INTRODUCTION
Polypharmacy, the concurrent use of multiple medications in elderly patients is common worldwide.1 Polypharmacy increases the risk of negative health outcomes, including adverse drug events (ADEs), drug-disease or drug-syndrome interactions, and potentially inappropriate medications (PIMs).2,3 PIMs are medications that can potentially harm patients and are associated with a 44% increase in mortality compared to no PIM usage.4 5-6
A systematic review reported there were up to 46 assessment tools for identifying PIMs: 28 criteria-based or explicit tools, eight judgment-based or implicit tools, and 10 assessment tools using both approaches.7 Of those reported in the review, only 19 tools applied consensus methods. Two explicit tools with the most citations are the Beers criteria, developed in America, and the Screening Tool of Older People’s Prescriptions (STOPP) criteria, developed in Europe. The latest versions of the Beers criteria and the STOPP were published in 2019 and 2015, respectively.5,8
High proportions of PIMs are reported in various settings, such as 30-50% in primary care and 43% in nursing homes.9 10-11 Differences in the prevalence rates of PIMs in different settings and countries may be a result of different assessment tools used to identify PIMs. Many studies have found that the prevalence rates of PIMs identified by the Beers criteria were higher than those identified by the STOPP criteria. Using the STOPP version 1 in primary care, the prevalence of PIMs was 37.3% and 33.8% in Ireland and Brazil, respectively.9,12 However, the prevalence of PIMs in Brazil was higher using the Beers criteria than using the STOPP criteria: 51.8% vs. 33.8%, respectively.9 Another study found that 50% of community-dwelling older adults reported the use of at least one PIM based on the Beers criteria compared to 46% based on the STOPP criteria.10
In Thailand, an assessment tool for PIMs was published in 2008 based on a literature review of high-risk medications and the Delphi method with 17 geriatricians.13 This assessment tool, though based on theoretically sound methods, is not widely used. PIMs in the Thai elderly were reported in secondary and tertiary hospitals with different prevalence rates, partly because of different criteria used in each study. Of 430 outpatients in a district tertiary hospital in southern Thailand, 28.1% received at least one PIM, as identified by the 2012 Beers criteria.14 In contrast, based on lists of risk drugs for Thai elderly adults, 79% of Thai elderly adults in a community secondary hospital received at least one PIM.15
There are no data in Thailand regarding PIMs in primary care. Therefore, we conducted the study reported herein to close this knowledge gap. The primary objective of the study was to examine and compare the prevalence of PIMs in primary care using three different criteria for PIMs. The secondary objective was to examine associations between PIM and other independent factors, such as age and gender.
METHODS
This study was approved by the Human Research Ethics Committee of Thammasat University No.1, Faculty of Medicine (MTU-EC-CF-1-230/59). The permission to review and collect data from medical electronic records was approved by the director of the primary care unit (PCU). To protect patients’ confidentiality, PCU staff removed all personal identifications, including national identification numbers, names, and addresses, before sending the data to the authors.
Study design and setting
This was a retrospective cross-sectional study exploring PIMs among elderly patients in a semiurban PCU in Pathum Thani, Thailand, which is overseen by the Faculty of Medicine of Thammasat University. Doctors in the PCU were family physicians, general practitioners (doctors without specialty training), residents in family medicine, and internists.
Participants
Patients aged >65 years were eligible for this study. Those without a prescription in the past year were excluded. The sample size was calculated to be 369 patients, with an estimated PIM prevalence of 40%, a 95% confidence interval, and a 5% margin of error.11 We rounded this number up to recruit 400 patients to increase estimation accuracy.
Eligible patients were selected using systematic random sampling. The patient identification numbers of all patients were listed in order and the first patient was selected randomly. Every sixth patient was then selected until the sample size reached 400 patients.
Data collection
The electronic medical records of the patients were reviewed. The following data from the previous year (2016-2017) were collected: demographic data, including age and gender; diagnoses according to the 10th Revision of International Classification of Diseases and Health-Related Problems (ICD-10), medical prescriptions, and creatinine levels. Other laboratory findings were not collected because ICD-10 codes for abnormal findings, such as hypokalemia, were used to identify conditions that meet PIM criteria. ICD-10 codes starting with V to Z (V to Y for external causes of morbidity and mortality and Z for factors influencing health status and contact with health services) were not collected for data analysis. Quality control for data entry was performed. If input errors were found, the first author rechecked the errors with a data source and correct every mistake.
PIMs
Three sets of criteria were used to identify PIMs: the 2015 Beers, the STOPP version 2 (v2), and the Winit-Watjana.4,5,13 The 2015 Beers criteria comprise of 5 groups of PIMs, including 1) medications for many or most older adults to avoid, 2) medications for older adults with specific diseases or syndromes to avoid, 3) medications to be used with caution, 4) potentially clinically important non-anti-infective drug-drug interactions, and 5) non-anti-infective medications to avoid or the dosage of which should be adjusted based on the individual’s kidney function.
In contrast to the Beers criteria, the STOPP v2 does not divide PIMs into five groups. All criteria are divided according to organ systems or medication classes. For the Winit-Watjana criteria, PIMs were divided into two groups: high-risk medications with potential adverse reactions and high-risk medications with drug interactions (drug-drug and drug-disease). Each Winit-Watjana criterion comprises one of four recommendations: 1) should be avoided, 2) rarely appropriate, 3) with some indications for older patients, and 4) unclassified. Inhaled and topical medications were not collected for data analysis because they were not in the criteria (except estrogen patch and vaginal cream) and may lead to misinterpretation of results.
All authors manually identified PIMs in each prescription using both clinical (ICD-10 codes and laboratory findings) and prescription (dosages and numbers of medications) data. The first author (VP) verified all prescriptions to detect discrepancies in PIM identification. The authors discussed resolving all discrepancies and, if needed, verified PIM identification with prescriptions and patients’ medical records.
Analysis
Both descriptive and analytical analyses were conducted. For descriptive analyses, medians and interquartile ranges (IQRs) were used to describe the following variables: age, number of diseases, medications, and prescriptions. Proportions were used for gender and PIMs. Persons and prescriptions were used as units of analysis for the prevalence of PIMs. The prevalence of prescriptions with PIM was calculated to answer how often doctors prescribed PIMs. A person was coded as 1 if the person received at least one PIM according to at least one of the three criteria in the past year, and a prescription was coded as 1 if the prescription contained at least one PIM according to at least one of the three tools. The use of ≥2 medications that were identified as PIMs according to these three criteria were subsequently analyzed. Comparisons for PIMs prevalence from each criterion were done using Chi-square tests.
For bivariate analyses, Student’s t-test and chi-square or Fisher’s exact test were used to compare the means and proportions of independent variables between individuals with PIMs and those without PIMs. A p-value of 0.05 was used to indicate statistical significance in this study. Variables with p<0.05 were included in multiple logistic regression analysis to explore associations between independent variables and PIMs. Adjusted odds ratio with 95% confidence interval was calculated. All analyses were conducted using STATA software version 13 (Stata, College Station, TX, USA).
RESULTS
The records of 400 elderly patients were retrieved with the median age of 70.5 years (Table 1). A total of 1,640 prescriptions, comprising 6,852 medications, were reviewed. Of all the prescriptions, almost 40% had polypharmacy or a concurrent use of ≥5 medications.
Table 1. Characteristics of 400 elderly patients
| Variables | Median | (P25, P75, IQR) |
|---|---|---|
| Age | 70.5 | (67, 75, 8) |
| Female – n (%) | 264 | (66) |
| Number of diseases | 3 | (2, 4, 2) |
| Number of medications | 11 | (5, 25, 20) |
| Number of prescriptions | 3 | (2, 6, 4) |
IQR: interquartile range; P25: percentile 25%; P75: percentile 75%
Of 400 patients, 301 (75.3%) received PIMs in either criterion at least once in the past year (Figure 1). The Winit-Watjana criteria identified the highest proportion of patients with PIMs in the samples (66.8%), which was 7.8% and 25% higher than those identified by the 2015 Beers criteria and the STOPP v2, respectively (p <0.001).
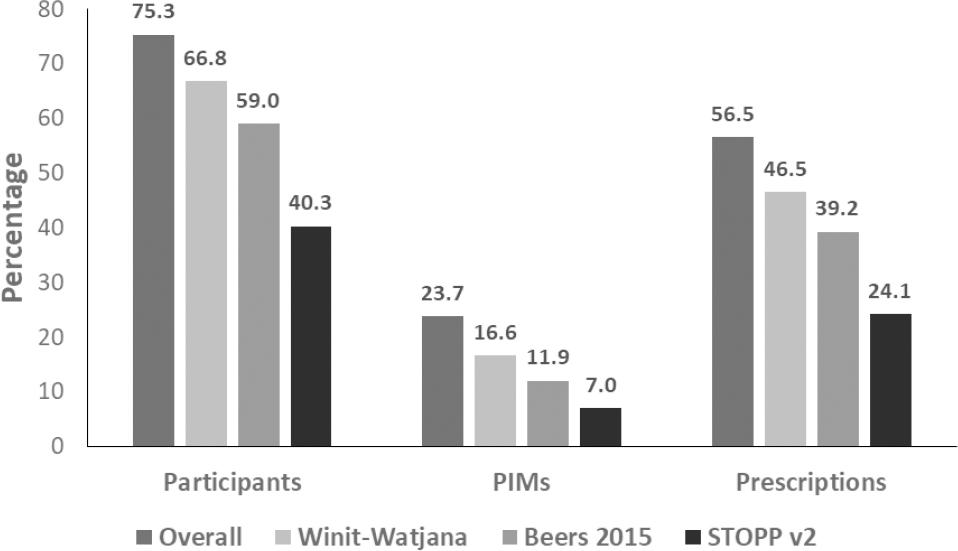
Figure 1. Proportions of PIMs by criteria.Chi-square tests shows statistically significant differences in the PIMs proportion from each criterion with p<0.001.
When considering the proportions of PIMs per prescription, 56.5% of the 1,640 prescriptions included at least one PIM based on any of the three criteria (Figure 1). Of these criteria, Winit-Watjana identified the highest proportion of PIMs (46.5%), followed by the 2015 Beers and the STOPP v2.
The most common PIMs identified by any criteria were orphenadrine, nonsteroidal anti-inflammatory drugs (NSAIDs), and angiotensin-converting enzyme inhibitors (ACEIs). Table 2 shows the top five PIMs in each criterion. Overall, orphenadrine, a first-generation antihistamine, was the most common PIM identified by all three criteria. ACEIs and flunarizine were identified only by the Winit-Watjana criteria, while dimenhydrinate and tramadol were identified only by the 2015 Beers criteria and the STOPP v2, respectively.
Table 2. Top five PIMs by criteria (2016-2017)
| Rank | Overall | Rank | Winit-Watjana | ||||
|---|---|---|---|---|---|---|---|
| PIMs | N | (%) | PIMs | n | (%) | ||
| 1 | Orphenadrine | 255 | (15.9) | 1 | Orphenadrine | 255 | (22.7) |
| 2 | NSAIDs a | 231 | (14.4) | 2 | NSAIDs a, e | 231 | (20.5) |
| 3 | ACEI b | 208 | (13.0) | 3 | ACEI b | 208 | (18.5) |
| 4 | Dimenhydrinate | 155 | (9.7) | 4 | Benzodiazepine c | 112 | (10.0) |
| 5 | Benzodiazepine | 112 | (7.0) | 5 | Flunarizine | 101 | (9.0) |
| 6 | Others | 642 | (40.0) | 6 | Others | 218 | (19.4) |
| Total | 1603 | (100.0) | Total | 1125 | (100.0) | ||
| Rank | 2015 Beers | Rank | STOPP version 2 | ||||
| PIMs | n | (%) | PIMs | n | (%) | ||
| 1 | Orphenadrine | 255 | (32.7) | 1 | Benzodiazepine | 112 | (22.2) |
| 2 | Dimenhydrinate | 155 | (19.8) | 2 | 1st-generation antihistamine | 108 | (21.4) |
| 3 | Benzodiazepine | 112 | (14.3) | 3 | Opioid | 96 | (19.0) |
| 4 | 1st-generation antihistamine d | 108 | (13.8) | 4 | NSAIDs e | 79 | (15.6) |
| 5 | Omeprazole | 78 | (10.0) | 5 | Omeprazole | 78 | (15.4) |
| 6 | Others | 73 | (9.3) | 6 | Others | 32 | (6.3) |
| Total | 781 | (100.0) | Total | 496 | (100.0) | ||
anonsteroidal anti-inflammatory drugs
bangiotensin-converting enzyme inhibitors
cexamples of benzodiazepine: lorazepam and alprazolam
dexamples of 1st-generation antihistamine: brompheniramine, chlorpheniramine and hydroxyzine
eNumbers of NSAIDS differed depending on criteria
The use of ≥2 medications that were identified as PIMs were identified against each criterion. Of all the patients, 15.5% received these PIMs at least once in the past year (Figure 2). The STOPP v2 identified the highest proportion of the use of ≥2 medications in the samples (14%), which were 7.2% and 11.2% higher than those identified by the 2015 Beers and Winit-Watjana criteria, respectively.
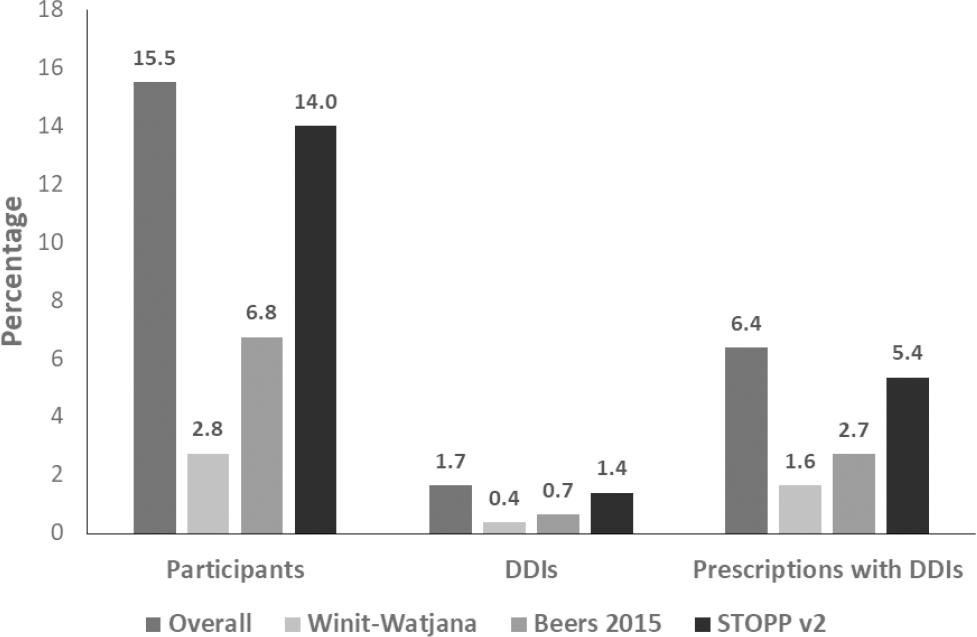
Figure 2. Proportions of the use of ≥2 medications that were identified as PIMs according to these three criteria were subsequently analyzed by criteria.Chi-square tests shows statistically significant differences in the proportions from each criterion with p<0.001.
Of the 1,640 prescriptions, 6.4% had the use of ≥2 medications that were identified as PIMs. The STOPP v2 identified 88 prescriptions with these PIMs (5.4%), which were 43 and 61 more prescriptions with these PIMs than those identified by the 2015 Beers and Winit-Watjana criteria, respectively (Figure 2). The most common uses of ≥2 medications that were identified as PIMs by any of the three criteria were duplicate drug classes (41.3%), two medications with anticholinergic activity (33.9%), aspirin and NSAIDs (12.8%), warfarin and NSAIDs (8.3%), and warfarin and aspirin (3.7%). An example of duplicate drug classes was chlorpheniramine and hydroxyzine. An example of two medications with anticholinergic activity was dimenhydrinate and amitriptyline.
Bivariate analysis showed that all variables, except gender and age, were associated with PIM prescriptions for any criteria. The associations were similar in each criterion. However, age was associated with PIM prescriptions using the STOPP v2, and gender was associated with PIM prescriptions using the 2015 Beers and Winit-Watjana criteria.
With adjusted analyses, ORs were 3.93 (95%CI 2.17-7.12), 2.78 (1.39-5.56), and 0.97 (0.50-1.87) for polypharmacy, four or more diagnoses, and four or more prescriptions in the past year, respectively. In a multivariate analysis, polypharmacy and ≥ 4 diagnoses were associated with PIM prescriptions in any criteria (Table 3). The findings were similar for PIMs that were identified by each category. Female gender was associated with PIM prescriptions when PIMs were identified by the 2015 Beers [adjusted OR = 1.72 (1.10-2.70)] and Winit-Watjana [adjusted OR = 1.78 (1.12-2.84)] criteria.
Table 3. Multiple regression analysis by criteria (2016-2017)
| Variables | Adjusted OR | 95%CI | p-values |
|---|---|---|---|
| Age | |||
| Overall | 1.01 | 0.96 - 1.05 | 0.708 |
| Winit-Watjana | 1.01 | 0.97 - 1.05 | 0.622 |
| 2015 Beers | 1.00 | 0.96 - 1.04 | 0.957 |
| STOPP version 2 | 1.03 | 0.99 - 1.06 | 0.165 |
| Female | |||
| Overall | 1.40 | 0.84 - 2.32 | 0.197 |
| Winit-Watjana | 1.01 | 0.97 - 1.05 | 0.622 |
| 2015 Beers | 1.00 | 0.96 - 1.04 | 0.957 |
| STOPP version 2 | 1.03 | 0.99 - 1.06 | 0.165 |
| Polypharmacy | |||
| Overall | 3.93 | 2.17 - 7.12 | <0.001 |
| Winit-Watjana | 2.78 | 1.66 - 4.65 | <0.001 |
| 2015 Beers | 2.19 | 1.34 - 3.58 | 0.002 |
| STOPP version 2 | 1.87 | 1.14 - 3.05 | 0.013 |
| ≥4 diseases | |||
| Overall | 2.78 | 1.39 - 5.56 | 0.004 |
| Winit-Watjana | 2.43 | 1.35 - 4.37 | 0.003 |
| 2015 Beers | 3.04 | 1.76 - 5.26 | <0.001 |
| STOPP version 2 | 1.01 | 0.58 - 1.75 | 0.980 |
| ≥4 prescriptions | |||
| Overall | 0.97 | 0.50 - 1.87 | 0.927 |
| Winit-Watjana | 1.10 | 0.61 - 1.98 | 0.743 |
| 2015 Beers | 2.79 | 1.67 - 4.67 | <0.001 |
| STOPP version 2 | 1.20 | 0.70 - 2.04 | 0.507 |
DISCUSSION
This study examined the prevalence of PIMs in a PCU in Thailand using three criteria: the 2015 Beers, the STOPP v2, and Winit-Watjana. In general, three out of four patients aged >65 years were prescribed PIMs at least once, and more than half of all prescriptions included at least one PIM.
Using 2015 Beers, the prevalence of PIMs per person was high (59%) compared to other studies, such as 22.6% in Europe, 38.5% in Ireland and 51.8% in Brazil.9,12,16 However, all of these studies used previous versions of Beers criteria that comprised less criteria for PIMs than the 2015 versions. The prevalence of PIMs in our study may have been similar to these studies if the same criterion was applied. The most common PIMs identified by the 2015 Beers criteria in our study included orphenadrine, dimenhydrinate, and benzodiazepine, which, in the majority of cases, were prescribed for acute symptoms.
Using the STOPP v2, the prevalence of PIMs per person was similar or lower (40.3%) when compared to other studies, such as 40.4% in Spain and 51.0% in Ireland.17,18 The most common PIMs identified by STOPP v 2 in our study were similar to those by the study in Ireland including Benzodiazepine, PPIs and the first-generation antihistamine.18 When compared to the 2015 Beers criteria, the STOPP v2 was better in identifying the use of strong opioids and NSAIDs but not in identifying the use of medications with anticholinergic effect.
To the best of our knowledge, no prior studies have used Winit-Watjana’s criteria, so our findings cannot be compared. Similar to the 2015 Beers criteria, orphenadrine and benzodiazepines were common PIMs. Of the five most common PIMs, three were medications for acute conditions, such as muscle strain and vertigo.
In comparing the findings from the three criteria, Winit-Watjana identified the highest prevalence of PIMs, followed by the 2015 Beers and the STOPP v2. Flunarizine and ACEIs, which were not PIMs according to the 2015 Beers and the STOPP v2 criteria, accounted for 27.5% of PIMs according to the Winit-Watjana criteria. The three criteria used in our study identified the following different PIMs: NSAIDs were best detected by the Winit-Watjana criteria; orphenadrine by the Winit-Watjana and the 2015 Beers criteria; and 1st generation antihistamine and PPIs by the 2015 Beers and the STOPP v2 criteria. All three sets of criteria identified benzodiazepines as PIMs, while only the STOPP v2 detected opioid use as PIMs. A difference in the PIMs prevalence among these three criteria does not reflect which criteria are better than the others. Instead, the findings show differences in the usefulness and limitations of these criteria. For example, the Beers and STOPP v2 criteria include medications that are unavailable in our country, while the Winit-Watjana criteria are out-of-date and need a revision.
For all the criteria, the most common PIMs were for acute conditions, such as muscle strain and dizziness, and for chronic conditions that required an intermittent use of medications such as insomnia and low back pain. In contrast, PIMs for chronic diseases that required continuous use of medications, such as cardiovascular disease and diabetes, were less common than PIMs for acute conditions. Thus, compared to medications for chronic conditions, elderly patients with acute conditions have a greater risk of receiving PIMs.
There are two possible reasons for the high prevalence of PIMs in our study. First, the PCU did not have alternative medications, such as diazepam, so doctors had no choice but to prescribe PIMs. Limited knowledge about PIMs among physicians, especially residents in family medicine and internists, might be another reason contributing to the prescription of PIMs. A previous study documented a higher prescribing error rate in younger than in older physicians.19 Young physicians in the PCU in our study, specifically internists, rotated monthly with other specialties, such as surgery and pediatrics. Therefore, the limited time spent in the PCU may compromise their learning.
For some common PIMs in our study, such as PPI, ACEIs, and orphenadrine, there is evidence supporting the use of these PIMs with indications. Avoidance of these medications, therefore, may be inevitable. For example, the long-term use of PPIs (≥8 weeks) is indicated in patients with chronic relapsing GERD and NERD and those that require PPIs for gastroprotection.20,21 Similarly, ACEIs are recommended medications listed in standard guidelines for hypertension.22 Unlike PPI and ACEIs, skeletal muscle relaxants, such as orphenadrine, in combination with NSAIDs are indicated for the short-term use in acute musculoskeletal pain when pain relief is not achieved with NSAIDs alone and patients tolerate adverse drug reactions.23 The intermittent use of orphenadrine in Thai primary care is also preferable for patients with chronic musculoskeletal pain despite a lack of evidence.23 The use and avoidance of PIMs, therefore, do not depend only on these criteria but also other evidence and physicians’ clinical knowledge and experiences. For those, who prefer a patient-centered approach, may continue prescribing PIMs until a patient feels confidence to stop taking the PIMs.24
The prevalence of the use of 2 or more medications that were identified as PIMs was low compared to that of other studies.25 All three criteria include a list of concurrent use of 2 or more medications, but the criteria are not comprehensive.4,5,13 Doctors should be aware of this limitation. However, the use of 2 or more medications included in these criteria are common in the elderly, potentially resulting in negative health outcomes, and increased awareness among doctors is necessary for them to avoid the use of 2 or more medications that were identified as PIMs.
The most common use of 2 or more medications in our study were duplicate drug classes and two medications with anticholinergic effects, which may lead to unpleasant symptoms, such as dry mouth and acute urinary retention. Both were identified by the STOPP v2, while the 2015 Beers criteria included only the latter. The STOPP v2, therefore, is better than the 2015 Beers and Winit-Watjana criteria for detecting drug class duplications. Our study also identified the use of NSAIDs with other medications, including aspirin and warfarin, which leads to a higher bleeding risk if combined.
Our findings support those of other studies showing that polypharmacy is associated with PIMs.3,26 In comparing the three criteria, the association between polypharmacy and PIMs was highest according to Winit-Watjana, followed by the 2015 Beers and the STOPP v2, which can be explained by a difference in PIM proportions in each criterion. Thus, reducing unnecessary medications may prevent doctors from prescribing PIMs. Nevertheless, findings from a recent review of the effectiveness of interventions to reduce PIMs are inconclusive.27 In addition, the lack of alternative medications to PIMs may leave doctors no choice but to prescribe PIMs. Hence, offering alternative medications in PCUs will provide an opportunity for doctors to prescribe safer medications for the elderly.28
While an increased number of comorbidities is associated with polypharmacy, its association with PIMs is unclear.11,29 Our study found a positive association, consistent with the findings of Prasert et al.15 The small difference between our study and that of Prasert et al. was the number of diagnoses: ≥4 vs. ≥5, respectively.15 Hence, doctors need to be aware of and assess PIMs in patients with multiple comorbidities. This notion is especially relevant in Thailand, where patients have the freedom to visit multiple doctors for each health condition. The number of prescriptions as a risk factor for PIMs has not been well examined in other studies.3,11,15 Our study found that the number of prescriptions was not associated with PIMs.
In our study, age and gender were not associated with PIMs across all criteria. Previous studies have reported mixed results.3,11,15 However, the findings in our study indicate that gender is associated with the PIMs identified by the Winit-Watjana and 2015 Beers criteria, in which female patients had a slightly higher likelihood of using PIMs than did male patients.
The strengths of our study include that prescriptions from the past 12 months were collected to decrease the effects of seasonal influence and that clinical reviews were conducted with two doctors and one pharmacist to ensure the accuracy of PIM identification. Our data were collected from a PCU where doctors work full-time. This situation is different from that of some PCUs in Thailand, where doctors generally visit at least twice a month. In addition, available medications in the Kukot PCU may differ from those in other PCUs without full-time doctors. Hence, the findings from our study may not be generalized to other PCUs with different contexts. However, the findings indicate a need to explore PIMs in general PCUs and implement evidence-based interventions to reduce PIMs in the elderly.














