INTRODUCTION
As the traditional drug delivery systems (DDSs) often associated with many side effects, there as an always requirement of approaches to develop new formulations for transporting medicine at required level when it needed. This type of on demand basis drug delivery also called smart drug delivery or intelligent drug delivery. These novel techniques enhance the therapeutic values and also reduce various side effects. In comparison to the conventional DDSs, the smart controlled DDSs can effectively reduce the dosage frequency, while maintaining the drug concentration in targeted organs/tissues for a longer period of time. In this sense, the controlled DDSs provide broad insights and fascinating properties for decreasing drug concentration fluctuation, reducing drug toxicities and improving therapeutic efficacy.1
This new class of intelligent or smart and responsive delivery systems is designed to perform various functions like detection, isolation and/or release of therapeutic agent for the treatment of various diseases. Intelligent drug delivery systems are mostly based on stimuli- responsive polymers which sense a change in a specific variable and activate the delivery; this phenomenon being reversible. A large number of materials such as polymers, lipids and inorganic materials, have been developed to control the drug release in this mannered to create smart drug delivery system. 2)-(7
Benefits of intelligent drug delivery systems
1.Intelligent drug delivery systems are capable to deliver the drug as per the physiological needs.
2.This system help to maintain the plasma drug concentration within therapeutic range.
3.Localized delivery of drug to particular compartment (targeted site) can be achieve.
4.Preserve the drugs that are rapidly destroy, enhance the stability.
5.Improve patient compliance.
TYPE OF INTELLIGENT DRUG DELIVERY SYSTEMS3)-(7
Broadly classify in two categories
Open-loop system
These are also known as externally controlled system or pulsatile systems . These system employed external triggers to deliver the drug such as magnet, temperature, ultrasound, electric effects etc.
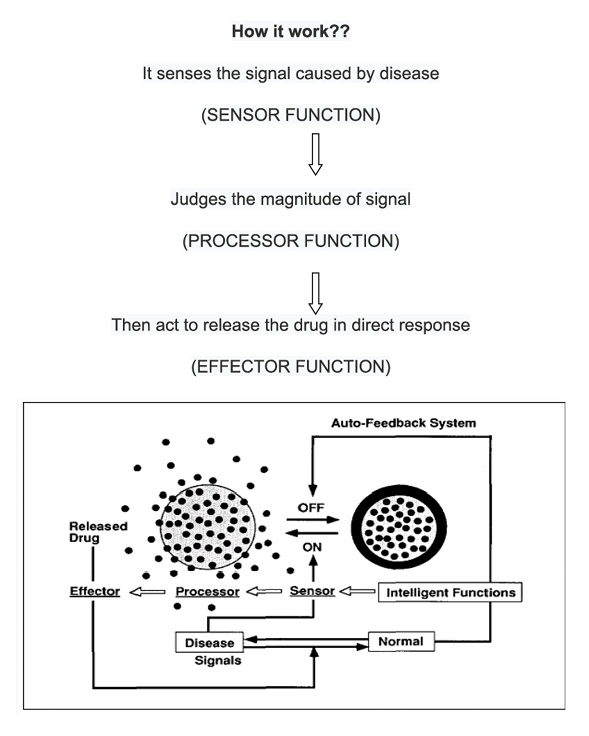
Close-loop system
These are also known as self regulated drug delivery systems or responsive systems. Rate of drug release controlled by feedback information without external intervention. These are uses various approached such as pH-responsive, glucose responsive, thermo-sensitive, urea responsive, inflammation sensitive etc.
Some are discussed below:
a. pH-responsive system
In this type of system pH use as a trigger to release the drug. Alteration in pH of the environment will cause swelling or de-swelling of the polymer. pH-responsive carriers is used to sense the pH at different region of body such as stomach (pH≈ 2) and intestinal tract (pH≈7).8)-(11
b. Redox-responsive system
Oxidation-reduction responsive system have gained huge interest for disease therapy.12),(13 The redox potential in microenvironments is multivariate in different tissues, which can be exploited to design redox-sensitive delivery systems. The design and fabrication of nanoparticles responsive to Glutathione can be a promising approach for targeting drug delivery.14
c. Enzyme-responsive system
Enzymes used as triggers in the design of smart drug delivery systems and gain attentention due to its its unique superiorities, such as substrate specificity and high selectivity15)-(18. Since enzymes (such as glycosidases, lipase, phospoholipases or proteases) are related to almost all the biological and metabolic process, they can be use to get enzyme-mediated drug release17.
d. Thermo responsive system
Temperature is one of the most suitable and efficient factors to control the drug release compared with other stimuli.19)-(21 Poly (acrylamide) gels swell with increasing temperature, while poly(N-alkyl substituted acrylamide) gels deswell. Usually, disease state body temperature raised as compared to normal state some conditions, such as inflammation and tumors, have higher temperatures than normal tissues 22). By use of this temperature difference between cancer tissues and normal tissues, smart drug delivery can be use to enhance their drug release in tumors 23),(24). Another temperature- responsive strategy is that the tumor site could be heated by external triggers (Ultra sound, magnetic field, etc.) to improve the drug release within the tumor vasculature microenvironment.25
e. Light, magnetic, and ultra sound responsive
Photo sensitive carriers are used to design light responsive drug delivery system and the drug release accompanied at desired site by external light illumination. In the magnetic and ultra sound responsive drug delivery system external magnetic field and ultra sound is used respectively to trigger drug molecules at targeted site.26
f. Other responsive systems
Glucose and other Saccharide Sensitive Systems, electro-responsive systems, Inflammation Responsive Systems, Electrically Regulated Systems, Urea Responsive Delivery Systems, Morphine Triggered Naltrexone Delivery Systems, System Utilizing Antibody Interactions, System Utilizing Chelation have also been employed to control the release of drug from smart system. Dual stimuli-responsive DDSs are most common systems and have been investigated, such as thermo- and pH- responsive systems, redox- and pH- -responsive systems thermo- and light- responsive systems, ultrasonic and magnetic responsive systems.27)-(39
Recent advancements
Insulin Pump
The ultimate goal for diabetes mellitus is to control the blood glucose level by insulin delivery and minimized the long-term diabetic complications. Currently main therapy is to administered insulin through subcutaneous injection for diabetes patients. Two or three injections are required a day to maintain the normal blood glucose level. Because this method is burdensome and invasive to living organisms, the patient’s situation would not be good regarding the quality of life. Therefore, an electrical and mechanical controlled insulin pump that injects insulin automatically into the bloodstream has been developed. An insulin pump constructed with polymer materials has been studied. Many researchers working on deliver insulin by smart device.
Gluco Watch
A GlucoWatch™ biographer is non-invasive, watch like device that measures glucose. A plastic part of Gluco watch that snaps into the biographer and sticks to the skin. Automatic reading every 10 min up to 13 hrs is taken by it. Gluco watch presently takes the lead among user-friendly techniques aimed at glucose monitoring. This system is based upon the principle of reverse iontophoresis.
Stimuli-responsive drug-delivery systems in clinical trials:
-a. AuroShell: Thermosensitive gold nanoshell are prepared by Nanospectra Biosciences for Intracranial tumors40.
-b. Opaxio: Enzyme-activated polymeric NP are prepared by Cell Therapeutics, Inc. for treatment of Ovarian cancer. 41
-c. ThermoDox: Thermosensitive liposomal doxorubicin are prepared by Celsion Corporation are useful in Breast cancer and primary liver cancer.42
-d. NanoTherm: Magnetic sensitive iron oxide NPs are prepared by MagForce Nanotechnologies AG for Glioblastoma, prostata cancer, eosphageal cancer and pancreatic cancer.43
RESULTS AND DISCUSSION
Current researches in the field of to intelligent drug delivery summarized here in tabular form (Table 1).
Table 1: current researches in the field of the intelligent drug delivery.
| Researchers / Year | Title of work | Work summary and Findings |
|---|---|---|
| Li et al., (2018) | One-Step Preparation of pH-Responsive Polymeric Nanogels as Intelligent Drug Delivery Systems for Tumor Therapy | In this work, pH-responsive polypeptide-based nanogels are reported as potential drug delivery systems. At pH 7.4, MPEG-b-PNLG nanogels exhibit high stabilities with hydrophobic inner cores, which allow encapsulation of hydrophobic therapeutic agents. Under tumoral acidic environments (pH ∼6.4), the cleavage of benzoic imine bonds induces the destruction of MPEG-b-PNLG nanogels and leads to rapid release of their payloads. Doxorubicin is used as a model drug to evaluate drug encapsulation and release.44 |
| Zhang et al., (2018) | Mesoporous silica nanoparticle-based intelligent drug delivery system for bienzyme-responsive tumour targeting and controlled release | This paper proposes a novel type of multifunctional envelope-type mesoporous silica nanoparticle (MSN) to achieve cancer cell targeting and drug-controlled release. In this system, MSNs were first modified by active targeting moiety hyaluronic acid (HA) for breast cancer cell targeting and hyaluronidases (Hyal)-induced intracellular drug release. Then gelatin, a proteinaceous biopolymer, was grafted onto the MSNs to form a capping layer via glutaraldehyde-mediated cross-linking. This system presents successful bienzyme-responsive targeting drug delivery in an optimal fashion and provides potential applications for targeted cancer therapy.45). |
| Zhao et al., (2018) | An intelligent dual stimuli-responsive photosensitizer delivery system with O2-supplying for efficient photodynamic therapy | In this paper, a new type of biocompatible multifunctional photo sensitizer delivery system was fabricated to relieve tumor hypoxia and improve the efficacy of PDT.46 |
| Li et al., (2018) | Dual-responsive drug delivery systems prepared by blend electrospinning | Temperature and pH dual-responsive drug delivery systems prepared by the thermosensitive polymer poly(N-isopropylacrylamide) (PNIPAAm) by polymerization. It was then co-dissolved with the pH-sensitive polymer Eudragit® L100-55 (EL100-55) and processed into fibers using electrospinning. Ketoprofen, a model drug, was also incorporated into the composite fibers, and fibers based on a single polymer additionally prepared.47 |
| Kumar et al., (2017) | Mesoporous silica nanoparticle based enzyme responsive system for colon specific drug delivery through guar gum capping | Herein, Researchers have developed a mesoporous silica nanoparticle (MSN) based enzyme responsive materials for colon specific drug delivery. They have utilized guar gum, a natural carbohydrate polymer as a capping layer to contain a model drug, such as 5-flurouracil (5FU) within the mesoporous channels of MSN. The drug loaded GG-MSN system also demonstrated near perfect ‘zero release’ property in absence of enzymes in different simulated conditions of the gastrointestinal tract.48 |
| Yang et al., (2016) | Glucose and magnetic-responsive approach toward in situ nitric oxide bubbles controlled generation for hyperglycemia theranostics. | Magnetic microvesicles with glucose oxidase designed as a novel theranostic approach for restoring glucose homeostasis and spatiotemporally control nitric oxide release for maintaining good overall diabetic health.49 |
| Stumpel et al., (2015) | Stimuli-Responsive Materials Based on Interpenetrating Polymer Liquid Crystal Hydrogels | Stimuli-responsive materials based on interpenetrating liquid crystal-hydrogel polymer networks are fabricated. These materials consist of a cholesteric liquid crystalline network that reflects color and an interwoven poly(acrylic acid) network that provides a humidity and pH response. The volume change in the cross-linked hydrogel polymer results in a dimensional alteration in the cholesteric network as well, which, in turn, leads to a color change yielding a dual-responsive photonic material.7) |
| Yu et al., (2015) | Reversal of doxorubicin resistance in breast cancer by mitochondria-targeted pH-responsive micelles. | Researchers reported an effective approach to overcome doxorubicin (DOX) resistance in MCF-7/ADR breast cancer using DOX-loaded pH-responsive micelles. The micelles were prepared from a pH-responsive diblock copolymer, poly(ethylene glycol)-block-poly(2-(diisopropylamino)ethyl methacrylate) (PEG-b-PDPA), and a vitamin E derivate (D--tocopheryl polyethylene glycol 1000 succinate, TPGS) (denoted as PDPA/TPGS micelles).24 |
| Nguyen et al., (2015) | Enzyme-Responsive Nanoparticles for Targeted Accumulation and Prolonged Retention in Heart Tissue after Myocardial Infarction. | Enzyme-responsive peptide-polymer amphiphiles are assembled as spherical micellar nanoparticles, and undergo a morphological transition from spherical-shaped, discrete materials to network-like assemblies when acted upon by matrix metalloproteinases (MMP-2 and MMP-9), which are up-regulated in heart tissue post-myocardial infarction.36 |
| Callmann et al., (2015) | Therapeutic Enzyme-Responsive Nanoparticles for Targeted Delivery and Accumulation in Tumors | An enzyme responsive, paclitaxel loaded nanoparticle is described and assessed in vivo in a human fibrosarcoma murine xenograft. This work represents a proof-of-concept study demonstrating the utility of enzyme-responsive nanoscale drug carriers capable of targeted accumulation and retention in tumor tissue in response to overexpressed endogenous enzymes.16 |
| Paris et al.,(2015) | Polymer-Grafted Mesoporous Silica Nanoparticles as Ultrasound-Responsive Drug Carriers. | A new ultrasound-responsive system based on mesoporous silica nanoparticles was developed for biomedical applications, grafting a copolymer on their surface that acts as gate keeper of the pores. Upon ultrasound irradiation, the sensitive polymer changes its hydrophobicity and, therefore, its conformation toward coil-like opening the gates and releasing the drug.50 |
| Curcio et al., (2015) | On demand delivery of ionic drugs from electro-responsive CNT hybrid films. | Electro-responsive hybrid hydrogel films were prepared by UV-induced polymerization of Acrylamide and N,N′-ethylene bisacrylamide to be applied for the on/off delivery of therapeutics. Swelling behaviour and drug release profiles were evaluated in the presence of an external applied electric voltage and mathematically modelled. The application of an external electric voltage resulted in a faster release of anionic drugs, while a slower release was recorded for cationic drugs.32 |
| Alvarez-Lorenzo et al., (2014) | Smart drug delivery systems: from fundamentals to the clinic | This review offers a critical overview of therapeutically-interesting stimuli to trigger drug release and the evolution of responsive materials suitable as functional excipients, illustrated with recent examples of formulations in clinical trials or already commercially available, which can provide a perspective on the current state of the art on smart drug delivery systems.51 |
| Liu et al., (2014) | pH-Sensitive nano-systems for drug delivery in cancer therapy. | Paper shoed that pH-Sensitive nano-systems have now been developed in which drug release is specifically triggered by the acidic tumor environment. Studies have demonstrated that novel pH-sensitive drug delivery systems are capable of improving the efficiency of cancer treatment. A number of these have been translated from bench to clinical application and have been approved by the food and drug administration for treatment of various cancerous diseases.8 |
| Ganesh et al., (2014) | Smart functional polymers-a new route towards creating a sustainable environment. | This review indicated recent developments in the area of smart functional polymers with a focus on various (physical, chemical and biological) stimuli responsive systems and their applications. Furthermore, article also provides useful insights and in-depth analysis on the feasibility of utilizing stimuli responsive polymeric materials/composites in anti-fouling and water harvesting applications which hold tremendous potential to create a sustainable environment.9 |
| Torchilin et al., (2014) | Multifunctional, stimuli-sensitive nanoparticulate systems for drug delivery | Here, researchers highlighted recent developments with multifunctional and stimuli-sensitive novel drug delivery systems and their therapeutic potential for diseases including cancer, cardiovascular diseases and infectious diseases.25 |
| Chiang et al., (2014) | pH-responsive polymer-liposomes for intracellular drug delivery and tumor extracellular matrix switched-on targeted cancer therapy | In this study, ECM-targeting liposomes were completely investigated their pH-responsibility, drug releasing behaviors, anticancer efficiencies and the time-dependent organ distribution and toxic effects.52 |
| Mura et al., (2013) | Stimuli-responsive nanocarriers for drug delivery | In this Review, researchers have discussed recent advances in the design of nanoscale stimuli-responsive systems that are able to control drug biodistribution in response to specific stimuli, either exogenous (variations in temperature, magnetic field, ultrasound intensity, light or electric pulses) or endogenous (changes in pH, enzyme concentration or redox gradients).12 |
| Kelley et al., (2013) | Stimuli-responsive copolymer solution and surface assemblies for biomedical applications. | In this review, researchers have discuss several methods that have been employed to synthesize self-assembling and stimuli-responsive copolymers for biomedical applications, and we identify common themes in the response mechanisms among the targeted stimuli. Additionally, we highlight parallels between the chemistries used for generating solution assemblies and those employed for creating copolymer surfaces.53 |
| Cheng et al., (2013) | Dual and multi-stimuli responsive polymeric nanoparticles for programmed site-specific drug delivery | Researchers studied novel dual and multi-stimuli responsive polymeric nanoparticles that respond to a combination of two or more signals such as pH/temperature, pH/redox, pH/magnetic field, temperature/reduction, double pH, pH and diols, temperature/magnetic field, temperature/enzyme, temperature/pH/redox, temperature/pH/magnetic, pH/redox/magnetic, temperature/redox/guest molecules, and temperature/pH/guest molecules have recently been developed. These dual and multi-stimuli responsive polymeric nanoparticles have shown unprecedented control over drug delivery and release leading to superior in vitro and/or in vivo anti-cancer efficacy.54 |
| Duanet al., (2013) | Smart pH-sensitive and temporal-controlled polymeric micelles for effective combination therapy of doxorubicin and disulfiram | A smart pH-sensitive polymeric micelles system with high drug loading and precise drug ratio was designed and prepared by conjugating doxorubicin (DOX) to poly(styrene-co-maleic anhydride) (SMA) derivative with adipic dihydrazide (ADH) through a acid-cleavable hydrazone bond, and then encapsulating disulfiram (DSF), a P-gp inhibitor as well as an apoptosis inducer, into the micelles formed by the self-assembly of SMA-ADH-DOX (SAD) conjugate. The pH-sensitive polymeric micelles system enabled a temporal release of two drugs 55 |
| Pan et al, (2012) | Redox/pH dual stimuli-responsive biodegradable nanohydrogels with varying responses to dithiothreitol and glutathione for controlled drug release | A new type of redox/pH dual stimuli-responsive poly(methacrylic acid) (PMAA)-based nanohydrogels was prepared from methacrylic acid and N,N-bis(acryloyl)cystamine crosslinker via distillation-precipitation polymerization. These nanohydrogels possess many favorable traits, such as excellent biocompatibility and biodegradability, minimal drug release under an extracellular condition (non-reductive), adequate drug loading capacity, and rapid drug release.56 |
| Mo et al., (2012) | Multistage pH-Responsive Liposomes for Mitochondrial-Targeted Anticancer Drug Delivery. | Zwitterionic oligopeptide liposomes (HHG2C18-L) containing a smart lipid (1,5-dioctadecyl-L-glutamyl 2-histidyl-hexahydrobenzoic acid, HHG2C18) are developed to overcome the barriers faced by anticancer drugs on the route from the site of injection into the body to the final antitumor target within transport steps with multiple physiological and biological barriers. HHG2C18-L show the multistage pH-responsive to the tumor cell (mitochondria in this case).57 |
| Leung et al., (2012) | Light-activated content release from liposomes | This review focuses on the development of liposome-based functional materials, where payload release is activated by light. Methods of sensitizing liposomes to light have progressed from the use of organic molecular moieties to the use of metallic plasmon resonant structures. This development has facilitated application of near infrared light for activation, which is preferred for its deep penetration and low phototoxicity in biological tissues.58 |
| Yudina et al., (2011) | Ultrasound-mediated intracellular drug delivery using microbubbles and temperature-sensitive liposomes. | In this study, ECM-targeting liposomes were completely investigated their pH-responsibility, drug releasing behaviors, anticancer efficiencies and the time-dependent organ distribution and toxic effects. Experimental results indicate that ECM-targeting liposomes showed rapid drug releasing profiles in acidic conditions.59 |
| Delcea et al., (2011) | Stimuli-responsive LbL capsules and nanoshells for drug delivery | Review of basic principles and recent developments in the area of stimuli responsive polymeric capsules and nanoshells formed via layer-by-layer (LbL) is presented. The stimuli can be logically divided into three categories: physical (light, electric, magnetic, ultrasound, mechanical, and temperature), chemical (pH, ionic strength, solvent, and electrochemical) and biological (enzymes and receptors). Using these stimuli, numerous functionalities of nanoshells have been demonstrated.60 |
| Zhao et al., (2011) | PEGylated thermo-sensitive poly (amidoamine) dendritic drug delivery systems | Researchers engineered PEGylated thermo-sensitive dendritic derivatives and investigated their temperature sensitivity and drug release behaviour therein.24 |
| Wu et al., (2011) | Organization of glucose-responsive systems and their properties | In this review, researchers described various glucose-responsive systems and their properties ant their future prospectus.28 |
| Yun et al., (2011) | Electro-responsive transdermal drug delivery behavior of PVA/PAA/MWCNT nanofibers. | The electro-responsive transdermal drug delivery system was prepared by electrospinning of poly(vinyl alcohol)/poly(acrylic acid)/multi-walled carbon nanotubes (MWCNTs) nanocomposites. The conductivity of nanofibers increased by increasing the content of MWCNTs and performing oxyfluorination with higher oxygen content. (30 |
| Liu et al., (2010) | Magnetically sensitive alginate-templated polyelectrolyte multilayer microcapsules for controlled release of doxorubicin | Magnetically sensitive alginate-templated polyelectrolyte multilayer microcapsules were successfully synthesized by a novel process combining emulsification and layer-by-layer self-assembly techniques. The in vitro release behavior under a high-frequency magnetic field (HFMF) indicated that the applied HFMF accelerated significantly the drug release from the microcapsules, which might be related to the microcapsules’ magnetic properties.61 |
| Gao et al., (2010) | pH-responsive nanoparticles for drug delivery | Nanoparticles whose cores become soluble or change their structures to release the carried drugs at the tumor extracellular pH or lysosomal pH have been developed for fast drug release into the extracellular fluid or cytosol. Such pH-responsive nanoparticles have therapeutic advantages over the conventional pH-insensitive counterpart.10 |
| Patel et al., (2008) | Enzyme-responsive snap-top covered silica nanocontainers | Mesoporous silica nanoparticles, capable of storing a payload of small molecules and releasing it following specific catalytic activation by an esterase, have been designed and fabricated. The storage and release of the payload is controlled by the presence of rotaxanes.62 |
| Zhang et al., (2007) | Thermo and pH Dual-Responsive Nanoparticles for Anti-Cancer Drug Delivery | Thermo and pH dual-responsive nanoparticles for an anti-cancer drug were prepared. These nanoparticles aggregated at body temperature under a slightly acidic pH of 6.9 (see figure), and a faster drug release was found to be associated with higher temperature and lower pH, both of which are advantageous for tumor-targeted anti-cancer drug delivery.33 |
| Murdan et al., (2003) | Electro-responsive drug delivery from hydrogels. | Electrically-responsive drug release is influenced by a number of factors such as the nature of the drug and of the gel, the experimental set-up, magnitude of the electric field etc. In this paper, electrically-responsive hydrogels, response of gels to an electric field and electrically-stimulated drug release are discussed. 63 |
CONCLUSION
Intelligent drug deliver means deliver of drug to selectively to targeted cells and tissues while leaving other healthy parts of the body untouched. Although research working on many areas to improve drug delivery to targeted site and therefore drug delivery systems gained wide attention in biomedical research due to their potential to significantly reduce the side effects of drugs and control the concentration and location of active drugs released in the body over long periods of time. Clearly new strategies for preparing drug delivery systems are capable of selectively releasing drug at targeted site will help improve many of the current problems in drug therapy.