INTRODUCTION
Peripartum cardiomyopathy (PPCM) is a rare, idiopathic, and often dilated cardiomyopathy (DCM) with a conspicuous systolic dysfunction that affects childbearing women during pregnancy or in the early postpartum period. Sometimes, there is a delay in diagnosis that may occur because the symptoms and signs of PPCM can mimic the normal findings of late pregnancy and the peripartum period [1]. Albeit a few patients harbor mild disease and have complete recuperation, others experience chronic cardiomyopathy with paramount morbidity and mortality [1, 2]. Physicians are frequently presented with diagnostic and therapeutic dilemmas in the face of limited evidence-based therapy alternatives [1]. For PPCM, this review provides the most up-to-date information on risk factors, etiology, pathophysiology, and prognostic variables; it also highlights management of PPCM as well as the future research directions.
OVERVIEW
PPCM is defined as an idiopathic cardiomyopathy that presents with HF secondary to left ventricular (LV) systolic dysfunction toward the end of pregnancy or in the months after delivery, in the absence of any other cause of HF [1, 2]. PPCM is an exclusionary diagnosis, with the majority of patients being diagnosed postpartum. However, other potential causes of HF (e.g., pregnancy-associated myocardial infarction or pulmonary embolism) and pre-existing heart diseases (e.g., congenital heart disease or chemotherapy induced toxic cardiomyopathy) should be ruled out [3]. Although it resembles the features of DCM, it is considered as an independent entity [4, 5]. Although the LV may not be dilated, the ejection fraction is nearly always reduced below 45% [1, 2].
PPCM commonly observed in multiparous black older (more than 30 years) women, women with pregnancy associated hypertensive disorders such as gestational hypertension, pre-eclampsia, or HELLP syndrome (hemolysis, elevated liver enzymes and low platelets) [6]. Smoking and malnutrition are additional risk factors. The overlap between PPCM and pre-eclampsia is clinically paramount, as patients with pre-eclampsia can present with noncardiogenic pulmonary edema and the coexistence of these two conditions highlights potential shared pathogenic mechanisms [6].
PATHOPHYSIOLOGY
The etiology of PPCM is largely unclear and is likely to be multifactorial [2]. Potential mechanisms include nutritional deficiencies [7, 8], viral myocarditis [9, 10], and autoimmune processes [11, 12]. Hemodynamic stress of pregnancy has been argued as a potential etiology. However, the maximal cardiovascular changes take place in the second trimester [13] when most women with pre-existing cardiac diseases develop symptomatic HF [14]. In contrast, the majority of women with PPCM become symptomatic during late pregnancy or postpartum [15].
Recent research has pitched into “two hit” model of PPCM pathogenesis, whereby a vascular insult caused by antivascular or hormonal effects leads to cardiomyopathy in women with an underlying predisposition. The first model is a STAT3 knockout mouse in which oxidative stress generates the cleavage of the nursing hormone, prolactin. The 16-kDa prolactin fragment has vasculotoxic and pro-apoptotic properties that cause vascular and myocardial dysfunction [16]. This ultimately leads to systolic HF, which is potentially reversible. The diseased myocardium is salvaged by specific treatment with bromocriptine that blocks the prolactin release from the pituitary gland and, therefore, prevents the cleavage of the full-length prolactin into the toxic 16-kDa fragment [16].
The soluble VEGF receptor sFlt-1 (soluble fms-like tyrosine kinase-1) is produced in the placenta and has been linked to a systemic angiogenic imbalance, among other things, in late pregnancy [17, 18]. In a mouse model lacking cardiac peroxisome proliferator-activated receptor gamma coactivator 1-alpha, vascular dysfunction mediated by elevated sFlt-1 negatively impacts heart function (PGC-1) [19]. Patients with pre-eclampsia or twin pregnancies are more likely to develop PPCM because sFlt-1 has a role in all conditions. Together with VEGF, bromocriptine and VEGF both partly alleviate pregnancy-related cardiomyopathy, but only when administered separately [17, 20].
GENETICS
More than 15% of women with PPCM had a family history of heart disease, according to the German registry. Cardiac myosin heavy chain (MYH) is one of the most often impacted genes, together with titin (TTN) and SCN5 [4, 21]. Most of the mutation carriers are asymptomatic prior to the pregnancy. When women are exposed to high levels of hemodynamic stress during late pregnancy, delivery and the early postpartum period, genetic cardiomyopathies can be discovered [21].
CLINICAL PRESENTATION AND DIAGNOSIS
Women with PPCM often have congestion symptoms including dyspnea on exertion, orthopnea, paroxysmal nocturnal dyspnea, fatigue, chest tightness and edema of the lower extremities [15]. Physical examinations often reveal tachypnea, tachycardia, elevated jugular venous pressure, pulmonary rales, and oedema.. Cardiogenic shock, unstable arrhythmias [22, 23] and arterial thromboembolism are less prevalent [24, 25]. Most women present postpartum, mainly during the first month after delivery; a considerable number present in the late antepartum period; and rare cases present as early as the second trimester [15, 26].
In PPCM, echocardiography continues to be the most effective imaging technique. It is diagnosed by echocardiographic evidence of left ventricular dysfunction with left ventricular ejection fraction (LVEF) <45% and often (but not always) left ventricular dilatation; presentation in peripartum or in the early postpartum period; and absence of an alternative explanation. Aside from right ventricular dilatation and dysfunction, an echocardiogram may also detect pulmonary hypertension, left atrial or biatrial dilatation, functional regurgitation of the mitral and tricuspid valves. In addition, it could be also observed in rare, an intracardiac thrombus [27]. Normal pregnancies would not have much of an effect on BNP and N-terminal BNP levels, whereas PPCM generally has a considerable increase in BNP and N-terminal BNP levels [28, 29, 30, 31]. Even if the ECG is normal, it does not rule out the possibility of PPCM [32]. Pulmonary venous congestion may be seen on a chest x-ray. Cardiac magnetic resonance imaging provides accurate ejection fraction and chamber measurements when the echocardiogram is inadequate, but gadolinium is avoided during pregnancy as it enters the placenta and may be teratogenic. An endomyocardial biopsy is not often required, and there are no diagnostic histological results. When another cause of HF is suspected it may merit biopsy when an alternative diagnosis, such as heart block and ventricular tachyarrhythmias suggesting giant cell myocarditis or cardiac sarcoidosis, would change the management [26].
DIFFERENTIAL DIAGNOSIS
PPCM is an exclusionary diagnosis. The step to preventing overdiagnosis is to pay close attention to suspected pre-existing cardiac conditions, such as cardiomyopathies and valvular disease. Diastolic dysfunction and HF may be caused by severe pre-eclampsia. However, systolic dysfunction is required for PPCM to be diagnosed. A number of different conditions, as shown in Table 1 can cause HF during pregnancy.
Table 1. Differential diagnosis for heart failure during pregnancy
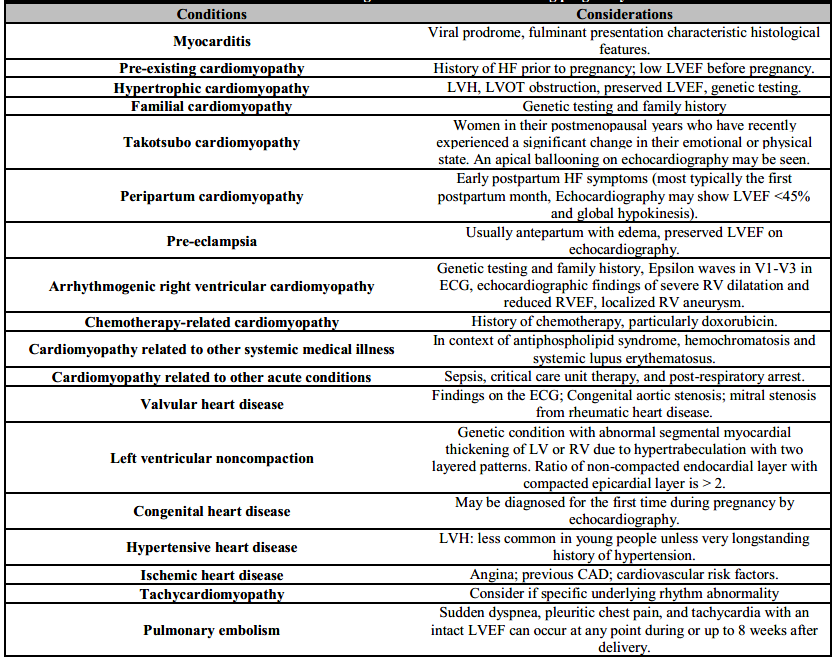
CAD: Coronary artery disease; HF: Heart failure; LV: Left ventricle; LVEF: Left ventricular ejection fraction; LVOT: Left ventricular outflow tract; LVH: Left ventricular hypertrophy; RV: Right ventricle; RVEF: Right ventricular ejection fraction.
COMPLICATIONS AND PROGNOSTIC FACTORS
The most frequent severe complication of PPCM is thromboembolism, which affects 6.6% to 6.8% of women. Cardiovascular thrombosis may affect the left or right side of the heart. Cardiac dilatation and hypocontractility, leading to blood stasis as well as endothelial damage, may contribute to the formation of intracardiac thrombosis [33]. Arrhythmias play a major role in PPCM morbidity and mortality in PPCM and a sudden cardiac death from ventricular tachyarrhythmia is believed to be the most prevalent cause of mortality in this group [34]. As a result, there are many adverse outcomes associated with PPCM, including brain damage, cardiac arrest, pulmonary edema, mechanical circulatory support and death.
Of the various prognostic factors, LVEF <30% upon diagnosis is the most accurate indicator of adverse events or long-term good recovery. The presence of LV dilatation, right ventricular systolic dysfunction, obesity, African American ethnicity, simultaneous pre-eclampsia, and raised troponin, NT-proBNP, and sFlt1 levels are all associated with adverse prognosis. In comparison to other types of HF with reduced LVEF, PPCM has been shown to have a better recovery rate [35, 36], and recovery often occurs within first 3 to 6 months of treatment [37]. In certain cases, recovery might be delayed for as long as two years after the diagnosis [38, 39, 40, 41].
MANAGEMENT
PPCM therapy is typically extrapolated from other types of heart failure with a reduced LVEF or expert opinion (or a combination of both), with special attention to avoiding adverse fetal effects in women who are still pregnant (Table 2).
Table 2. Safety of medications for peripartum cardiomiopathy during pregnancy and lactation
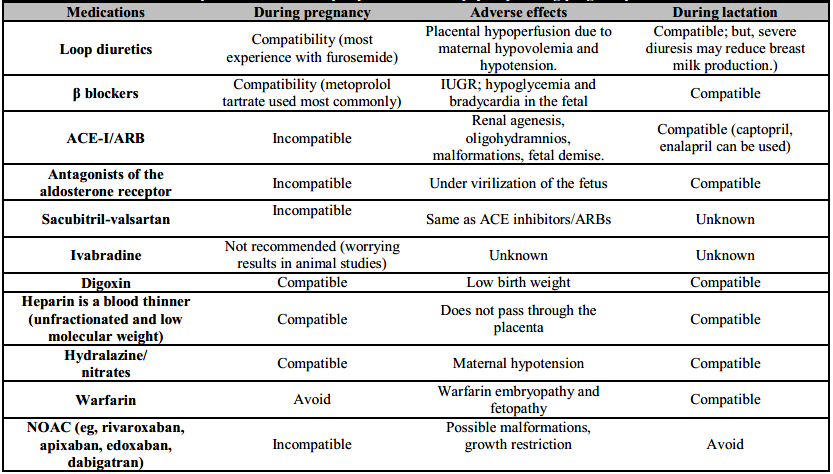
IUGR: Intra-uterine growth retardation; ACE-I: Angiotensin converting enzyme inhibitor; ARB: Angiotensin receptor blocker; NOAC: Novel oral anticoagulants.
The basis of volume management is sodium restriction. Symptomatic pulmonary or peripheral edema may benefit from the use of a loop diuretic [42]. Over-diuresis during pregnancy should be avoided at all costs to avoid the dangers of maternal hypotension and uterine hypoperfusion. If hemodynamics allow, β-blockers are administered, with a preference for β1 selective ones to avoid stimulating uterine contraction via β2 innervation [15]. All angiotensin converting enzyme inhibitors, angiotensin receptor blockers, including sacubitril-valsartan are contraindicated in pregnant women [42]. Mineralocorticoid receptor antagonists should be avoided during pregnancy in view of their anti-androgenic effects, although they may be taken during breast feeding [43]. Digoxin may be safely used in pregnancy [44].
Mechanical circulatory support with intra-aortic balloon pump, percutaneous ventricular assist device therapy, and extracorporeal membrane oxygenation have been used successfully in PPCM and should be considered early in patients with hemodynamic instability despite inotropic support [35]. Additionally, termination of pregnancy by urgent delivery via caesarean section is mandatory in these cases. To aid in fetal lung development, women should be administered corticosteroids until they reach the 34th week of gestation. Levosimendan is a calcium sensitizing agent which leads to improvement in systolic function and hemodynamics in the PPCM subset [45].
In view of the increased incidence of LV thrombi and systemic thromboembolism, anticoagulation is recommended in the setting of severely decreased LVEF during late pregnancy and 6 to 8 weeks postpartum [46].
The European Society of Cardiology (ESC) recommends anticoagulation if the LVEF is ≤ 35% [47], whereas the American Heart Association (AHA) recommends using LVEF is ≤ 30% as theshhold [48]. Warfarin crosses the placenta and is teratogenic (Table 2). Due to a paucity of human evidence, the newest direct-acting oral anticoagulants should be avoided during pregnancy and breastfeeding. The anticoagulants of choice for pregnant women with PPCM include low molecular weight heparin and unfractionated heparin since they do not cross the placenta. It is safe to use warfarin and low molecular weight heparin during lactation [48].
Women with PPCM complicated by arrhythmias may require acute or chronic administration (or both) of antiarrhythmic drugs. Cardioversion and defibrillation are considered safe throughout the pregnancy and should be performed without delay in emergencies [49]. The ESC and AHA recommend consideration of these devices in women with PPCM and LVEF ≤35% as a bridge to LV recovery or to implantable cardioverter-defibrillator (ICD) implantation after 3 to 6 months [2, 42, 47]. Early implantation an ICD is generally discouraged as most women recover [2].
BROMOCRIPTINE
Bromocriptine provides PPCM patients with three advantages. Firstly, ablactation is required to avoid the high metabolic demands of lactation and breastfeeding. Secondly, ablactation is advised to ensure that the usual oral HF medicine is properly and safely administered. Thirdly, bromocriptine is used to suppress the secretion of prolactin as a therapy for a particular condition [19].
In an observational German registry, 72% of women who “improved” after PPCM had received bromocriptine, compared with 35% of women who did not improve [19]. A recent randomized controlled trial (RCT) of 63 German women with PPCM that compared one week and eight week regimens of bromocriptine found similar improvements in LVEF [50]. No women died or required left ventricular assist device or transplantation. However, it is not clear if bromocriptine's prolactin-inhibiting effects extend to all women with PPCM and should thus become standard therapy. The 2018 ESC guidelines include a Class IIb, Level of Evidence: B recommendation for the use of bromocriptine [2]. Due to the association with thrombotic complications, therapeutic anticoagulation is recommended in conjunction with bromocriptine. This therapy may be best justified in women with PPCM who have severe cardiomyopathy (LVEF <25%) or cardiogenic shock (or both) [2].
LABOR AND DELIVERY
It is reasonable to make an effort to stabilize the mother in order to minimize prematurity-related issues for the fetus. Hemodynamic instability despite medical therapy should prompt early delivery (or termination if prior to fetal viability). The possibility of vaginal delivery is considered for stable patients unless obstetric reasons for cesarean section exist. An increased risk of bleeding, infection, and thromboembolic consequences is related with caesarean delivery. When making pre-delivery arrangements, it is important to consult the patient and an experienced multidisciplinary team. Unstable patients may benefit from invasive hemodynamic optimization prior to delivery and monitoring during delivery and the early postpartum period. [35].
Venous return increases following delivery, removal of caval compression by the fetus, autotransfusion due to uterine contractions, and fluid mobilization and resorption. The post-partum risk of fluid overload and pulmonary edema must be anticipated [35].
PRE-DISCHARGE ADVICE
BREASTFEEDING
Breastfeeding is safe in PPCM, suggesting that increasing prolactin production continually is also not necessarily harmful. Most HF medications are safe to use during breastfeeding (Table 2). When left ventricular failure (e.g., an LVEF of less than 25 percent) or cardiogenic shock is present, it is reasonable to consider the use of bromocriptine unless further data available [48].
SUDDEN CARDIAC DEATH PREVENTION
There is paucity of published data guiding the timing of ICD implantation. Within the first six months after postpartum, most patients recover to LVEF >35 percent and do not meet the criteria for ICD implant. There should be no rush to make an order [51]. Women with PPCM confront an increased risk of cardiac arrest in the months following their diagnosis, so extra precautions should be taken to keep them safe. In a small prospective study of women with newly diagnosed PPCM, 3 of the 7 patients with severely reduce LV function were compliant with a wearable cardioverter/defibrillator were found to have ventricular fibrillation that was appropriately shocked [52]. Wearable cardioverter/defibrillators, on the other hand, did not deliver a single shock throughout the four months of follow-up in patients with PPCM [53]. Despite conflicting data, women with new onset PPCM and severe LV failure may benefit from wearable cardioverter/defibrillators as a bridge to recovery or until an implantable ICD is required. Cardiac resynchronization treatment (CRT) may be beneficial for certain patients [54].
COUNSELING AND MANAGEMENT OF SUBSEQUENT PREGNANCY
The risks associated with a subsequent pregnancy depend primarily upon whether the myocardial function has fully recovered and the pre-pregnancy LVEF is the strongest predictor of outcomes (Table 3). The patients should be advised on the reported high risk of recurrent HF, durable deterioration of cardiac function, and mortality in case of persistent myocardial dysfunction (LVEF <50%). There is an increased risk of stillbirth, abortion, and pre-term delivery among women with persistent LV dysfunction. If the LVEF is not more than or equal to 50% to 55%, the ESC advises against further pregnancies [2]. Pregnant women who recover LVEF of at least >50% have a decreased risk of problems, but there is still an increased chance of recurrence of HF in the future pregnancy [55]. Pre-conception counseling should include discussion of the potential risk of recurrent mocardial dysfunction, which may persist after the pregnancy [55].
Table 3. Subsequent pregnancy management
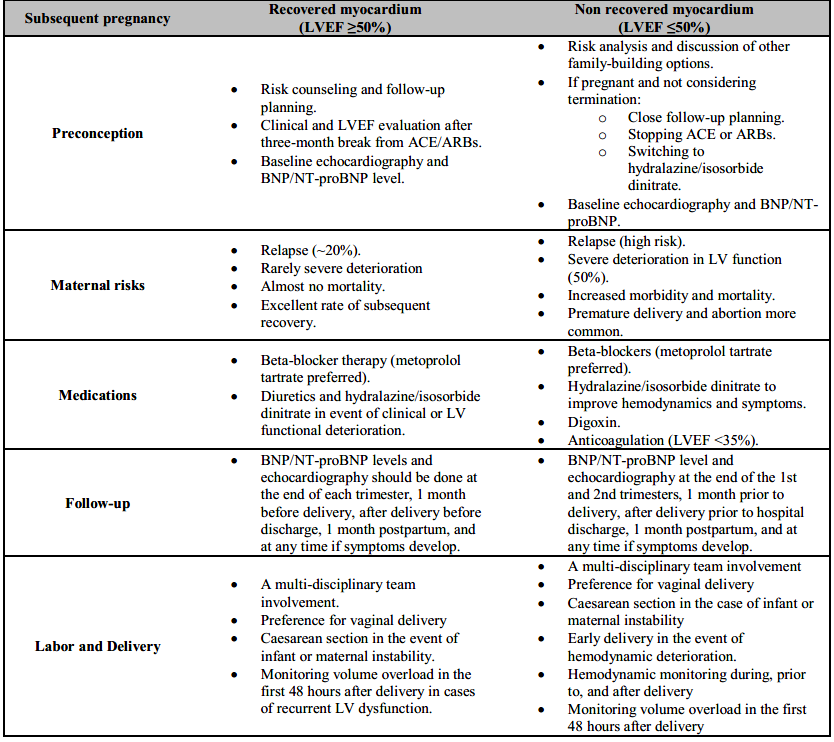
ACE: Angiotensin-converting enzyme inhibitor; ARB: Angiotensin receptor blocker; LV: Left ventricle; LVEF: Left ventricular ejection fraction; BNP: brain natriuretic peptide.
In women with recovered LV function who are on medications for HF, ACE inhibitors/angiotensin receptor blockers, and aldosterone receptor antagonists should be discontinued prior to conception (Table 3).
The stability of LV function must be ensured after at least 3 months off these medications prior to considering the LV recovered. There is paucity of data to support prophylactic use of beta-blockers during subsequent pregnancies.
CONCLUSIONS
PPCM is an uncommon yet potentially life-threatening condition with a significant maternal morbidity and mortality. Transthoracic echocardiography and natriuretic peptide levels may provide a quick and accurate diagnosis. Vasculo-hormonal impacts and genetic predisposition are likely to have some role in the disease's pathogenesis, but the key steps involved need to be clarified. The role of bromocriptine remains unclear, and further RCTs are warranted for determining the potential benefits. Important gaps in knowledge need to be answered, such as the optimal anticoagulation strategy, timing of ICD implantation, risk prediction and management during a subsequent pregnancy and the long-term duration of medications after myocardial recovery. Ongoing efforts at the bench, bedside, and population level that leverage international collaborations are critical to enabling the identification of novel treatments and improving patient outcomes.














