INTRODUCTION
Diabetes mellitus (DM) is a chronic metabolic disorder caused by dysfunction of the β-cells of the pancreatic islets that induces elevated plasma glucose levels. This disease affects more than 425 million people around the world without predilection for either sex. Two types of diabetes mellitus have been described: type 1 and 2. Type 2 DM is the most common, accounting for 90% of cases and is mainly caused by a lifestyle with high-calorie diets, low physical activity, or smoking [1]. Diabetic patients are more susceptible to infections, especially fungal infections. Oral manifestations in diabetics include a higher prevalence and severity of both dental caries and periodontal disease, salivary flow dysfunction, impaired healing, and opportunistic infections. Patients with diabetes mellitus are more prone to fungal infections, probably due to immune disturbance and salivary composition changes. Considering fungal infections, diabetics show a higher prevalence of oral lesions associated with Candida infection, especially denture stomatitis, pseudomembranous candidiasis, median rhomboid glossitis and angular cheilitis. Moreover, diabetics usually have systemic medications that favor the reduction of salivary flow, facilitating the proliferation of microorganisms in oral biofilms. Approximately 30% of diabetics have an oral yeast infection at some point in their life [2]. The main microorganism of oral candidiasis is Candida albicans, a polymorphic fungus with the ability to grow in the form of hyphae that colonize and invade the tissues. There is a direct relationship between the increase in blood glucose levels and the number of Candida hyphae in the oral mucosa [3]. This study aimed to evaluate oral candidiasis and the different Candida species found in patients with and without diabetes mellitus.
METHODS
A search for studies on oral candidiasis and diabetes mellitus was conducted in the following databases: PubMed (MEDLINE, Cochrane Library), Web of Science (WoS) and Google Scholar. Search strategies were developed for each database with a combination of Medical Subjects Headings (MeSH) terms and free text terms. The search terms were: "candidiasis, oral" [MeSH Terms] AND "diabetes mellitus" [MeSH Terms]; "oral candid*" AND "diabet *"; allintitle: "oral" ("candidiasis" OR "candida") ("diabetes" OR "diabetic"). After this initial search, 481 articles (145 in PubMed, 241 in WoS and 95 in Google Scholar) were found between 1967 and 2020; 173 of them duplicates, leaving 308 articles for elegibility. Two researchers (ARA and CPR) examined the titles and abstracts of the articles independently, and later, both selected the papers that were included in this meta-analysis. The inclusion criterion was subjects of any age with a diagnosis of either type 1 or type 2 diabetes mellitus. The exclusion criteria were: a) articles without full-text availability (n = 174), b) articles with a score below 6 stars from a maximum of 9 on the Newcastle–Ottawa methodological quality assessment scale [4] (n = 52), and c) studies with non-usable data (n = 57). Finally, twenty-five studies were included in this meta-analysis (Figure 1).
STATISTICAL ANALYSIS
Data were processed with the RevMan 5.4 meta-analysis software (The Cochrane Collaboration, Oxford, UK). For dichotomous outcomes, the odds ratio (OR) with the Mantel-Haenszel Chi-square formula (M-H) and 95% confidence interval (95% CI) was used. Heterogeneity was determined according to the P values and the Higgins statistic (I2%). In cases of high heterogeneity (I2 > 50%), the random-effects model was applied. Pearson Chi-square test with Fisher's exact test was also used when required. Tables and a forest plot graph were used to present the results. The minimum level of significance was set at p < 0.05.
RESULTS
Only articles with low to moderate risk of bias (≥6 stars from a maximum of 9 stars) according to the Newcastle-Ottawa (NOS) quality scale [4], were considered in this study.
Table 1 presents the twenty-five studies [5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29] that reported oral Candida species detection in patients with and without diabetes mellitus, considering the different Candida detection methods used. Oral Candida species were detected in 1453 (47.3%) of 3073 diabetics (range: 16.4% [28] – 83.7% [16]) and 488 (25.0%) of 1953 non-diabetics (range: 4.0% [23] – 81.4% [25]).
Table 1. Characteristic of the patients and effects of diphenhydramine ointment
Ref: References; NOS: Newcastle-Ottawa quality scale; n/N: number of Candida positive cases/total number of cases; (%): Percentage of positive cases; USA: United States of America; SDA: Sabouraud's dextrose agar; CAC: CHROMagar Candida; PAS: Periodic Acid-Schiff stain; PCR: Polymerase chain reaction.
Twenty-two studies [5-11, 13, 15-25, 27-29] assessed the oral detection of Candida species in patients with and without diabetes mellitus (Figure 2). Diabetic patients were 3.16 times more likely to have Candida species in their oral microbiota, finding highly significant statistical differences (OR = 3.16; 95% CI: 2.21 to 4.52; p < 0.001).
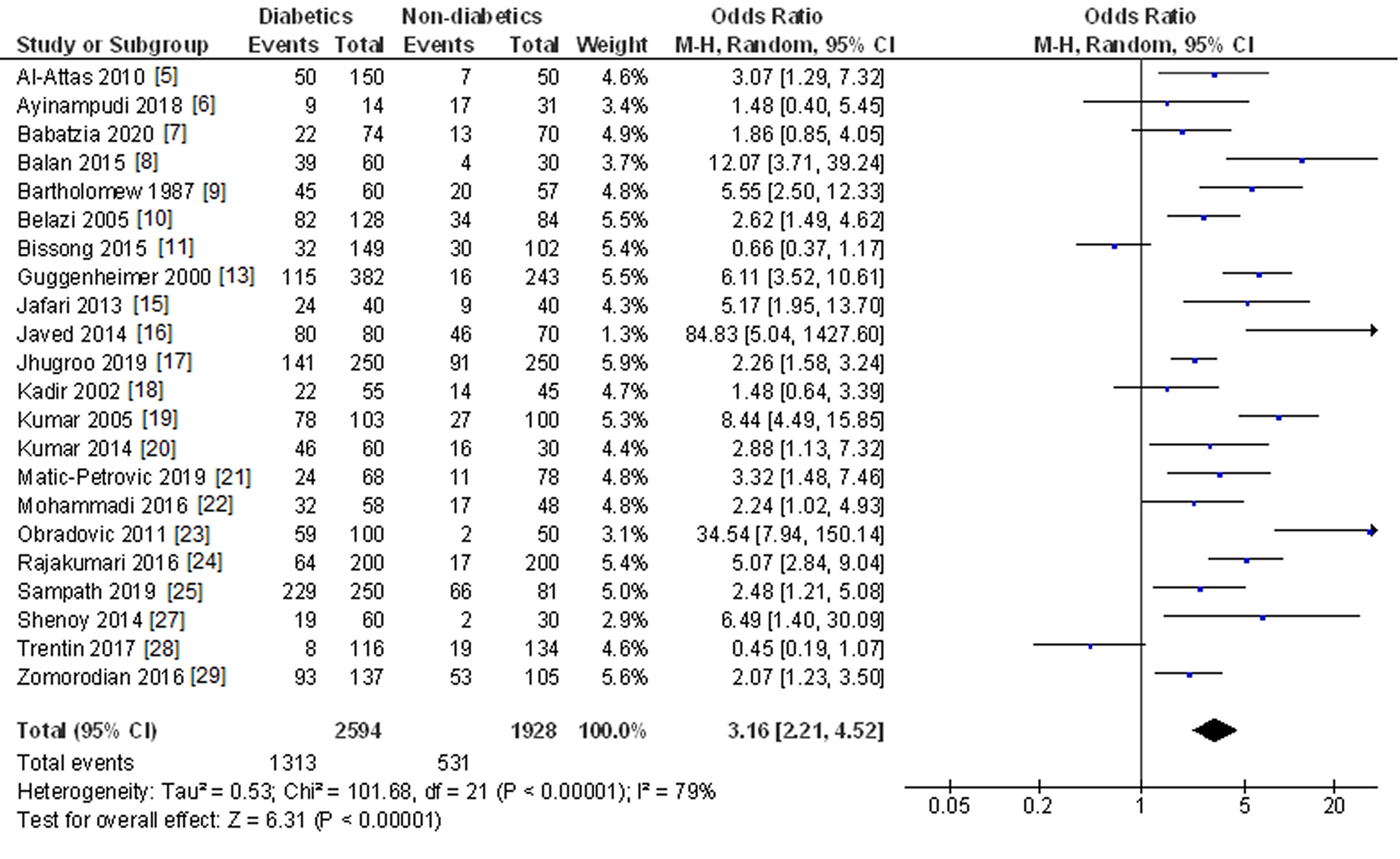
Figure 2. Study data and forest plot graph for the Candida species detection in subjects with and without diabetes mellitus.
The main risk factors related to oral Candida infection in patients with diabetes mellitus are shown in Table 2. Eight studies [10, 12, 14, 15, 19, 23, 25, 26] examined the possible influence of gender on the probability of Candida oral infection. Gender did not affect to fungal infectionwith no statistically significant association 8OR = 1.40; 95% CI: 0.95 to 2.08; p = 0.09). In the case of the diaebetes mellitus type, four studies [5, 19, 27, 29] evaluated this paramenter, noticing a higher prevalence of oral candidiasis in type 1 diabetes patients, although without reaching statistical significance (OR = 1.55; 95%CI: 0.74 to 3.24; p = 0.24).
Table 2. Risk factors related to oral Candida infection in patients with diabetes mellitus (DM)
Ref: References; OR: Odds Ratio; [95%CI]: 95% confidence interval; I2: Higgins statistic for heterogeneity (percentage); *statistically significant.
Respect to glycemic control, five studies [7, 8, 14, 19, 20] pointed out that poor glycemic control increased 2.94 times the risk of oral Candida species infection, with highly significant statistical differences (OR = 2.94; 95% CI: 1.73 to 5.01; p < 0.001).
Other four studies [10, 14, 25, 26] also corroborated denture wearers were more than twice as likely to be infected with Candida species. In the statistical analysis, a highly significant association was found (OR = 2.22; 95% CI: 1.48 to 3.33; p < 0.001).
Table 3 displays the distribution of the different Candida species found in oral microbiota between diabetic and non-diabetic subjects. The most prevalent species was C. albicans in both diabetics (78.0%) and non-diabetics (71.2%), followed by C. tropicalis (9.4% in diabetics and 12.8% in non-diabetics) and, the least prevalent, C. kefyr (0.5 % in diabetics and 1.9% in non-diabetics). When the different Candida species are classified in two groups (C. albicans species and C. non-albicans species), non-albicans species were more frequent in non-diabetics (28.8%) than in diabetics (21.9%), with statistically significant differences (p = 0.04).
DISCUSSION
Data from twenty-five studies on oral candidiasis and Candida species detection in diabetics have been included in the present meta-analysis.
In this study, diabetic patients were 3.16 times more likely to have Candida species in their oral microbiota than non-diabetics, with a highly significant statistical relationship (p < 0.001). Of the 22 studies that studied this variable, 20 of them [5, 6, 7, 8, 9, 10, 13, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 27, 29] found a higher prevalence of Candida species detection in diabetics compared to the two studies [11, 28] that found oral Candida species more frequently in non-diabetics, although without statistical significance. Candida species have a predilection for colonizing the oral cavity, particularly in patients with diabetes mellitus, with percentages ranging between 60% and 80% of diabetics. Oral mucosa in diabetics provides a less hostile ecosystem for oral colonization by Candida species. This fact could be related to factors such as hyposialia, the use of dentures, the degree of glycemic control and the intake of drugs. In diabetics with poor metabolic control, an oral environment rich in sugars permits high levels of glucose in saliva and can contribute to the persistence of aciduric yeasts in the oral cavity. Moreover, carbohydrates in the diet may be a contributing factor, promoting adhesion, biofilm formation, and yeast colonization in the oral environment [25]. Some studies [11] report a lower frequency of oral candidiasis in diabetics because they do not establish well-defined diagnostic criteria and the diagnosis is not made by calibrated examiners or specialists. The diagnosis made by a calibrated examiner guarantees a correct evaluation of oral candidiasis, increasing the reliability of the clinical data obtained [28].
Gender did not condition the probability of Candida species oral infection or detection, with no statistically significant association (p = 0.09). Six studies (4 with statistically non-significant results [12, 14, 19, 23] and 2 with significant results [25, 26] that considered this factor, found a greater predisposition in the female gender. In contrast, two other studies [10, 15] observed a higher susceptibility to Candida infection in males. The association between gender and oral Candida species colonization is quite poorly defined with conflicting findings. Some studies [25] have observed that, in women with periodontitis, where inflammation of the oral mucosa is favored, there was a greater probability of fungal infection compared to men. In fact, Candida species colonization was observed in 88% of women and 73.1% of men. This increased fungi detection in women could be due to the hormonal changes observed in menopause that induces a series of changes in the oral and vaginal mucosa with a higher prone to candidiasis [26].
In the present study, the possible influence of diabetes mellitus (type 1 or type 2) type on the risk of suffering from candidiasis was also analyzed. A higher prevalence of oral candidiasis in patients with type 1 diabetes was found, although statistical significance was not achieved (p = 0.24). Of the four studies that analyzed this parameter, two [5, 19] observed a higher prevalence of type 1 diabetes compared to type 2, but only one [5] had statistical significance. On the other hand, the other two studies [27, 29] did not observe this higher prevalence in type 1 diabetes patients. Although oral candidal colonization appears to be greater in type 1 diabetics than in type 2, it has not been possible to establish a correlation between the rates of Candida species carriers, their concentration, the type of diabetes mellitus or antidiabetic drugs used [5]. Another possible explanation for this higher frequency in type 1 diabetics could lie in the resistance to some antifungals observed in these patients, in whom the treatment appears to be less effective [29].
In this study, a poor glycemic control increased 2.94 times the risk of Candida species oral infection with a highly significant statistical relationship (p < 0.001). All studies [7, 8, 14, 19, 20] that evaluated this variable were in favor of this inverse relationship between glycemic control and the Candida infection risk. Several studies [14, 20] have shown that poorly controlled diabetics or those without metabolic control are significantly more susceptible to having oral candidiasis. Salivary glucose forms chemically reversible glycosylation products with proteins in tissues during hyperglycemic episodes and this leads to accumulation of glycosylation products in oral epithelial cells, which in turn may increase the number of receptors available for Candida. This finding suggests the fact that uncontrolled or poorly controlled diabetes increases susceptibility to oral opportunistic infections, such as oral candidiasis [20]. Poor glycemic control determines higher mean glycosylated hemoglobin (HbA1c). This hyperglycemia could contribute to the risk of Candida oral infection by also increasing salivary glucose levels, and promoting the proliferation of Candida in the oral cavity. Furthermore, this conditions a decrease in salivary pH, creating an ideal environment for fungal growth [29].
Denture wearers' diabetics were more than twice as likely to also be Candida carriers compared to diabetics no denture wearers, with highly significant statistical differences (p < 0.001). The four studies [10, 14, 25, 26] that delved into this parameter confirmed this higher prevalence of Candida species in denture wearers. Candida species avidly bind and adhere to acrylic surfaces, and dentures may act as a reservoir for these organisms, forming a bacterial-fungal biofilm layer that cannot be easily eradicated. Wearing dentures promotes the Candida species growth beneath denture, with low oxygen levels, an anaerobic environment, and a low pH. These conditions, together with a reduced salivary flow under the denture or poor oral and prosthetic hygiene, favor greater adherence of Candida species to the acrylic substrates of the denture [25]. All denture wearers, and especially diabetic, must maintain the highest level of oral health and remove the denture at night to sleep, to reduce the risk of candidal infection [10].
The oral distribution of the different Candida species between diabetics and non-diabetics was also established, without observing a statistically significant association (p = 0.17). The most prevalent Candida species in both groups was C. albicans, present in 78.1% of diabetics and 71.2% of non-diabetics. On the other hand, when C. albicans were compared with the rest of non-albicans Candida species, diabetics showed less frequent of C. non-albicans species oral detection, with statistically significant differences (p = 0.04). C. tropicalis and C. parapsilosis were the non-albicans species most commonly found in both diabetics and non-diabetics. Most of the studies [17, 21] state the percentage of detection of C. albicans in diabetics around 70%-85%, data that agrees with that indicated in this study. In the oral cavity of diabetics, the increase in sugar concentrations creates an ideal microenvironment for the colonization and proliferation of C. albicans [16].
Considering C. non-albicans species in diabetics, other studies [29], unlike the present one, place C. glabrata as the second Candida species detected in diabetics. There appears to be symbiotic cooperation between C. albicans and C. glabrata. The secretion of proteolytic and lipolytic enzymes by C. glabrata contributes to the invasiveness of the hyphae of C. albicans which, in turn, acts as a promotor for the invasion of C. glabrata, contributing to the increase in the pathogenesis of both species [25]. The pathogenic synergy among different Candida species, generating biofilms of mixed species, allows each other to benefit, resulting in the perpetuation of the infection, with greater difficulty in eradication and higher resistance to antifungal treatment [26].
CONCLUSIONS
R: 2.22, p < 0.001). In contrast, neither gender nor diabetes type conditioned fungal infections (p > 0.05). The most prevalent Candida species in both diabetics and controls were C. albicans and C. tropicalis. Diabetics had significantly fewer Candida non-albicans infections than non-diabetics (p = 0.04).
LIMITATIONS OF THE STUDY
The methods for sample collection (swab, rinse, smear, etc.), which could influence Candida counts, could not be assessed. Different criteria were also found to distinguish between being a Candida carrier without disease and having oral candidiasis.
The results of this meta-analysis should be interpreted with caution due to the high heterogeneity found in some comparisons. The studies differences may be conditioned by the study design type, the methods used to collect information, the type of analysis used or by the characteristics of the populations studied.
New studies are needed to evaluate the factors related to the increased susceptibility of diabetics to oral candidiasis.















