INTRODUCTION
Oxygen therapy consists of administering oxygen at a higher concentration than atmospheric. These concentration values are directly proportional to the altitude. Its indications are varied, having an essential role in both acute and chronic pathologies. The main objectives of oxygen therapy are: a) to treat and/or prevent hypoxemia; b) to decrease myocardial demand; c) to decrease respiratory effort, and d) to treat pulmonary hypertension [1]. Likewise, it is important to define several concepts to know their indications. The arterial partial pressure of oxygen (PaO.) is the amount of dissolved oxygen in the blood. Its normal values are between 70-90 mmHg, PaO2 is related to alveolar ventilation, ventilation/perfusion ratio, and the fraction of oxygen in the inspired air (FiO.). Normal PaO. values are approximately equivalent to arterial oxygen saturation (SatO2), which is defined as the arterial fraction of hemoglobin bound to oxygen [1], of 95-100%.
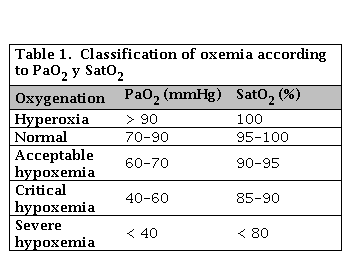
Once these basic concepts have been defined, it is necessary to clarify the difference between hypoxemia and hypoxia. Hypoxemia is defined as the decrease in PaO. below 80 mmHg. On the other hand, hypoxia is described as a decrease in tissue oxygenation [2]. Considering PaO2 values, different levels of hypoxemia can be defined, as shown in Table 1.
RESPIRATORY PHYSIOLOGY
The primary function of the lung parenchyma is to supply oxygen to the arterial blood and to remove carbon dioxide from the mixed venous blood of the pulmonary artery produced by cellular metabolism and gas exchange [3]. PaO2 determines arterial oxygenation, while PaCO2 (arterial partial pressure of carbon dioxide) indicates carbon dioxide levels dissolved in the arteries (34-38 mmHg under standard conditions). Adequate gas exchange is measured by several factors, including control of ventilation (V), alveolar ventilation (VA), alveolar-capillary diffusion, and pulmonary perfusion (Q) [4]. The deterioration of any of them or the incorrect coordination between them will determine respiratory failure situations with or without hypercapnia [5].
In each respiratory cycle, approximately 500 ml are mobilized, the inspired volume being slightly higher (1%) than the exhaled volume due to the amount of oxygen being inhaled is greater than the carbon dioxide that is exhaled. When talking about ventilation, the effective measure is VA, since V also encompasses the dead space (DV) [6]. The dead space is the anatomical (trachea and large airways) and the alveolar dead space, which increases with age. The DV represents 1/3 of the total V [5, 6].
After ventilation, gas exchange occurs; this is determined by alveolar-capillary diffusion [7], which is based on a pressure system that mobilizes air from the areas of higher pressure to those of lower pressure. Gas diffusion depends on several factors:
The distance to be traveled by gas molecules
The exchange surface
The partial pressure of the gas on each side of the interface (pressure gradient)
Gas diffusibility [8]
Once the gas exchange has been carried out, the O2 and CO2 are dissolved in the plasma or bound to hemoglobin [9]. Under normal conditions, pulmonary O2 transfer is limited by perfusion, and only in very particular situations (such as extreme exercise) is this transfer limited by diffusion [10]. VA and Q are higher at the bases than at the vertices. In addition, the VA/Q gradient differs according to anatomical location, being higher at the vertices (> 3) than at the bases (< 1). This gradient is because the VA is much higher than Q at the vertices [11]. An important concept is the arterial alveolus difference or gradient of O2 (AaPO2), which is understood as the difference between PAO2 and PaO2. Its levels reflect the efficiency of the lung as a gas exchanger, orienting us on the possible mechanism of alterations in gas exchange [12].
HYPOXEMIA AND RESPIRATORY INSUFFICIENCY
Hypoxemia (PaO2 < 70 mmHg) and respiratory failure may be due to various mechanics with different clinical significance (Table 2) [13]. The onset of respiratory failure may be acute or chronic, and the appearance of compensation mechanisms is possible if the onset is carried out more gradually [13]. The diagnosis of respiratory failure is made by arterial blood gas and, according to CO2 levels, it is classified as type I (without hypercapnia) or type II (with hypercapnia) (Table 3) [14].
Table 2. Pathophysiological mechanisms of respiratory failure
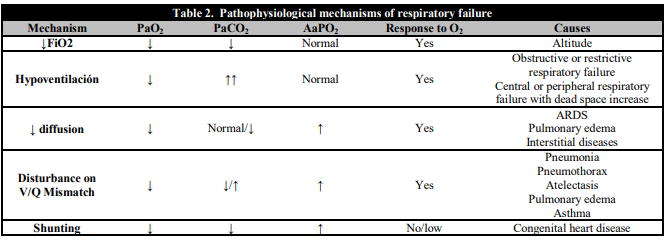
PaO2: Arterial partial pressure of oxygen; AaPO2: Arterial alveolus difference or gradient of O2; FiO2: Fraction of inspired oxygen in the air; V/Q: Ventilation/perfusion ratio. ARDS: Adult respiratory distress syndrome.
HYPOXIA CLINICAL MANIFESTATIONS
Hypoxia symptoms are nonspecific and related to the causative disease and the levels of oxygen and carbon dioxide. Therefore cases of sudden onset hypoxemia produce more significant symptomatology since compensation mechanisms are not generated [15]. Among the clinical manifestations is an increase in respiratory effort, peripheral cyanosis, and neurological involvement, which varies from lack of concentration and apathy (chronic onset) to clouding and stupor (acute symptoms) [16]. Furthermore, hypercapnia causes disorientation, motor agitation, facial diaphoresis, headache, clouding, and even coma in the abrupt symptoms, whereas, in the most chronic symptoms, only mild drowsiness. [17]. Table 4 lists the most characteristic clinical signs.
As we have already mentioned, in chronic hypoxia symptoms, compensation mechanisms are generated. These are based on the increase in heart rate and cardiac output, thus improving the V/Q ratio through an improved blood flow through pulmonary vasculature [18]. On the other hand, an increase in the renal secretion of erythropoietin causes polycythemia that increases the oxygen-carrying capacity of the blood [19]. However, these mechanisms are harmful in the medium-long term, since pulmonary vasoconstriction generates irreversible anatomical alterations such as hypertrophy of the middle muscular layer of the pulmonary arterioles and fibrosis of the intima, which leads to pulmonary hypertension and right heart failure [20]. All this is favored by the increase in cardiac output and polycythemia, producing an elevation in myocardial demand and higher energy consumption, which can cause a malnutrition status that favors the appearance of some general manifestations [21].
INDICATIONS AND BENEFITS OF OXYGEN THERAPY
Primary objective of oxygen therapy is the prevention or reversal of the consequences of hypoxemia, ultimately improving tissue oxygenation. Over time, several studies have been carried out to verify its benefits in different pathologies, highlighting studies in chronic obstructive pulmonary disease (COPD) [22, 23]. Both studies established that mortality decreased when giving oxygen for 15 hours a day compared to not giving it for that period. It further decreased when applying a continuous regimen of 17.7 hours/day. Based on these results, the criteria for the indication of continuous home oxygen therapy were defined; based on: PaO2 < 55 mmHg, or between 55 and 60 mmHg with the presence of peripheral edema, cor pulmonale, hematocrit values above 55%, stocks of cardiac arrhythmias and mental or central nervous system disorders that could be attributed to hypoxemia [24].
In the presence of these results, the use of oxygen therapy has spread to other pathologies that present with chronic respiratory failure with a pattern similar to COPD. However, no improvement in survival has been observed in patients with idiopathic pulmonary fibrosis [25], pulmonary hypertension [26], cystic fibrosis [27], or heart failure [28]. Despite this, it is recommended to consider oxygen therapy in these entities in the presence of severe hypoxemia (PaO2 < 60 mmHg).
Taking into account this point of general indication, we can assess acute or chronic situations in need of treatment with oxygen therapy. Among the acute situations, the most recognizable are the situations of arterial hypoxemia, imbalances in the V/Q, short-circuits or right-left shunts, and decreased FiO2 in ambient air (high altitudes). Likewise, other symptoms are observed where there may be a PaO2 > 60 mmHg with a significant deterioration of the tissue supply, such as in situations of low expenditure secondary to cardiogenic shock, in acute anemia or carbon monoxide poisoning (competes in binding to hemoglobin) [29, 30].
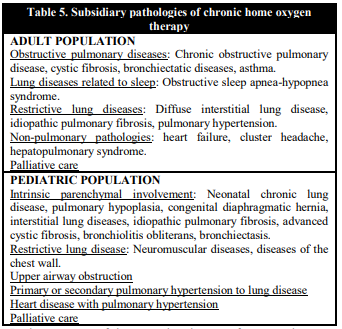
The chronic use of oxygen therapy differs depending on whether the pediatric or adult population is involved. It should be noted that childhood pathologies are different, and it may be necessary to use it temporarily, although, if needed, it is required both at home and when traveling. Its indication is related to development, which is why its prognosis is better than in adulthood. In addition, it may be necessary only for night-time use, a different criterion than that of the adult population (> 15 h daily) [29, 31]. Table 5 summarizes the most prevalent pathologies in adults and pediatric populations in which chronic oxygen therapy is indicated.
OXYGEN THERAPY SYSTEMS
The different oxygen administration formats are classified according to the flow that reaches the patient, differentiating high and low flow systems [32].
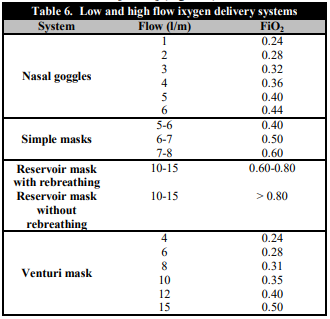
Low flow systems are characterized by not supplying all the air required by the inspiratory flow demand, which is why the use of ambient air, in addition to the air provided by the system, is required to satisfy this need [33]. Within this group, we find nasal goggles, simple masks, and reservoir masks (Figure 1). The most important feature of these systems is that they do not ensure stable levels of FiO2, achieving variable oxygen concentrations in the inspired air depending on the flow of oxygen supplied and the ventilatory pattern of each patient (Table 6) [34].
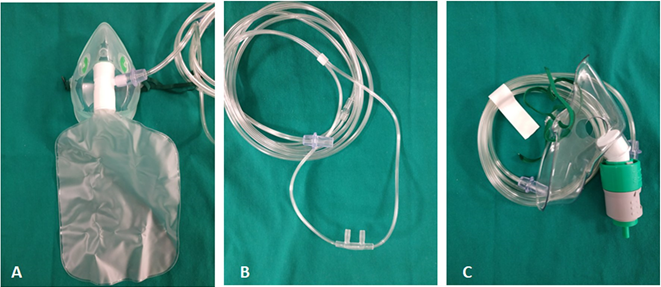
Figure 1. Oxygen therapy systems low flow systems like reservoir masks (A), nasal goggles (B) as well as high flow systems (Venturi mask, C)
On the other hand, high flow systems provide the patient with inspiratory flows higher than their peak demand; that is, the system provides all the inspired air. In this way, they allow us to keep FiO2 constant independently of the patient's ventilatory pattern. Within this group are the Venturi mask and high flow oxygen therapy systems and active humidification [33, 35] (Figure 1).
HIGH FLOW OXYGEN THERAPY (HFO)
High flow oxygen therapy systems provides to the patient an adjustable air and oxygen mixture. It is a system with increasing use in recent years due to its excellent tolerance and ease of use. It consists on the administration of high oxygen flows through nasal cannulas. To promote tolerance in patients, this air is administered humidified (humidity between 95-100%) and hots (34-40 °C). The benefits of this system are due to the humidification and temperature of the gas itself, which reduces metabolic expenditure, improves compliance and lung elasticity, and facilitates tolerance and comfort. Furthermore, other advantages of these systems are that they decrease the nasopharyngeal dead space, decrease the inspiratory resistance and provide a certain degree of pulmonary distension pressure (CPAP), which is neither measurable nor adjustable, but which has numerous benefits (decreases atelectasis, improves the V/Q ratio and in premature patients reduces apnea).
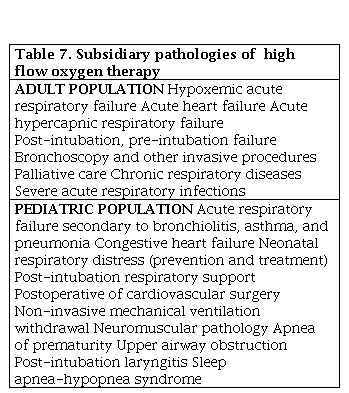
According to age, the limits that define high-flow oxygen therapy are 1-8 lpm in newborns, in children 1-2 lpm/kg, and more than 30 lpm in adults [33]. The indications for this system have increased in recent years, being accepted generically in cases of moderate acute respiratory failure or in cases of hypoxemia that do not respond to conventional oxygen treatment. Table 7 shows pathologies in children and adults in which the HFO has shown utility.
Contraindications are few, limited to anatomical causes (nasal obstruction or choanal atresia, trauma or surgery in the nasopharynx, etc.) or severe respiratory failure requiring mechanical ventilation. Caution is also recommended in the use of this type of system in cases of hypoxemia with hypercapnia since the use of oxygen may cancel the respiratory stimulus generated by hypoxia [33].