INTRODUCTION
Bladder carcinoma is the 7th most common malignancies in the world and is also the second most common neoplasm of the genitourinary system after prostatic cancer. In Bulgaria, bladder cancer (BC) ranks 18th in frequency [1, 2]
According to literature data, men have a risk of developing BC from 0.45% to 2.81% before the age of 60 and women from 0.14% to 0.82%. After the age of 75, this risk increases equally in both sexes [3]. In the etiopathogenetic aspect, numerous factors are listed that have a direct impact on the process of carcinogenesis, with smoking and chemical carcinogenesis taking the lead. Such are arylamines, benzidines, 4-aminobenzene, 2-naphthylamine, and others, which act as pro-carcinogens. Like bilirubin, the compounds listed herein are conjugated to glucuronic acid in the liver and excreted in the urine in which they are activated [4]. In Chile, Argentina, and China, the impact of arsenic is also indicated as a risk factor. In some countries of the Middle East and parts of Africa, the widespread infections caused by parasites - trematodes of the genus Schistosomes also lead to an increased risk of bladder neoplasms, 75% of which are squamous cell carcinomas [5, 6].
HISTORICAL NOTES
In the last few years, with the entry of new trends in oncology, innovative therapeutic approaches in the treatment of patients with urothelial BC, special attention is paid to the detailed pathomorphological description of the presence and severity of the stromal reaction. The stromal response in neoplastic diseases is part of the adaptive immune response, and it helps to understand the immunobiology of the tumor microenvironment. At the same time, it is a predictive indicator of overall patient survival.
The history of immunotherapy began in 1921, when French scientists Albert Calmett and Camille Guerin first discovered the Bacillus Calmette-Guérin tuberculosis vaccine (BCG, Mycobacterium bovis) at the Pastor Institute in Lille, France. Eventually, Old, Clark, and Benaceraf, intrigued by Pearl's hypothesis that "patients with tuberculosis have a lower incidence of tumors," conducted a series of experimental studies in the 1950s and concluded that BCG administration also prevented the growth of experimental tumors [9].
Twenty years later Mate et al. observed a clinical outcome with BCG treatment in a leukemia patient, while, Morton et al. demonstrate the positive effect of BCG against human malignant melanoma [11]. Ten years later, in 1975, Bloomberg et al. observed severe inflammatory reactions in the healthy bladder of dogs after local injection of BCG [12]. However, a year later, Morales et al. reported the first successful intravesical use of BCG for the treatment of bladder carcinomas. Since then, BCG has become the gold standard for the treatment of non-muscular invasive bladder cancer (NMIBC) [13]. Intravesical therapy with BCG has been shown to reduce the recurrence rate and the risk of progression to invasive muscle disease. Patients with high-grade muscular-invasive urothelial bladder carcinoma treated with BCG after transurethral resection of a bladder tumor (TURBT) have lower levels of cystectomy. BCG also prevents recurrence of the disease in 60% to 100% of patients in one year and in 55% to 75% in two years.
Then, in 1980, Lam and colleagues completed a comparative study between standard surgical therapy for NMIBC and standard therapy combined with adjunctive BCG [14]. Furthermore, Brosman et al. [15] modified Morales' intravesical regimen of BCG [13], and in 1990 BCG was approved by the Food and Drug Administration for the treatment of in situ bladder cancer (CIS), non-invasive BC (pTa) and BC with invasion of the lamina propria (pT1) [16].
Therapy with BCG induces a chronic nonspecific inflammatory reaction with influx mainly of CD8 T lymphocytes (CD8 cells) and CD4 T lymphocytes (CD4 cells), as well as macrophages and production of cytokines in the bladder mucosa that leads to an immune response against tumor cells [7]. Long-term follow-up studies have been performed of the urine of patients treated with BCG and revealed elevated amounts of IL-1, -2, -6, -8 and -12, TNF, INF-γ, and Granulocyte-macrophage colony-stimulating factor (GM-CSF) [7]. Also, the Alhunaidi el al. study revealed that bladder tumor cell killing involves immune-mediated cytotoxicity, including NK cells, NK T cells, CD8 T cells, macrophages, and TRAIL (granulocytes) among many others [7]. They have previously investigated the immunologically active components of BCG in the therapy of BC and observed that numerous BCG subcomponents provide positive stimuli for CD 4 cell differentiation and enhance the cytotoxicity against bladder tumor cells [17].
Here, we present low grade urothelial carcinoma of the bladder before and after application of BCG (Figure 1).
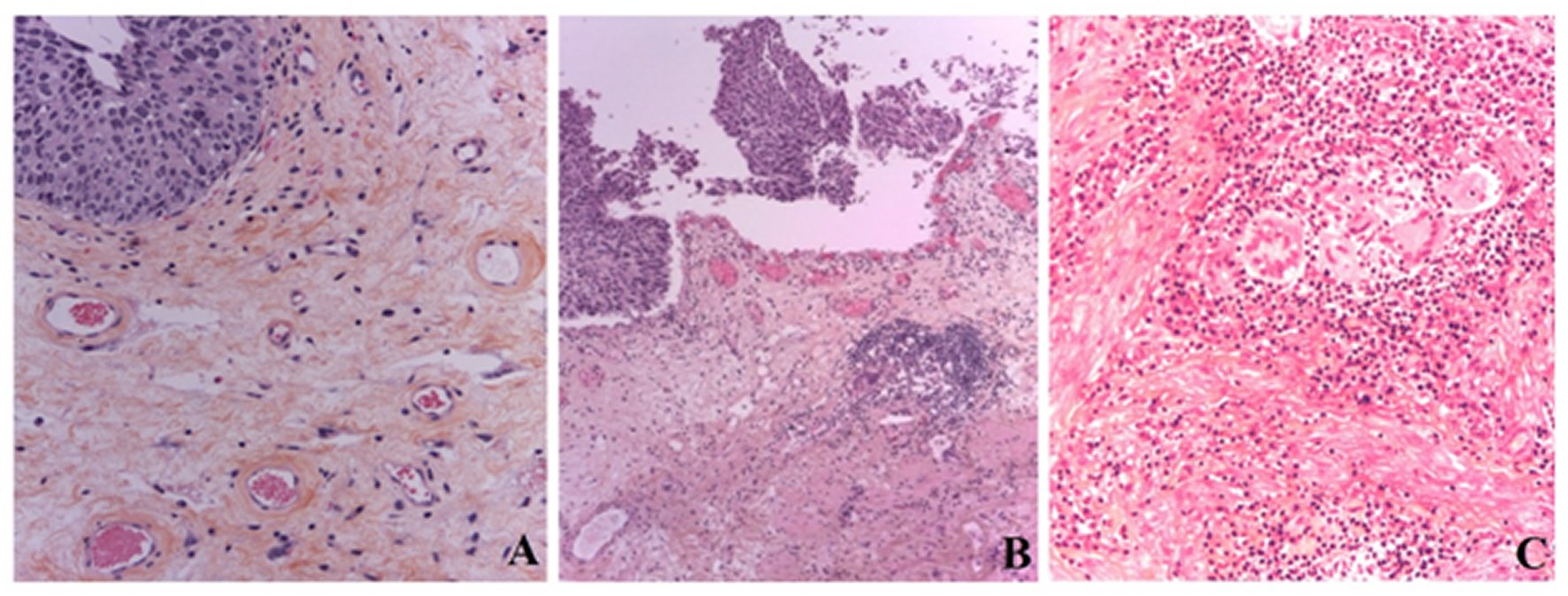
Figure 1. Low grade urothelial carcinoma of the bladder before (A) and after (B and C) application of BCG. (A). with scanty lymphocytic infiltration in lamina propria; with single, isolated giant multinucleated cells of stromal origin, without formation of granulomas, which reflecting chronic non-specific irritation of mucosa. After application of BCG-therapy, it was also revealed formation of tertiary lymphoid structure (TLS) (→) (B), also, in the mucous lamina propria was observed pronounced lymphocytic infiltration with epithelioid cell and giant cell granulomas of tuberculoid type; the giant multinucleated cells are Langhans type of monocyte origin, grouped in granulomas and reflect an Type IV hypersensitivity reaction (C);. H&E x200; x100; x400.
Since 1990, the therapeutic approach has included the combined use of Methotrexate, Vinblastine, Adriamycin, and Cisplatin [18]. At present, chemotherapy is the mainstay of treatment for patients with invasive urothelial BCs, with platinum compounds being the mainstay of many therapeutic regimens of oncotherapy. However, chemotherapy, in patients with metastatic disease, remains the only palliative and the median survival rarely exceeds 12 months [19]
The modest success of studies on the human body's immune response to tumor cells and the introduction of immunotherapy represent the first significant improvements in the treatment of these patients in decades [20].
In 2016, the United States Food and Drug Administration (FDA) and in 2017, European Medicines Agency (EMA) introduced a checkpoint inhibitor as a first- and second-line therapeutic agent for the treatment of advanced BCs. Today, when oncology focuses on personalized therapy, identifying cases of treatment with checkpoint inhibitors is crucial. The latter is done by immunohistochemical examination of PD-L1 expression on tumor cells and tumor-infiltrating immune cells. This identifies PD-L1 as a predictive tissue biomarker for research and to restore the potential for antitumor immunotherapy in patients with invasive urothelial BC [21].
CONCLUSION
The last few years have seen an extremely rapid expansion of our knowledge of immune regulation based on the underlying principles established in laboratory models of infection, autoimmunity, and transplantation. It is these principles and knowledge that can be applied to the study of the restoration of antitumor immunity and the establishment of immunotherapy with checkpoint inhibitors as a viable treatment for patients with advanced urothelial BC. It should be borne in mind that in clinical practice, treatment and prognosis decisions are guided and directly dependent on histopathological assessment and directly related immunohistochemistry. Immunohistochemistry and subsequent personalized immunotherapy with checkpoint inhibitors "open doors" for the development of new generations of immune modulators that would be most effective in combination. There is only one goal - to prolong the patient's life.














