 3 as it reviewed concepts related to general and key quality aspects, including dimensions or values, and the importance of indicator development along the lines put forward by Donabedian as well as from an endoscopic perspective 1,2,3,4. Consequently, this study was meant to suggest the structure, process, and outcome procedures and indicators necessary to implement and evaluate quality in the colonoscopy setting.
METHODOLOGY
The study was developed along two phases. First, a multidisciplinary task force was formed, based at Hospital Clínico Universitario Virgen de la Arrixaca, Murcia, to review the literature and design a procedure for diagnostic colonoscopy. In a second phase, the team's proposals were reviewed and assessed by a panel of experts selected by the SEPD until a definitive version was established. Subsequently, spec sheets were developed for each indicator in order to assess the procedures involved.
Literature search and study selection strategies
Two strategies were used for searching the literature: one was directed to clinical practice guidelines (CPGs); the other, to original and review papers. Colonoscopy-related CPGs were taken from three international sources (Agency for Healthcare Research and Quality [AHRQ], National Institute for Health and Care Excellence [NICE], Scottish Intercollegiate Guidelines Network [SIGN]) and a Spanish one (GuíaSalud), as well as from reviews found in the web sites of endoscopy and gastroenterology societies (American Society for Gastrointestinal Endoscopy [ASGE], American Gastroenterological Association [AGA], European Society of Gastrointestinal Endoscopy [ESGE], Sociedad Española de Endoscopia Digestiva [SEED], SEPD, and Asociación Española de Gastroenterología [AEG]). Original papers were obtained from the Web of Knowledge (WoK), PubMed, and Cochrane databases using the following search strategy: selection of all documents dated from January 1, 2006 to August 10, 2016 including any of the following descriptors: [colonoscop*] plus [informed consent, quality, safety, (security), assessment, assurance, indicators, criteria]. Active filters included: clinical trial, controlled clinical trial, meta-analysis, randomized controlled trial, review, guideline, practice guideline, publication date from 2006/01/01 to 2016/08/10; humans; adults; language: English, Spanish. Complementarily, a review was undertaken of the references included in the original papers, guidelines, and meta-analyses selected and included in the analysis, and those deemed interesting and previously overlooked were retrieved. Once the search protocol was completed, all articles obtained were reviewed and assessed separately by two reviewers. Each reviewer screened the selected studies using any of the following criteria: a) the paper includes recommendations on appropriate preparation, performance, and follow-up; b) the paper includes or suggests quality indicators for structure, process, or outcomes. Studies selected by only one reviewer were checked out by the other in order to decide their inclusion or otherwise. To provide a homogeneous assessment of every study selected, a table was designed to collect the information included on structure, process, and outcome indicators, and whether that information was explicit or otherwise. Furthermore, the table also included the type of study referenced (clinical trial, observational study, meta-analysis, etc.), which was identified as related to colonoscopy.
Colonoscopy procedure design
Taking the selected references and author experience as a point of departure, the activities required for each procedure were collected and sorted. Regarding the procedures shared by all endoscopic examinations, the logical structural, functional, and organizational differences found among clinical digestive endoscopy units limit their development to a minimum. Similarly, descriptions of specific techniques exclusively applied to some situations were excluded, as they were not included in the goals of the present work. The result was diagrammed in flowcharts or parallel path flowcharts. Proposals were reviewed and assessed by a panel of professionals selected by the SEPD until a definitive version was agreed.
Indicator design
In order to obtain valid indicators, quality was measured for the knowledge available regarding procedure-related activities and for the documents selected after the literature search. This was accomplished using the GRADE system. In this system, quality of "evidence" (hereafter used without quotations to mean the best proof-based knowledge available) is initially classified as high or low according to its origin in experimental or observational studies. Then, by considering a number of items that may diminish or increase the baseline score, quality is graded as high, moderate, low, or very low 4,5. To ensure reliability and to facilitate scoring for the indicators selected by clinical units, each indicator is provided with a datasheet including scope of applicability (procedure[s] where it may be applied); denomination; calculation formula; type of indicator according to Donabedian's model 3; temporal relationship with procedure (pre-procedure, procedure, post-procedure); quality dimension it relates to; justification, exclusions and clarifications; and supporting level of evidence.
RESULTS AND DISCUSSION
Search results
The selection process began by suppressing duplication and by excluding studies explicitly dealing with colonoscopy as cancer screening tool, poorly referenced studies, abstract-only publications, papers unavailable or published before 2006, papers dealing with pediatric topics, veterinary medicine or nondigestive endoscopy, and papers in a language other than Spanish or English. In our search and assessment, we made an exception for the definition of interval cancer, which specifically refers to screening colonoscopy, because of its being a direct outcome indicator (albeit not the only one as will be seen later) of colonoscopy quality.
A total of 260 studies were included in the paired analysis and assessed full-text, including both randomized and non-randomized clinical trials, as well as high-quality series of cases, reviews and meta-analysis for a total of 107.
Colonoscopy procedure
The procedure has been plotted in a diagram (Fig. 1) with the following correlative steps:
1. Place patient in appropriate position:
- Left lateral position, although a supine position is also feasible.
- Head in neutral position with pillow support.
- If venous access is required, preferably use the right arm.
2. Check functioning of endoscope:
- Adequate quality visualization.
- Tip angulation.
- Air and water.
- Suction.
3. Lubricate endoscope:
4. Insert endoscope:
- Rectal digital examination with generous lubrication.
- Select insertion technique (may be hybrid or mixed, by sections):
One-person method. The endoscopist is autonomous for insertion. Assistance is only needed for abdominal compression, position changes or support.
Two-person method. An assistant handles the tube; the endoscopist operates the controls.
Pass anal sphincter. Three options:
Oblique pressure with forefinger until sphincter relaxation.
Introduce thumb tip and replace with tube.
Direct insertion.
5. Advance to cecal fundus:
Maneuver properly through the colon:
Advance tube in short steps to avoid loop formation.
Always keep endoscope centered within the colonic lumen.
Coordinate appropriately rotation (torque) and angulation movements using both hands.
Negotiate angles with mild pushes followed by short withdrawals with clockwise rotation on the tube to maintain rectification.
Improve visibility by insufflating while avoiding overdistension.
Position changes may help passage through difficult colonic areas.
Respond adequately to findings:
Excise or biopsy lesions according to type, size, and consistency.
Reference lesions, including aspect, shape, size, and presumptive diagnosis.
lesions using distance from anus/cecum or anatomic hallmarks when feasible (transverse colon, cecum, hepatic and splenic flexures, anastomosis, scars, etc.).
Pass through rectum by pushing the endoscope.
Pass through sigmoid colon by pushing the tube counterclockwise, usually forming an alpha loop, with help from an assistant.
Release alpha loop in descending colon by withdrawing colonoscope and applying clockwise torque.
Advance by pushing tube to transverse colon.
Go through mid-transverse colon angulation by withdrawing endoscope with clockwise torque while aspirating gently.
Use aspiration and mild torque to go beyond hepatic flexure. Epigastric compression may be needed to prevent loops in transverse colon.
Push and aspirate along ascending colon down to cecal fundus.
Consider imaging.
6. Assess entry into ileum:
- Weigh according to test indication, findings, and anatomic difficulty.
7. Withdraw tube:
- Maneuver slowly.
- Clean thoroughly, remove residual mucus.
- Look behind every haustrum.
- Observe attentively to identify haustral irregularities and submucosal circulation changes.
- Respond adequately to findings:
Polyps:
- Describe morphology (Paris classification), size, and location.
- Consider magnification to improve diagnosis.
- Excise or biopsy.
Cancer lesions:
- Locate and biopsy.
- Tattoo with India ink (except lower rectum and cecum).
- Advance to cecum if lesion passable.
Colitis:
- Collect biopsy simple.
- Describe extent, patchy or continuous involvement.
- Try and visualize ileum.
Vascular lesions (angiodysplasia, etc.)
- Describe.
- Consider electrocoagulation.
Diverticula:
- Describe.
- Avoid insufflation, particularly if inflammatory component present.
- Aspirate as much air as possible by segments.
8. Visualize rectum:
- Rest tube on wall and turn to obtain retroversion in rectal ampoule.
- Inspect mucosa.
9. End examination:
- Withdraw tube completely.
- Pass endoscope over to ancillary personnel for cleansing and new preparation.
Indicators
Table 1 offers an overall view of colonoscopy-specific indicators (boldface) and general indicators common to all endoscopic procedures.
Among quality dimensions, efficiency is commonly overlooked by colonoscopy quality guidelines. Here, efficiency has been considered as a cross-sectional dimension reflecting the way our proposed indicators are evaluated. For instance, "all polyps excised" was included as uncommon indicator to emphasize the need to satisfy patient requirements in one single procedure. Efficiency also influenced the definition of indicators regarding adenoma excision, as based exclusively on the index colonoscopy (excluding repeat colonoscopies to complete a prior procedure).
B.06. Appropriate interval between colonoscopies
Several clinical practice guidelines have been published regarding follow-up for these patients 7,8,9. Such guidelines are primarily based on population studies and clinical trials with different types of follow-up, and take into account the risk for recurrent lesions after an index colonoscopy. This study recommends that every digestive endoscopy unit adheres to one of these guidelines to establish their own practice, and assess the proposed indicator.
Excision of adenomas or serrated polyps during colonoscopy is a risk factor for the subsequent development of new adenomas and colorectal cancer. The indication of colonoscopy for the follow-up of previously excised adenomas or serrated polyps is currently a very usual one. Overindication is here common, which represents an unnecessary workload. Furthermore, inadequate or lax follow-up represents a risk factor for the development of new lesions or even colorectal cancer. Adequate follow-up will prevent colorectal cancer development, but not completely. Follow-up after excision of a colon adenoma will depend upon three factors: index colonoscopy quality, characteristics and number of excised lesions, and excision technique or difficulty 10,11.
C.04. Colon preparation
Achieving adequate colonic cleansing or preparation is one of the most common quality issues in colonoscopy 12. Good or, at least, moderately good colonic cleansing facilitates identification of colon adenomas, as was confirmed by several clinical trials and meta-analyses 13,14. Thus, it indirectly prevents colorectal cancer development, as will be seen below. Clinical practice guidelines, based on systematic reviews and meta-analyses, recommend using split doses or the entire cleansing dose just a few hours before the procedure 15,16,17,18. Colon preparation must be individualized according to personal risks 19. The recommended standard for adequate cleansing ranges from 85% to 90%, the latter figure being recommended for screening colonoscopy 20,21. This paper suggests as recommendation using the Boston scale 22,23. A score of two or three for each segment is recommended when using this scale 13.
C.05. Complete colonoscopy
A technical goal for any colonoscopy is complete colonic examination. Incomplete examinations are associated with increased risk for interval cancer 24. In clinical practice, meeting this indicator was perhaps highly variable until recently 25,26,27,28,12. Colonoscopy is considered as complete when the tip of the endoscope reaches beyond the ileocecal valve, which allows to assess the cecum's medial aspect (a common site for interval cancer) and the area around the appendicular orifice. The report must state that cecal structures (ileocecal valve, appendicular orifice) were identified, and an image including the appendicular orifice and characteristic cecal fold should also be attached 20,21,29,30,31.
C.06. Excision of all polyps smaller than 20 mm
Colonoscopy, as any other medical procedure, must be cost-effective, if possible on the first attempt. A technical goal is excision of all polyps within the reach of any average endoscopist 12,25. While scales are available to assess the difficulty of polyp excision, they remain inadequately validated 11. Although evidence is sparse, an indicator based on polyp size (20 mm) is considered to reflect the overall technical quality of polyp excision by an endoscopist. Some patients may have complex or multiple polyps. An endoscopist may choose to initially excise the more challenging ones and then leave the easier polyps for a second colonoscopy procedure; or he/she may excise all polyps in some specific segment 30. As with other indicators that will be seen below, this indicator should only be assessed for index procedures. An index colonoscopy is the first colonoscopic procedure performed in a given patient (whether for symptoms, screening, follow-up, etc.).
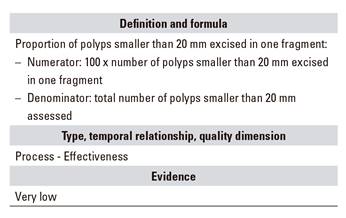
C.07. All polyps smaller than 20 mm excised in one fragment
Among indicators to assess the technical ability of an endoscopist as regards polyp excision, the following may be considered: the percentage of polyps excised in one fragment and the fragment rate per mucosectomy above 2 cm. For most endoscopists, it is technically possible to excise most polyps smaller than 20 mm in one fragment 32. However, some polyps smaller than 20 mm may be challenging to access, and excision in two or more fragments may be then advisable.
C.08. Withdrawal time
Withdrawal time is considered as an indirect quality indicator related to adenoma and interval cancer identification rates 33,34. It is therefore an indicator to consider especially for physicians with low adenoma detection rates. This indicator is assessed based on colonoscopies where no polyps are found. Standard average time ranges from six to eight minutes for one endoscopist 35,36. Importantly, colon length is variable, and withdrawal time might be shorter in an adequately cleansed shorter colon. Not all studies find this indicator valuable 37,38.
C.09. Biopsy sample collection in patients with chronic diarrhea
In patients with intermittent or chronic watery diarrhea, the prevalence of microscopic colitis has been estimated around 9.5-13.7% 39,40. Diagnosing these conditions may improve quality of life through the use of specific therapy with drugs such as oral budesonide 41. Thus far, the only way to diagnose microscopic colitis (whether collagenous or lymphocytic) is systematic biopsy collection during colonoscopy. Several schemes are recommended, including biopsy sample collection all through the colon, which involves at least four samples from the proximal colon up to the splenic flexure 42,43.
C.10. Number and distribution of biopsies in patients with inflammatory bowel disease (IBD)
Patients with IBD and colon involvement are at higher risk of colorectal cancer 44. This risk increases with colonic involvement extension and time since disease onset. In these patients, cancer is preceded by dysplasia. Follow-up programs for these patients including multiple biopsies may improve life expectancy. Recently, several clinical trials found an increased effectiveness in dysplasia detection using high-resolution endoscopy and chromoendoscopy 45, but results may be conflicting in clinical practice 46,47. In this study, a program using chromoendoscopy and targeted biopsies, or a classic regimen of four-quadrant biopsy samples every 10 cm, is recommended for patients with chronic IBD on follow-up for the screening of dysplasia and colorectal cancer.
D.04. Colon preparation report
In order to establish an appropriate follow-up interval, or merely to know whether a colonoscopy was high-quality, this information should be available in the procedure's report. Colonic cleansing quality should be recorded using a validated scale and by segments 22,23,36,48. However, there is often significant variability, and this and other relevant aspects of colonoscopy occasionally remain outside the scope of reports 39.
D.05. Withdrawal time report
We suggest that this indicator be included to provide information on procedure quality in the colonoscopy report. However, as already mentioned, what matters is the average withdrawal time per endoscopist performing colonoscopies. However, including this indicator may lead endoscopists to maintain higher average withdrawal times 36.
E.01. Incidence of adverse effects
In order to facilitate improved endoscopist performance, digestive endoscopy units have increasingly measured adverse events (AEs). While severe AEs are usually perceived as uncommon or rare, a considerable number of unexpected hospital admission may result from colonoscopy procedures, as well as considerable health care costs, and mortality in a healthy population. Lower digestive endoscopy, including colonoscopy, has been increasingly used as CRC screening instrument both in Europe and the US during the past few years, supported by growing evidence of its effectiveness to reduce CRC rates and CRC-related mortality. Therefore, awareness of colonoscopy-related adverse events in routine practice is key for the decision-making process of policy makers 49,50.
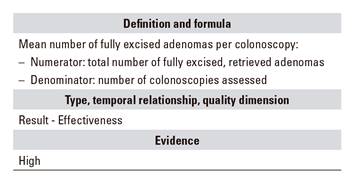
E.04 - Mean number of adenomas excised per colonoscopy procedure
These two indicators complement each other. While terms such as adenoma detection index or adenoma detection rate have been used, such denominations are confusing and we had better refer to these markers as percentage of colonoscopies with at least one adenoma excised and number of adenomas excised per colonoscopy. We aim at using natural common language that may be understood by all those involved in the colonoscopy: general public, managers, and any other person involved regardless of education.
Traditionally, percentage of colonoscopies with at least one adenoma has been used as key indicator; this has been the most commonly studied indicator and the one best correlated with the prevention of colorectal and interval cancer, both in screening and non-screening colonoscopies 34,49,50. We included the indicator "mean number of excised adenomas per colonoscopy" as an additional marker of endoscopist technical quality 51,52. Currently, no appropriate standards are available for the general population. In waiting for newer data to emerge, we suggest a standard of 20% as percentage of colonoscopies with at least one adenoma excised among all index colonoscopies, regardless of age or indication.
Other potential indicators would include: percentage of colonoscopies with advanced adenoma, mean number of advanced adenomas per colonoscopy, number of adenomas in patients with adenomas or number of adenomas minus the index lesion; while they allow differentiations in quality between endoscopists, their required number of patients is very high.
E.05. Incidence of interval cancer after colonoscopy
This may well be the best outcome indicator for colonoscopy, particularly so in colorectal cancer screening programs 5,7. Though analyzing indicators for colonoscopy in general really is outside our scope, this is theoretically the marker with the strongest impact since a key goal for any colonoscopy is the long-term prevention of colorectal cancer development. Appropriate mucosal inspection, accompanied by excision of identified lesions, prevents colorectal cancer from developing. However, a significant number of undetected lesions is known to exist. Furthermore, some lesions, even flat or smaller lesions, may be highly aggressive, which results in a variable prevalence of post-colonoscopy colorectal cancer at three to five years after the procedure that oscillates between 0% and 9% 37,38,39. Prolonged withdrawal time, improved colon cleansing, adequate follow-up intervals for patients with adenoma, and higher adenoma excision rates will prevent colorectal cancer 39,40,48,50,51,52. This indicator has potential barriers, including patient loss to follow-up, prolonged interval between consecutive colonoscopies, and low incidence of post-colonoscopy cancer. The calculation approach for each endoscopy unit remains poorly established, but should ideally include all colonoscopies performed, even those with normal results. However, this research recommends that it be considered in the follow-up program of patients with colonic adenomas or serrated polyps in order to facilitate its monitoring 41,42,43,44,45,46.
CONCLUSIONS
This paper considered the use of a number of indicators to be included in colonoscopy quality improvement programs, together with the general indicators considered by the prior paper. The total number of indicators to assess one endoscopy may rise up to 30. Even with a data processing system, managing all these indicators in the setting of ongoing quality monitoring may represent a workload difficult to comply with. Therefore, it is important that assessment be based on key indicators. Various scientific societies agree on the significance of routinely measuring indicators relevant to endoscopy unit performance, thus allowing ongoing improvement cycles according to their results. However, if an endoscopy unit performs above standard during ongoing monitoring for a specific indicator, and keeps this up long-term, our monitoring activity will be unnecessary as it will identify no opportunities for improvement. Hence, flexibility is advisable, and indicator monitoring should be adapted to needs of improvement in each unit. For other quality-related activities, such as implementing an improvement cycle or redesigning health care activities, quality assessment should take the highest number possible of indicators into consideration.
BIBLIOGRAFÍA
1. Arcelay A. Adaptación de un modelo de calidad total a las instituciones sanitarias españolas. Rev Calid Asist 2000;15:184-92. [ Links ]
2. Donavedian A. La calidad de la asistencia. ¿Cómo podría ser evaluada? JANO 1989;864:103-10. [ Links ]
3. López-Picazo J, Alberca de las Parras F, Sánchez del Río A, et al. Quality indicators in digestive endoscopy: introduction to structure, process, and outcome common indicators. Rev Esp Enferm Dig 2017;109(6):435-50. DOI: 10.17235/reed.2017.5035/2017 [ Links ]
4. Petersen BT. Quality assurance for endoscopists. Best Pract Res Clin Gastroenterol 2011;25(3):349-60. DOI: 10.1016/j.bpg.2011.05.003 [ Links ]
5. Rees CJ, Bevan R, Zimmermann-Fraedrich K, et al. Expert opinions and scientific evidence for colonoscopy key performance indicators. Gut 2016;65(12):2045-60. DOI: 10.1136/gutjnl-2016-312043 [ Links ]
6. Rees CJ, Thomas Gibson S, Rutter MD, et al. UK key performance indicators and quality assurance standards for colonoscopy. Gut 2016;65(12):1923-9. DOI: 10.1136/gutjnl-2016-312044 [ Links ]
7. Atkin WS, Valori R, Kuipers EJ, et al. European guidelines for quality assurance in colorectal cancer screening and diagnosis. First edition - Colonoscopic surveillance following adenoma removal. Endoscopy 2012;44(Suppl 3):SE151-63. DOI: 10.1055/s-0032-1309795 [ Links ]
8. Hassan C, Quintero E, Dumonceau J-MM, et al. Post-polypectomy colonoscopy surveillance: European Society of Gastrointestinal Endoscopy (ESGE) Guideline. Endoscopy 2013;45(10):842-51. DOI: 10.1055/s-0033-1344548 [ Links ]
9. Lieberman DA, Rex DK, Winawer SJ, et al. Guidelines for colonoscopy surveillance after screening and polypectomy: a consensus update by the US Multi-Society Task Force on Colorectal Cancer. Gastroenterology 2012;143(3):844-57. DOI: 10.1053/j.gastro.2012.06.001 [ Links ]
10. Rutter MD, Chattree A, Barbour JA, et al. British Society of Gastroenterology/Association of Coloproctologists of Great Britain and Ireland guidelines for the management of large non-pedunculated colorectal polyps. Gut 2015;(Agree Ii):1-27. DOI: 10.1136/gutjnl-2015-309576 [ Links ]
11. Gupta S, Miskovic D, Bhandari P, et al. A novel method for determining the difficulty of colonoscopic polypectomy. Frontline Gastroenterol 2013;4(4):244-8. DOI: 10.1136/flgastro-2013-100331 [ Links ]
12. Sánchez del Río A, Campo R, Llach J, et al. Variation among endoscopy units in the achievement of the standards of colonoscopic performance indicators. Hepatogastroenterology 2008;55(86-87):1594-9. [ Links ]
13. Clark BT, Protiva P, Nagar A, et al. Quantification of adequate bowel preparation for screening or surveillance colonoscopy in men. Gastroenterology 2015;150(2):396-405. DOI: 10.1053/j.gastro.2015.09.041 [ Links ]
14. Clark BT, Rustagi T, Laine L. What level of bowel prep quality requires early repeat colonoscopy: systematic review and meta-analysis of the impact of preparation quality on adenoma detection rate. Am J Gastroenterol 2014;109(11):1714-23;quiz 1724. DOI: 10.1038/ajg.2014.232 [ Links ]
15. Saltzman JR, Cash BD, Pasha SF, et al. Bowel preparation before colonoscopy. Gastrointest Endosc 2015;81(4):781-94. DOI: 10.1016/j.gie.2014.09.048 [ Links ]
16. Hassan C, Bretthauer M, Kaminski MF, et al. Bowel preparation for colonoscopy: European Society of Digestive Endoscopy (ESGE) Guideline. Endoscopy 2013;(3):142-50. DOI: 10.1055/s-0032-1326186 [ Links ]
17. Johnson DA, Barkun AN, Cohen LB, et al. Optimizing adequacy of bowel cleansing for colonoscopy: recommendations from the U.S. Multi-Society Task Force on Colorectal Cancer. Gastrointest Endosc 2015;80(4):543-62. DOI: 10.1016/j.gie.2014.08.002 [ Links ]
18. Martel M, Barkun AN, Menard C, et al. Split-dose preparations are superior to day-before bowel cleansing regimens: a meta-analysis. Gastroenterology 2015;149(1):79-88. DOI: 10.1053/j.gastro.2015.04.004 [ Links ]
19. Connor A, Tolan D, Hughes S, et al. Consensus guidelines for the safe prescription and administration of oral bowel-cleansing agents. Gut 2012;61(11):1525-32. DOI: 10.1136/gutjnl-2011-300861 [ Links ]
20. Rex DK, Schoenfeld PS, Cohen J, et al. Quality indicators for colonoscopy. Gastrointest Endosc 2015;81(1):31-53. DOI: 10.1016/j.gie.2014.07.058 [ Links ]
21. Rembacken B, Hassan C, Riemann JF, et al. Quality in screening colonoscopy: position statement of the European Society of Gastrointestinal Endoscopy (ESGE). Endoscopy 2012;957-68. DOI: 10.1055/s-0032-1325686 [ Links ]
22. Parmar R, Martel M, Rostom A, et al. Validated scales for colon cleansing: a systematic review. Am J Gastroenterol 2016;111(2):197-204. DOI: 10.1038/ajg.2015.417 [ Links ]
23. Lai EJ, Calderwood AH, Doros G, et al. The Boston bowel preparation scale: a valid and reliable instrument for colonoscopy-oriented research. Gastrointest Endosc 2009;69(3 Pt 2):620-5. DOI: 10.1016/j.gie.2008.05.057 [ Links ]
24. Baxter NN, Sutradhar R, Forbes SS, et al. Analysis of administrative data finds endoscopist quality measures associated with postcolonoscopy colorectal cancer. Gastroenterology 2011;140(1):65-72. DOI: 10.1053/j.gastro.2010.09.006 [ Links ]
25. Sánchez del Río A, Baudet JS, Naranjo Rodríguez A, et al. Desarrollo y validación de indicadores y estándares de calidad en colonoscopia. Med Clin (Barc) 2010;134(2):49-56. DOI: 10.1016/j.medcli.2009.07.047 [ Links ]
26. Coriat R, Lecler A, Lamarque D, et al. Quality indicators for colonoscopy procedures: A prospective multicentre method for endoscopy units. PLoS One 2012;7(4). DOI: 10.1371/journal.pone.0033957 [ Links ]
27. Shah HA, Paszat LF, Saskin R, et al. Factors associated with incomplete colonoscopy: a population-based study. Gastroenterology 2007;132(7):2297-303. DOI: 10.1053/j.gastro.2007.03.032 [ Links ]
28. Gavin DR, Valori RM, Anderson JT, et al. The national colonoscopy audit: a nationwide assessment of the quality and safety of colonoscopy in the UK. Gut 2013;62(2):242-9. DOI: 10.1136/gutjnl-2011-301848 [ Links ]
29. Valori R, Rey J-F, Atkin W, et al. European guidelines for quality assurance in colorectal cancer screening and diagnosis. First edition - Quality assurance in endoscopy in colorectal cancer screening and diagnosis. Endoscopy 2012;44(S 03):SE88-SE105. DOI: 10.1055/s-0032-1309795 [ Links ]
30. Jover R, Herraiz M, Alarcón O, et al. Clinical practice guidelines: quality of colonoscopy in colorectal cancer screening. Endoscopy 2012;44(4):444-51. DOI: 10.1055/s-0032-1306690 [ Links ]
31. Rey J-F, Lambert R. ESGE recommendations for quality control in gastrointestinal endoscopy: guidelines for image documentation in upper and lower GI endoscopy. Endoscopy 2001;33(10):901-3. DOI: 10.1055/s-2001-42537 [ Links ]
32. Steele RJ, Pox C, Kuipers EJ, et al. International Agency for Research on C. European guidelines for quality assurance in colorectal cancer screening and diagnosis. First edition - Management of lesions detected in colorectal cancer screening. Endoscopy 2012;44(Suppl 3):SE140-50. DOI: 10.1055/s-0032-1309802 [ Links ]
33. Shaukat A, Rector TS, Church TR, et al. Longer Withdrawal time is associated with a reduced incidence of interval cancer after screening colonoscopy. Gastroenterology 2015;149(4):952-7. DOI: 10.1053/j.gastro.2015.06.044 [ Links ]
34. Hilsden RJ, Dube C, Heitman SJ, et al. The association of colonoscopy quality indicators with the detection of screen-relevant lesions, adverse events, and postcolonoscopy cancers in an asymptomatic Canadian colorectal cancer screening population. Gastrointest Endosc 2015;82(5):887-94. DOI: 10.1016/j.gie.2015.03.1914 [ Links ]
35. Rex DK, Schoenfeld PS, Cohen J, et al. Quality indicators for colonoscopy. Gastrointest Endosc 2015;81(1):31-53. DOI: 10.1016/j.gie.2014.07.058 [ Links ]
36. Barclay RL, Vicari JJ, Greenlaw RL. Effect of a time-dependent colonoscopic withdrawal protocol on adenoma detection during screening colonoscopy. Clin Gastroenterol Hepatol 2008;6(10):1091-8. DOI: 10.1016/j.cgh.2008.04.018 [ Links ]
37. Moritz V, Bretthauer M, Ruud HK, et al. Withdrawal time as a quality indicator for colonoscopy - A nationwide analysis. Endoscopy 2012;44(5):476-81. DOI: 10.1055/s-0032-1306898 [ Links ]
38. Gellad ZF, Weiss DG, Ahnen DJ, et al. Colonoscopy withdrawal time and risk of neoplasia at 5 years: results from VA Cooperative Studies Program 380. Am J Gastroenterol 2010;105(8):1746-52. DOI: 10.1038/ajg.2010.107 [ Links ]
39. Lieberman DA, Faigel DO, Logan JR, et al. Assessment of the quality of colonoscopy reports: results from a multicenter consortium. Gastrointest Endosc 2009;69(3 Pt 2):645-53. DOI: 10.1016/j.gie.2008.08.034 [ Links ]
40. Nayor J, Saltzman JR, Campbell EJ, et al. Impact of physician compliance with colonoscopy surveillance guidelines on interval colorectal cancer. Gastrointest Endosc 2017;85(6):1263-70. DOI: 10.1016/j.gie.2016.10.041 [ Links ]
41. Nishihara R, Wu K, Lochhead P, et al. Long-term colorectal-cancer incidence and mortality after lower endoscopy. N Engl J Med 2013;369(12):1095-105. DOI: 10.1056/NEJMoa1301969 [ Links ]
42. Morris EJA, Rutter MD, Finan PJ, et al. Post-colonoscopy colorectal cancer (PCCRC) rates vary considerably depending on the method used to calculate them: a retrospective observational population-based study of PCCRC in the English National Health Service. Gut 2014;64(8):1248-56. DOI: 10.1136/gutjnl-2014-308362 [ Links ]
43. Bressler B, Paszat LF, Chen Z, et al. Rates of new or missed colorectal cancers after colonoscopy and their risk factors: a population-based analysis. Gastroenterology 2007;132(1):96-102. DOI: 10.1053/j.gastro.2006.10.027 [ Links ]
44. Brenner H, Chang-Claude J, Seiler CM, et al. Protection from colorectal cancer after colonoscopy: a population-based, case-control study. Ann Intern Med 2011;154(1):22-30. [ Links ]
45. Singh S, Singh PP, Murad MH, et al. Prevalence, risk factors, and outcomes of interval colorectal cancers: a systematic review and meta-analysis. Am J Gastroenterol 2014;109(9):1375-89. DOI: 10.1038/ajg.2014.171 [ Links ]
46. Samadder NJ, Curtin K, Tuohy TMF, et al. Characteristics of missed or interval colorectal cancer and patient survival: a population-based study. Gastroenterology 2014;146(4). DOI: 10.1053/j.gastro.2014.01.013 [ Links ]
47. 4Institute of Medicine. Crossing the quality chasm: a new health system for the 21st century. Washington DC: National Academy Press; 2001. [ Links ]
48. Jain D, Momeni M, Krishnaiah M, et al. Importance of reporting segmental bowel preparation scores during colonoscopy in clinical practice. World J Gastroenterol 2015;21(13):3994-9. DOI: 10.3748/wjg.v21.i13.3994 [ Links ]
49. Corley DA, Jensen CD, Marks AR, et al. Adenoma detection rate and risk of colorectal cancer and death. N Engl J Med 2014;370(14):1298-306. DOI: 10.1056/NEJMoa1309086 [ Links ]
50. Kaminski MF, Regula J, Kraszewska E, et al. Quality indicators for colonoscopy and the risk of interval cancer. N Engl J Med 2010;362(19):1795-803. DOI: 10.1056/NEJMoa0907667 [ Links ]
51. Denis B, Sauleau EA, Gendre I, et al. The mean number of adenomas per procedure should become the gold standard to measure the neoplasia yield of colonoscopy: a population-based cohort study. Dig Liver Dis 2014;46(2):176-81. DOI: 10.1016/j.dld.2013.08.129 [ Links ]
52. Wang HS, Pisegna J, Modi R, et al. Adenoma detection rate is necessary but insufficient for distinguishing high versus low endoscopist performance. Gastrointest Endosc 2013;77(1):71-8. DOI: 10.1016/j.gie.2012. 08.038 [ Links ]
Received: December 11, 2017; Accepted: February 13, 2018











 texto en
texto en