INTRODUCTION
Cell proliferation is defined as the balance between cell divisions and cell loss through cell death or differentiation [1]. Uncontrolled cell proliferation is a hallmark of cancer [2]. Tumour growth and progression depend on the alterations of oncogenes and tumor suppressor genes that most likely accumulate during a multistage process leading to imbalance between proliferation and apoptosis [3].
Breast cancer is the most common cancer in women both in the developed and the developing nations [4]. Breast cancer survival rates vary significant globally, ranging from 80% or over in Sweden, North America, and Japan to around 60% in middle-income countries and below 40% in low income countries [5]. The little survival rates in less developed countries can be clarified mostly by the absence of early recognition program, ensuing in a high proportion of women presenting with late-stage disease, as well as by the nonexistence of suitable diagnosis and inclusive management services [4].
Nucleolar organizer regions (NORs) are loops of DNA that are accountable for ribosomal RNA (rRNA) transcription. They are situated in the nucleoli of cells and in the chromosomes 13–15 and 21, 22 in association with proteins [6]. As rRNA molecules are the core locations of protein synthesis, it follows that the number of NORs in each cell nucleus reflects cellular activity [7]. The number and size of argyrophilic nucleolar organizer region (AgNORs) can be measured with staining of tissues or cells with silver stains [8].
AgNORs is associated with the proliferative action of neoplasms. Rising in AgNOR counts may reflect increased proliferative activity of cells [9, 10]. AgNOR counts have been considered in breast carcinoma, but the outcomes have been contradictory. Some studies have proved that quantitative analysis of AgNORs produces a predictive factor in breast cancer [11]. AgNOR has been progressively useful in histopathology research, due to the fact that there may be a potential link between high AgNOR counts and malignant transformation [12, 13]. Hence, the shape, numbers, and distribution of AgNORs within the nucleus might be of possible use in predicting behavior in breast carcinomas.
The monoclonal antibody Ki67 recognizes a nuclear antigen in proliferating cells in the G1-, S-, M-, and G2-phases of the cell cycle [14]. It does not react with resting cells in the GO-phase. Proportion of tumor cells positive for Ki67 measures the tumor growth fraction, i.e., the percentage of tumor cells that are proliferating. Its core drawbacks are that frozen sections or fresh cells are needed, it does not permit identification of the proportion of cells occupying each phase of the cell cycle, other than the GO-phase, and it does not measure the length of the cell cycle. However, because it is a relatively simple technique, Ki67 evaluation is now the most popular method used to measure human tumor kinetics. In this study, we evaluated the correlation of Ki-67 and argyrophilic nucleolar organizer regions (AgNORs) staining expression as proliferation markers in both benign and malignant breast lesions with hormonal receptor status and other clinicopathological parameters.
METHODS
SAMPLE COLLECTION
The present study was carried out on 40 prospective biopsies taken from patients with different type of breast tumor attending histopathological laboratories at Alexandria University, Egypt. They were divided into benign and malignant group as well as control. The benign group entailed 10 cases, while the malignant group consisted of 30 cases. All participants were agreed to participate by signing the consent form and the study was approved by the ethical committee of the department of Histology. Alexandria University, Egypt. All the fresh tissue samples from both groups were fixed in 10% neutral buffered formalin, dehydrated and embedded in paraffin serial. A 5µm thick were subjected to following studies:
AgNOR SILVER STAINING METHOD
Deparaffinized paraffin sections were rehydrated and washed three times in pure water. Drops of silver solution (50% silver nitrate with formic acid gelatin; in ratio of 2:1) was put on each section in the slide. The slides were then placed horizontally in the humidified staining covered container in dark place and were incubated for 45 minutes. Later, the slides were washed in 5% thiosulphate solution for 5 minutes. Thereafter, the slides were washed in running tap water and countered with eosin. Finally, sections were dehydrated, cleared and mounted.
IMMUNOHISTOCHEMICAL TECHNIQUE
Immunohistochemical staining for ki-67 protein, a proliferation marker, was carried out using the labeling avidin biotin complex (ABC). A 5 µm-thick paraffin sections were placed on aminopropyltriethoxy saline-coated glass slides. Sections were deparaffinized, rehydrated and rinsed in distilled water. Slides were placed on a slide holder and submerged in large amount of antigen retrieval (citrate buffer pH 6) in an oven at 95 °C for 20 minutes. Slides were removed from the oven to room temperature and allowed to cool for 30 minutes before proceeding with normal staining procedure. A circle was drawn on the slide around the tissue with a hydrophobic barrier pen. Slides were washed in phosphate buffer saline (PBS) twice 5 minutes each. Endogenous peroxides activity was quenched using 3.0% H2O2 in PBS. Slides were washed in PBS twice 5 minutes each and were incubated with serum blocking reagent for 30 minutes to block nonspecific binding. Slides were washed in PBS twice 5 minutes each, excess buffer was blotted off using pieces of tissue paper. Slides were incubated with primary antibodies of ki-67 over night at 4°C in the humidified chamber. Negative control slides were incubated without adding primary antibody. After 24 hr, slides were allowed to reach the room temperature and then washed in PBS twice 5 minutes each. Slides incubated with conjugated ABC-HRP reagent for 30 minutes in humidified chamber at room temperature, then washed in PBS twice 5 minutes each. Working solution of DAB was prepared and applied to tissue sections, and the reaction was monitored as the chromomeric reaction turned the epitome sites brown. Next step was preceded when the intensity of the signal is appropriate for imaging. Slides were rinsed in distilled water, stained with hematoxylin as nuclear counter stain, and then rinsed in running tap-water until the appropriate color of the hematoxylin appeared. Finally, sections were dehydrated, cleared and mounted. The Semi-quantitative evaluation of the immunohistochemical results of ki-67 protein was arbitrarily graded as (0) none, (1) <1%, (2) 1-10%, (3) 10-50%, (4) >50%.
IMAGE ANALYSIS
The image of each slides of groups were captured using a 40 X objective (Bar = 50) with numerical aperture of a high resolution of (16-bit digital camera (1280X1024 pixel). Images were viewed and recorded using Olympus microscope – equipped with Spot digital camera, using computer program MATLAB software (image J). AgNOR dots numbers were counted per 100 cells through ten images collected under image analyzer by oil immersion magnification. Number of positive immunostaining nuclei by KI67 protein were counted through ten images collected from ten fields for each case under image analyzer by 40Xmagnification.
The measure of the image optical density (IOD) changes of both histochemistry and immunohistochemistry stain in the individual groups. The maximum, minimum and integrity of intensity color based on gray-level acquisition, analysis of the data was carried out by (reading 10 fixed areas in one image, five images for each case). The mean values of each reaction were based on the mean of pixel number. The IOD based on Gray-level transition probabilities in digitized images from dark to light (0 up to 250).
RESULTS
HISTOCHEMICAL RESULTS: AgNORs
Paraffin sections for 30 cases were stained by silver stain and counter stain by the eosin stain. The result appeared homogenous pink color in control slide as a negative stain and appositive black dots color of silver stain in the nuclei of both benign and malignant cases. The black dotes consist of small discrete intra-nuclear dots and large structures resembling nucleoli (uniformly stain) or may be aggregates of dots in nuclear matrix. The slides were examined under the light microscopy using an oil emersion magnifying as shown as Figure 1.
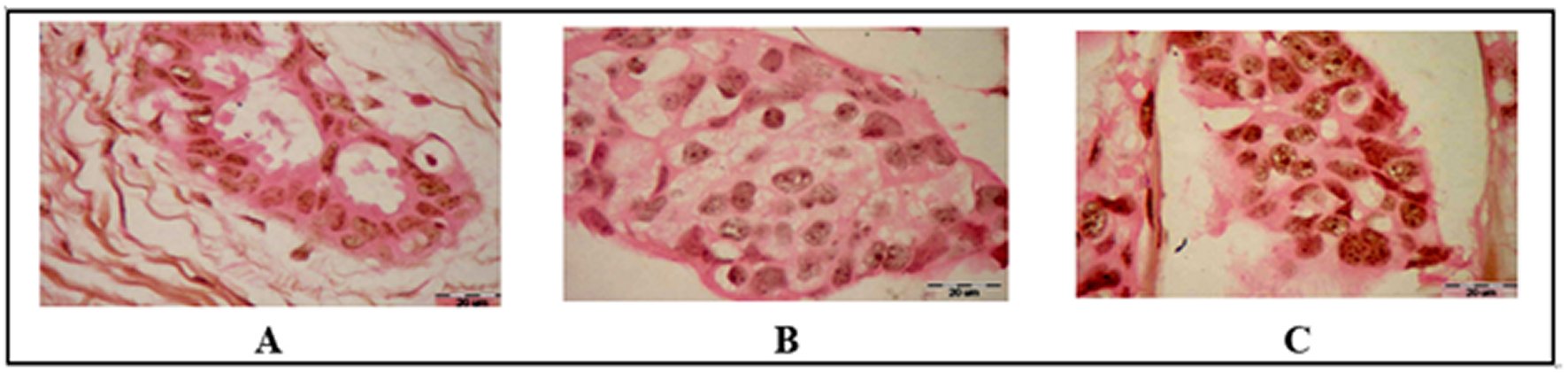
Figure 1. Histochemical Results of tested samples (Silver stain Bar=20 µm).A: Benign Fibroadenoma case show cluster black dotes of Ag NORs in few epithelial breast cells B: Malignant invasive ductal carcinoma (IDC) grade II case, note clusters of black dotes in nucleoli and increase the number of satellite Ag NORs dotes in nuclei matrix. C: Malignant IDC grade III case showing increased number of cluster black dotes of AgNORs in most of the invasive malignant cells.
The mean values of Ag NORs silver stain dots count for normal, benign, grade II and III were 1.28±0.17, 2.83±0.68, 5.23±0.87and 7.32±0.92 respectively. There was a statistically significant difference (p≤0.001) was noticed between the all individual groups, among the normal and breast lesion as well as among the GII and GIII, as shown in Table 1.
IMMUNOHISTOCHEMICAL RESULTS OF KI-67 PROTEIN
Paraffin sections for 40 cases were stained by Ki67 protein monoclonal antibody used avidin biotin complex (ABC) and DAB chromogen as precipitate color stain. The immunohistochemical staining appeared as brown granules stained in the nuclei and the negative control slide was stained with ommited the primary antibody (Ki67 protein) and the blue color indicated to the negative control nuclei. The expression of the Ki67 appeared homogenous golden-brown color in control case and a positive brown granules or diffuse dark brown color in the nuclei of both benign and malignant cases under the 400X magnify examined under the light microscope. Under the high magnify (1000X) the positive nuclei appeared as dark brown granules spread in the nuclei and some nuclei have a coarse granule in nucleoli surrounded by a small granule in nuclei matrix was observed. The results were illustrated in Figure 2 and Table 2.
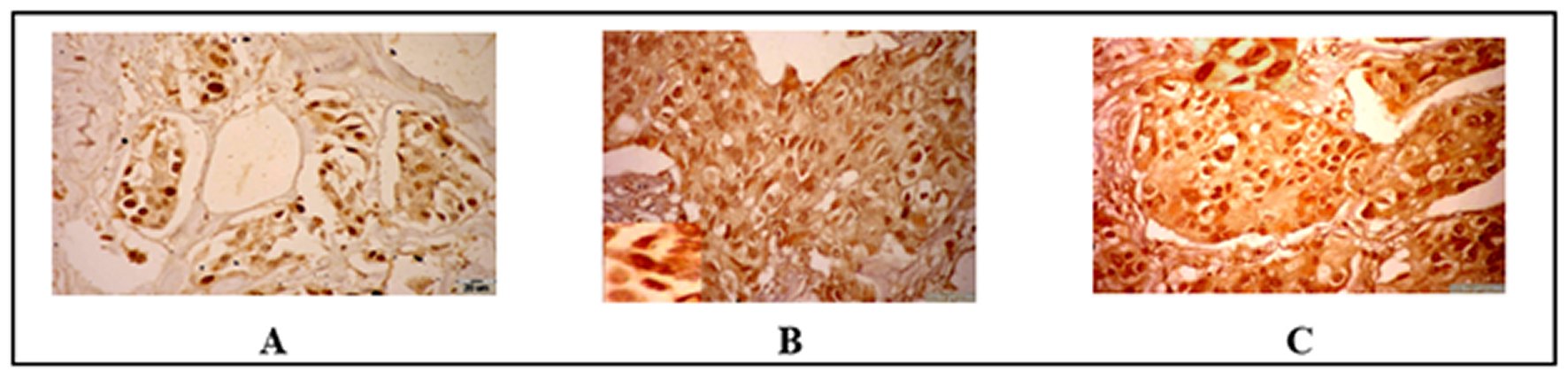
Figure 2. Immunohistochemical results of tested samples (ABC and DAB, BAR=20 µm).A: Benign Fibroadenoma case show a dark positive nuclei of Ki67 expression >20%. B: Malignant IDC grade II case show dark brown granules spread in nuclei ki-67 expression. C: Increased dark brown granules showed spread in nuclei of ki-67 expression. ABC: Avidin biotin complex.
DISCUSSION
Breast cancer is a global serious disease among women with reported annual rates of incidence and death increasing alarmingly [15]. The current outcomes of malignant group showed that 90% of the breast cancer cases were invasive ductal carcinoma (IDC), most of which were allocated to the age range of (>35-55) years and tumor size T2 (>2-5) cm was present (72.2%, 13/18) of GII and (6/12-50.0%) of GIII and lymph node involvement present in (91.6%) of GIII and (8.4%) of GII of malignant group.
Previous studies revealed that 98% of the breast cancer cases were invasive ductal carcinoma (IDC) had the age range of (>35-55) years and the tumor size T2 (>2-5) cm with lymph node involvement was present in 73% of the 30 malignant cases [16]. These results are consistent with several previous studies reported that IDC is the most common histological type of invasive breast cancer, and in the developing world it characterized by an early peak age of onset [17].
Likewise, the majority of the present studied cases were grade II (60%), followed by grade III (40%). Whereas the previous studied reported that the majority (75%) of their studied cases was grade II, followed by grade III (22%) [16]. The present result revealed that IDC grade II is the most common histological type of invasive breast cancer in our country. This result is supported by previous studies claimed that most of the breast cancer cases undergoing surgical resection are at grade II and III [18, 19]. This morphological assessment of the degree differentiation has been provided useful prognostic information in breast cancer and histological grade forms part of the multifactor Nottingham prognostic index together with tumor size and lymph node stage, which is used to satisfy individual patient for appropriate therapy [20].
The malignant cell possesses large irregular nucleoli, that the nucleolus plays a vital role in the control of cell proliferation and protein synthesis. The rapidly dividing cells and cells with mitotic activity have prominent nucleoli [21, 22]. The tumor cell proliferation is a key feature of tumor progression and is widely available for routine assessment by immuno-histochemistry using relevant antibodies [23]. Also, the silver stained nucleolar organization region is very useful tool in breast lesions [22]. Therefore, the present study aimed to assess the proliferation induce through histochemical and immunohistochemical studies and evaluate the correlation of proliferation markers in both benign and malignant breast lesions with hormonal receptor status and other clinicopathological parameters .
The present results showed the positive immunostaining of Ki67 appeared as a large positive coarse granule localized in the nucleolus and spread granules in nuclei matrix. In agreement to this result; the double indirect immunofluorescence studies have revealed that the Ki-67 antigen is associated with reforming nucleoli within discrete domains containing two of the major nucleolar antigens fibrillarin and RNA polymerase I during early to mid G1of the cell cycle, at mature nucleoli the Ki-67 antigen is absent from regions containing RNA polymerase I and appeared within domains containing either fibrillarin or B23/nucleophosmin [24]. In addition, the distribution of the Ki67 antigen in the mitotic cells was studied by the immunofluorescence, immunoelectron and confocal scanning laser microscopy. This study revealed that the ki67 antigen localized as a bright irregular meshwork throughout the nucleoplasm, the antigen appeared in a reticulate structure surrounding the condensed chromosome at metaphase. While it was observed entire nucleoplasm at telophase, that is preceded the typical nucleolar localization pattern in each of the two daughter cells. They have been suggested that the Ki-67 antigen associates with a fibrillarin-deficient region of the dense fibrillar component of the nucleolus. Integrity of this region is lost following either nucleolar dispersal or nucleolar segregation [25].
The current immunostaining examination under the light microscope with high magnify (100X) revealed that the Ki67 antigen is detected at a large number of granules detached foci throughout the nucleoplasm, extending to the nuclear envelope. This feature indicated that the rapidly proliferating cells induce high positive Ki67 protein immunostaining and localized within the nucleolus and nuclear region. In other hand, it was considered as a nuclear acid protein. So, the percentage of cells that display positive staining of Ki67 protein is reported as a board –certify for the pathologist or as prognosis marker . Moreover, the current results showed a high significant (p< 0.001) count of Ki67 positive nuclei in benign groups compared to malignant group in metaphase. Also, a high significant difference was showed in GIII compared to GII in anaphase. This result was documented by Kamel et al. [24] which reported that the monoclonal antibody Ki-67 detected in human nuclear antigen that is present in proliferating cells, but absent in quiescent cells. In the past biochemical, and molecular biology studies investigated on purification of the non-histone chromosome in nuclei and cytoplasm polypeptide, reported that a murine and human nonhistone chromatin protein as Ki67 monoclonal antibody is present in large amounts in malignant and normal proliferating cells, but not in resting cells [26, 27]. Therefore, it has been suggested that the ki67 protein is a nonhistone protein detected in proliferating cells in all phases of the cell division cycle throughout most of interphase .
In the current study, ki-67 protein antibody expression has shown to be highly correlated with tumor histological grade (F 158.400, p ≤ 0.001). This relationship is going in accordance with Dawsett et al. who stated that ki-67 expression was significantly correlated with more advanced grade [23]. However, the current study showed no statistically significant correlation between the immunohistochemical expression of ki-67 protein antibody and the patient's age, tumor size, LNM and LV. As well as the hormonal status stain (ER & PR) and Her2 immunopositively stain, this result is supported by Cazzaniga et al. who showed no difference between the groups regarding the expression of Ki-67, ER, PR, and Her-2\neu [28]. There were some explanations for the prognostic role of Ki-67 expression in breast cancer. Straver et al. reported an association between Ki-67 expression and other prognostic variables (tumor grade, nodal status, tumor size, age at diagnosis, hormone receptors (ER) status, and epidermal growth factor receptor expression, but, the pre-apoptotic marker p53 expression had an inverse association with favorable prognostic markers [29]. This might confirm that Ki-67 expression is an independent prognostic marker by multivariate analysis when other associated factors were used as covariates .
As regard to the Ag (NORs), the current study depended on the morphological appearance of Ag (NORs) dots and the mean AgNORs count/100 cells. AgNORs have been clearly visible in tissue sections as brown/black intranuclear dots upon 1000X magnification. The morphology of silver-stained nucleolar regions (dots) in malignancy was at variance with benign lesions. Dots were large, coarse, often deeply stained, and had variable size and distribution between the nucleoli and nuclear matrix. In agreement to our results, Ansari et al. reported that AgNORs were visible as dark brown or black dots in the nucleus, either singly or in clusters [30]. The dots were fine, round, discrete, and singly dispersed in benign tumors, whereas the dots were larger, coarse, irregularly distributed, and tended to form clusters in malignancy in smear of breast lesions. Therefore, the staining technique is a relatively simple and rapid one and can be applied to both aspiration smears and tissue sections. AgNORs proteins are a set of argyrophilic nucleolar proteins that accumulate in highly proliferating cells while their expression level is low in non-proliferating cells. Some of these proteins remain associated with the nucleolar organizer regions (NORs) during mitosis [31].
Our findings revealed that the mean number of AgNORs dotes/ nuclei determined to 100 cells having <5 AgNORs per nucleus in benign and >5 in malignant tumors in IDC.(98) determination to agree the present result was illustrated the percentage of cells having ≥5 AgNORs per nucleus in both benign breast tumors as well as in infiltrating ductal carcinomas. So, the standardized morphometric analysis has enabled precise and rapid interpretation of NOR counts [30]. The present results showed highly statistical significant (p<0.001) correlation was observed between Ag (NOR)s count/100 cells with normal, benign and malignant group (grade II, III ) respectively. Therefore, the current study revealed that the AgNORs count may be better and more consistent indicator of changes in the proliferative capacity of a breast tumor. This finding is in agreement by some investigators were reported that the number score of AgNORs carried out in different tumour types demonstrated that malignant cells frequently present a greater AgNORs protein amount than corresponding non-malignant cells [31]. A high significant value of Ag NOR (p<0.05) score in malignant with metastasis comparison to those without metastasis [22]. They concluded that AgNORs stain is proven predictability may become a trustworthy milestone . However, the present study showed no statistically significant correlation between the Ag NORs black dots count / 100 cells and the patient's age, tumor size, LNM and LV. As well as the hormonal status stain (ER & PR) and Her2 immunopositively stain. This finding is in agreement with those reported by other previous studies [32]. There is evidence that the quantitative distribution of Ag NORs proteins is a proliferation-related parameter that can be used as a prognostic index in tumor pathology.
Although Ag-NORs scores do not independently provide definite information in relation to prognosis, it is proposed that they could contribute towards prognostication if considered in combination with specific clinic-morphological m parameters [33]. That staining technique is a relatively simple and rapid one and can be applied to both aspiration smears and tissue sections. The manual evaluation of Ag NOR scores is a cost-effective alternative to automated methods of evaluation. In spite of the Ag NORs analysis is carried out through standardized morphometry. However, such techniques are not widely available in developing countries [34]. In addition, the concept of this study is to correlate the proliferation assessment by morphometry counting of AgNORs black dotes by silver histochemstain and positive nuclei of Ki67 immunostaining. The current study was noticed a parallel correlation in normal, benign in the correlation between the AgNORs and ki-67 positive stain were (p≤0.03), that were increased with an increase of the IDC grades (GII &GIII). This observation was agreement with Sharma et al. who attempted to correlate between Ki-67 scores and the mean AgNORs dote counts, there was no significant correlation was observed (p=0.606), although both the parameters (score and count) increased with an increase in the grade of the tumors [35]. Therefore, AgNOR analysis is a really important prognostic factor for several human neoplasms. The experimental and theoretical justifications for AgNOR as a prognostic factor are also reviewed [30]. Furthermore, the new concept of this work was quantitative assessment of the histochemistry and immunohistochemistry stains caries for calculating density of stains by integrated optical density (IOD) using digital image analysis software. The mean values of each reaction were based on the mean of pixel/µm. This concept may be provided the role of the image analysis methodology that influence the measurement and identified a potentially valuable clinical role for Ki67 and AgNORs measurement, as well as the Feulgen DNA stain .
Image analysis evaluation of the current study revealed an increased significant difference (p˂0.005) in malignant group compared to benign individual. Also, the digital analysis proved a high significant difference (p<0.001) between the GII and GIII of the malignant group studies. So, this result could be revealed that the digital data IOD for Ki67 measurement have identified a potentially valuable evidence role of proliferative fraction in breast carcinomas. agreement to this results, the Swiss Working Group of Breast- and Gyneco-pathologists has surveyed inter- and intra-observer consistency of Ki-67-based proliferative fraction in breast carcinomas through the image analysis method (IOD) and showed good to very good agreement in well (G1) and poorly (GIII) differentiated breast carcinomas [36]. Nevertheless, there was a high degree of inter and intra-observer inconsistency in the read-outs of Ki-67 (LI) in moderately differentiated (GII) carcinomas, which is particularly problematic because it is for these “intermediate carcinomas where guidance from the carcinoma's proliferative activity is expected for chemotherapy decisions [37].
Regarding to the IOD AgNORs the present study revealed an increased highly statistically significant in correlation between normal, benign and malignant group (grade II, III) (123.29±5.11) (137.54±4.92), respectively. This result revealed that the expression of AgNOR proteins were measured by quantification of the silver staining level using morphometry and image analysis reflected the role of the Image analysis as a valuable method for assessing tumor cell proliferation . Moreover, the current study Histochemical image optical density (IOD) of Feulgen DNA stain showed a decreased statistically significant correlation was observed in IOD Feulgen magenta color between normal and breast lesion cases. Also decreased significant difference was noticed in benign compared to the malignant individual groups. Therefore, our results could reveal that DNA densities reflect the regional protein association of the DNA. The results illustrated the reporting of the Feulgen dense regions may result from aggregation of a specific class of histones by the alkaline buffer with consequent condensation of the DNA bound to dark band of chromosomes. The Feulgen pale or negative regions may represent those in which non-aggregated proteins, histone and non-histone. This renders that the DNA of those regions subject to diffusion or vulnerable to fragmentation in the Feulgen hydrolysis [38].
In line to our results, the staining intensity is proportional to the DNA concentration through the applications of the Feulgen reaction are concerned with DNA quantification in cell nuclei by image cytometry for ploidy evaluation in tumor pathology, this measurements which are summarized to give the final integrated optical density of the DNA concentration [39]. Therefore, from the image view and recorded by image analyzer of the Feulgen stain or specific demonstration of DNA in cell structures at the light microscopic level may be used and measured the staining intensity proportional to the DNA concentration. Some investigators concluded that image analysis (IA) provides a valuable alternative method for assessing tumor cell proliferation may offer certain advantages over flow cytometry [40].














