INTRODUCTION
Metabolic syndrome (MS) is a term used to characterize cardiovascular risk factors and is characterized by the association of various abnormalities, such as central obesity, dyslipidemia, type 2 diabetes mellitus and systemic arterial hypertension (SAH). There are different definitions for diagnostic criteria, all using combinations of risk factors 18,25. MS frequency is significantly associated with favorable socioeconomic conditions, sedentary lifestyle, hypercaloric diets and high body mass index (BMI) 20. The global prevalence of MS varies depending on the region, urban or rural environment and characteristics of the population studied, like sex, age, race and ethnicity 16.
Considering that obesity is one of the risk factors for MS, the literature has pointed out the important role of genetic susceptibility for this disease development 23, including gene interactions and gene-environment interactions 19. Recent studies show the existence of several single nucleotide polymorphisms (SNPs) located in different genes involved in lipid metabolism and insulin resistance, which play a role in obesity development 22.
In this context, the proprotein convertase subtilisin/kexin 9 (PCSK9) gene encodes the glycoprotein PCSK9, which plays an important role in the cholesterol metabolism. Its expression modulates the number of low-density lipoprotein (LDLR) receptors 28 and affects the circulating levels of low-density lipoprotein cholesterol (LDL-cholesterol), the main carrier of cholesterol in humans 11. The polymorphism E670G (rs505151) of the PCSK9 gene promotes increase of PCSK9 activity (gain of function), leading to reduction of LDLR and consequently, raise in plasma LDL-cholesterol, contributing to hypercholesterolemia 11,28.
Another gene that regulates cholesterol synthesis is the insulin-induced gene 2 (INSIG2), which encodes the INSIG2 membrane protein 8. INSIG2 inhibits sterol synthesis in the liver when the cells have an adequate supply of these (29). Deregulation of this process may affect lipid metabolism and increase insulin resistance 3, which are closely related to MS 4. Some studies have shown an association between the polymorphism rs7566605 of INSIG2 gene and obesity 13,29.
Furthermore, the fat mass and obesity-associated gene (FTO) encode an important protein whose exact physiological function remains unknown. Studies indicate that this gene plays a role in the nervous and cardiovascular systems and has a strong association with BMI, obesity and type 2 diabetes development 30,31. The FTO gene has several know SNPs, many of them related to BMI, with emphasis in rs9939609. In this case, the A allele is directly related to a greater accumulation of body fat, especially in homozygous form (AA).
In line with this, the objective of the present study was to evaluate the association between polymorphisms of INSIG2, PCSK9 and FTO genes with anthropometric, biochemical characteristics and presence of metabolic syndrome in patients with severe obesity.
MATERIALS AND METHODS
SUBJECTS
This is a cross-sectional study, which enrolled individuals of an ethnic mixed population 27, aged between 18 and 60 years, with grade II or III obesity. The sampling procedure is fully described elsewhere and anthropometric and biochemical data of the studied population were already published by our group 21. Pregnant and lactating individuals, patients on hypocaloric diets and who underwent bariatric surgery were excluded. The study was conducted after approval by the Ethics Committee of the institution and according to the Declaration of Helsinki. All participants provided a written informed consent.
Questions about personal history of chronic diseases such as diabetes mellitus, systemic arterial hypertension, cardiovascular diseases and obesity were collected from medical records.
ANTHROPOMETRIC AND BLOOD PRESSURE (BP) MEASUREMENTS
For anthropometric evaluation, the following indicators were used: weight (kg), height (m), BMI (kg/m2) and abdominal circumference (AC). According to Nicoletti et al. (2016), patients were weighed in a Filizola® scale-type digital scale, with a capacity of 300 kg and an accuracy of 0.2 kg. For height measurement, a vertical rod with a graduation of 0.5 cm was used. BMI was obtained using the formula: BMI = P/A2, where P is the weight in kilograms and A is the height in meters. AC was measured by passing an inextensible metric tape with graduation of 0.1 mm on the largest circumference.
The systolic and diastolic blood pressure (BP) was measured using the indirect method with an auscultatory technique using a mercury column sphygmomanometer, with cuffs appropriate for obese individuals. The procedure was performed with the patient after five minutes of rest in a quiet environment, without having practiced physical exercises for at least 60 minutes and without having smoked in the 30 minutes before the procedure.
BIOCHEMICAL EVALUATION
Plasma total cholesterol (TC), low-density lipoprotein (LDL-cholesterol) and high-density lipoprotein (HDL-cholesterol), triglycerides (TG) and glycemia were analyzed in blood samples after 12 hours of fasting. As a protocol of the service, the dosages of TC, HDL-cholesterol and TG were performed by automated colorimetric method. LDL-cholesterol values were calculated by the Friedwald formula: LDL-cholesterol = TC - (HDL-cholesterol + TG/5), with TG values lower than 400 mg/dl 9.
METABOLIC SYNDROME IDENTIFICATION
MS was defined according to the criteria of the I Brazilian Guidelines for the Diagnosis and Treatment of Metabolic Syndrome, which uses diagnostic standards similar to the National Cholesterol Education Program-Adult Treatment Panel III (NCEP ATP III). In this case, MS was defined by a group of cardiovascular risk factors, characterized by at least three of the following components: a) abdominal circumference > 88 cm; b) fasting glycemia > 100 mg/dl; c) triglycerides > 150 mg/dl; d) HDL cholesterol < 50 mg/dl; and e) increased blood pressure: systolic blood pressure (SBP) ≥ 130 mmHg or diastolic blood pressure (DBP) ≥ 85 mmHg.
DNA EXTRACTION AND ANALYSIS OF GENETIC POLYMORPHISMS
DNA was extracted from peripheral blood samples using a GE Healthcare kit according to manufacturer's protocol. The genotypic determination was performed by the allele discrimination method in real time PCR (polymerase chain reaction), which allows the analysis of the variants in a specific segment of DNA. The single nucleotide polymorphisms rs505151, rs75666605 and rs9939609 were genotyped using TaqMan(tm) Pre-Designed SNP Genotyping Assays probes (Applied Biosystems, Foster City, CA), as specified by the manufacturer.
STATISTICAL ANALYSIS
Data were presented in mean and standard deviation. The Kolmogorov-Smirnov test was used to verify the data normality. The Hardy-Weinberg equilibrium calculations for the polymorphisms were performed by applying the Chi-square test. The t-test for independent samples was used to compare the phenotypic variables among the genotypes of each polymorphism. To analyze the dominant model (DM), individuals with at least one variant allele were grouped and compared with those of the reference genotype. For the recessive model (RM) analysis, individuals with at least one wild-type allele were grouped and compared with those with both variant alleles. The associations of continuous variables with genotypes were analyzed with general linear models controlling for sex and age. Statistical significance was set at 5%, all analyzes being performed in Statistical Package for Social Science software (SPSS version 22.0; Inc. Chicago, IL).
RESULTS
For this study, 150 severe obese subjects (80% females) with mean age of 47.2 ± 10.4 years were selected. The means of systolic and diastolic pressure were 122.2 ± 15.4 mmHg and 78.1 ± 10.0 mmHg, respectively. It was identified that 94.7% of patients presented grade III obesity, 73.3% systemic arterial hypertension, 25% high glycemic levels and 26.4% hypertriglyceridemia. Indeed, abdominal obesity was present in all patients. Thus, taking into account the criteria established by the NCEP, 72.7% of the patients selected for this study present metabolic syndrome.
Genotypic distributions of the polymorphisms rs505151 (X2 = 12.33, p = 0.92), rs7566605 (X2 = 1.72, p = 0.64) and rs9939609 (X2 = 0.82, p = 0.49) exhibited the Hardy-Weinberg equilibrium. The allelic and genotype frequencies of the polymorphisms are presented in Table 1. The genotypes AA homozygous for PCSK9 gene, CG heterozygote for INSIG2 gene prevailed. On the other hand, there was a similar distribution between the AA and TT genotypes for FTO gene.
Table I. Genotypic and allelic frequency of polymorphisms assessed in severe obese patients evaluated in the study (n = 150)

n: number of individuals.
Table 2, Table 3 and Table 4 show the anthropometric and metabolic variables for DM and RM for polymorphisms of PCSK9, FTO and INSIG2 gene, respectively. No difference was observed between the possible genotypes for PCSK9 and FTO gene. However, there was a difference between systolic and diastolic BP and triglycerides levels among the possible genotypes for INSIG2 gene, showing that individuals with at least one variant allele (G) had higher BP and triglycerides values. Linear regression analyses confirm the association between the polymorphism of INSIG2 gene and this variables, even when adjusted for gender and age (Table 5). However, none of the polymorphisms studied was directly related to the risk of MS developing in patients with severe obesity (Fig. 1).
Table II. Anthropometric and metabolic variables for genotypes of the rs505151 polymorphism in the PCSK9 gene in subjects with severe obesity (n = 150)
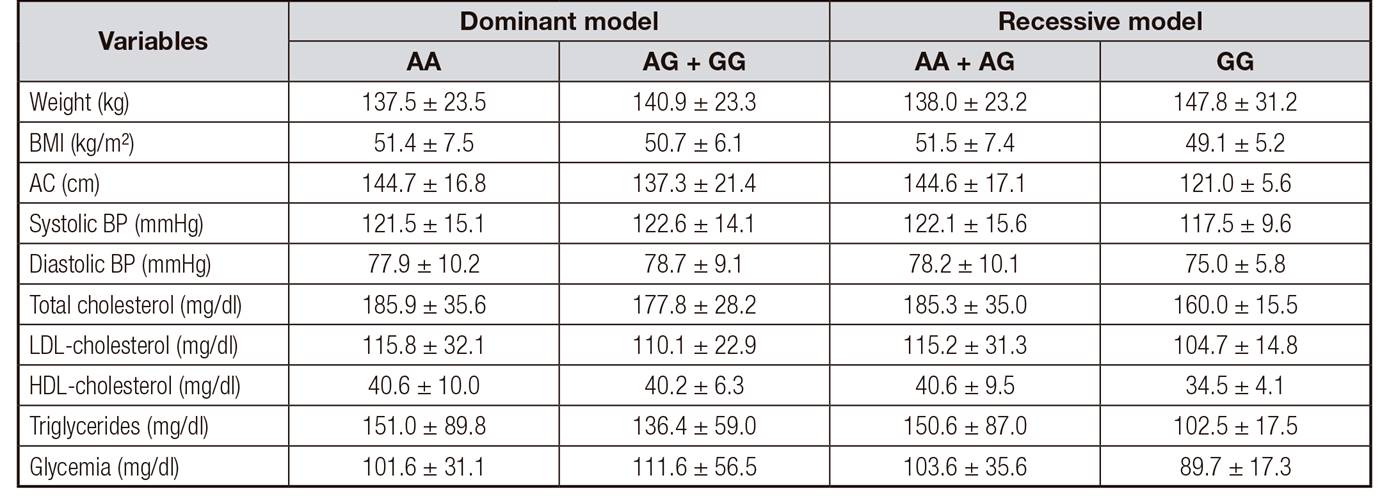
BMI: body mass index; AC: abdominal circumference; BP: blood pressure; LDL-cholesterol: low density lipoprotein; HDL-cholesterol: high density lipoprotein.
Table III. Anthropometric and metabolic variables for genotypes of the rs9939609 polymorphism of FTO gene in severe obese subjects evaluated in the study (n = 150)

BMI: body mass index; AC: abdominal circumference; BP: blood pressure; LDL-cholesterol: low density lipoprotein; HDL-cholesterol: high density lipoprotein
Table IV. Anthropometric and metabolic variables for genotypes of polymorphism rs7566605 of INSIG2 gene in severe obese evaluated in this study (n = 150)

BMI: body mass index; AC: abdominal circumference; BP: blood pressure; LDL-cholesterol: low density lipoprotein; HDL-cholesterol: high density lipoprotein.
*p<0.05
Table V. Multiple linear regression analysis, adjusted for sex and anthropometric variables, showing the contribution of the polymorphism rs 7566605 in the INSIG2 gene in the concentrations of triglycerides, systolic and diastolic pressure in subjects with severe obesity (n = 150)

AC: abdominal circumference; CI: confidence interval.
DISCUSSION
No direct relationship was found between the presence of polymorphisms rs505151 of PCSK9 gene, rs7566605 of INSIG2 gene and rs9939609 of FTO gene and the presence of MS. However, it was observed that rs7566605 of INSIG2 gene showed a significant relationship with three parameters analyzed: systolic blood pressure, diastolic blood pressure and triglyceride levels.
Although our findings do not show a direct relationship between PCSK9 polymorphism and BMI or abdominal obesity, some cases have already been well described in the literature 28,32. Moreover, a Chinese study showed an association between the variant allele and HDL-cholesterol and LDL-cholesterol concentrations 2. Evidence supports that in patients with the polymorphism, the expression of the protein was higher, and consequently the circulating LDL-cholesterol concentrations were increased 15. Furthermore, a study investigating the effect of this polymorphism on the risk of cardiovascular diseases (CVD) and stroke showed that in patients with a higher risk of CVD who had the variant allele, LDL-cholesterol concentrations were significantly higher 28.
Regarding the polymorphism of the INSIG2 gene, the literature is still controversial. Our findings corroborate previous studies that did not evidence associations of polymorphism with BMI or waist circumference 1. In contrast, Oki et al. (2009) evidenced that CC genotype is a protective genetic factor against the progression of hypercholesterolemia. The polymorphism of INSIG2 gene does not inhibit fatty acid synthesis, leading to an exaggerated production of cholesterol, fatty acids, triglycerides and phospholipids 3. Other studies found no association between polymorphism and severe obesity, or with phenotypes associated with hypertension 5,13. Thus, the present study is a pioneer to show association between blood pressure levels and this polymorphism. A possible mechanism of increased systolic and diastolic blood pressure would be the fact that the polymorphism increases the production of cholesterol and may contribute for atherosclerosis development, consequently raising the individual's blood pressure. As for the biochemical and metabolic parameters, the wild homozygous form of the INSIG2 (CC) gene was related to the reduction of waist/hip ratio and glycated hemoglobin, which may lead to an improvement in glucose intolerance 6.
The association between the FTO gene and obesity in humans is already well described 7,13. However, the evidence showing the role of this polymorphism in the development of type 2 diabetes mellitus is weak. Li et al. (2008) did not find associations between the gene and waist circumference and fasting glycemia. On the other hand, a recent study by Guclu-Geyik et al. (2016) found an association between polymorphism and MS in men. The mechanism by which the FTO gene leads to the development of obesity is not fully understood. The polymorphism of FTO gene was related to obesity, but not to MS; nonetheless, a predisposition to an increased risk of obesity may trigger other variables such as increased AC, altered fasting glycemia and dyslipidemia.
Thus, as a main finding, despite not being directly associated with the prevalence of MS, the presence of the variant allele of INSIG2 polymorphism could increase the chances of developing the disease. It should be emphasized that our sample size is limited and our analysis was performed in a population of mixed ethnic origin. Low power may have been responsible for lack of significant associations among severe obese individuals, and reaching significance for some of the potential interactions. Moreover, although adjustment by sex and age was performed, residual confounding such as physical active and dietary intake cannot be totally ruled out.
CONCLUSION
Polymorphisms of PCSK9, INSG2 and FTO genes are not associated with the MS prevalence; however, the polymorphism of INSIG2 gene is associated with higher concentrations of triglycerides and blood pressure values, which are also considered as risk factors for the development of the disease.















