INTRODUCTION
Total parenteral nutrition admixtures for neonates (TPNAn) should be tailored and adapted to immaturity, body weight, age, metabolic needs and co-existing diseases. The stability of these admixtures is a critical issue due to the high nutritional requirements involved (1-3). One of the major stability criteria is the particles size (4-6). It is recommended that admixtures should include less than 0.05 % of droplets of fat larger than 5 μm although a safer profile has set the criteria of instability (7) at a maximum droplet size of 1 μm (8,9). Since instability of lipid emulsion is related to high concentration of cations (10), some authors considered TPNAn unsuitable (11-13). Forchielli et al. (8,9) described over the last decade that admixtures for neonates are stable, except for those with fish oil emulsions and exceeding 4.5 mmol/L, where they observed that 12 % of lipid droplets exceeded 1 μm and 2 % exceeded 5 μm. The authors concluded that the interaction between fish oil and calcium strongly suggested an incontrovertible effect on droplet dimension, limiting their use with in order to prevent hepatic toxicity (14). Notwithstanding, the stability of admixtures with fish oil has been confirmed for admixtures with calcium concentration from 6.5 to 12.2 mmol/L in the pediatric age (13,15) and recently in adults (6).
Our Hospital Pharmacy Department compounds every year more than 2500 admixtures with fish oil emulsion and high content of calcium gluconate for a reference neonatal intensive care unit having 42 beds (16). Based on a detailed scientific literature review and on extensive clinical experience as well as practice evaluation, we hypothesized that every individualized admixture prepared with fish-oil emulsion and containing calcium at concentrations ranging from 10 to 20 mmol/L would reveal a distribution of droplet size matching the recommended safety profile for TPNAn.
The aim of this study was to check the stability (particles size < 1 μm) of different individualized TPNAn prepared with fish-oil emulsion and containing calcium at concentrations ranging from 10 to 20 mmol/L.
METHOD
The study material were samples of TPNAn prepared and tested for different storage periods at Madrid University Hospital 12 Octubre, Spain. The study protocol was approved by the reference Clinical Research Ethics Committee (CEIm 21/202).
ADMIXTURES
TPNAn were prepared in horizontal laminar flow hood following the protocols for the preparation of parenteral nutrition of the Hospital Pharmacy Department. Multi-layer ethylene-vinyl acetate bags and photoprotection overbags from Bexen Medical® were used. The sequence of compounding was as follows: amino acids, calcium gluconate, glucose, lipids, vitamins, magnesium sulfate, sodium and potassium salts, zinc, multi-trace element, sodium glycerophosphate.
We tested three types of admixtures:
Twelve different TPNAn formulations with SMOFlipid® 20 % (Fresenius, soybean-olive-fish oil LCT + MCT) and calcium content from 10 to 20 mmol/L (F1 to F12, Table I). These 12 admixtures vary in the amount of amino acids (Primene® 10 %, Baxter), lipids (SMOFlipid 20 %® Fresenius), sodium, potassium, chloride, phosphate and calcium gluconate. The same amount of dextrose, trace elements (Peditrace® Fresenius-Kabi), vitamins (Soluvit® + Vitalipid Infantil®, Fresenius-Kabi), zinc (Meinsol-oligozinc®, Fresenius-Kabi), magnesium (Sulfato de magnesio Altan® 150 mg/mL, Altan) were added to every admixture.
Table I. Composition of the TPNAn formulations under investigation (n = 18).
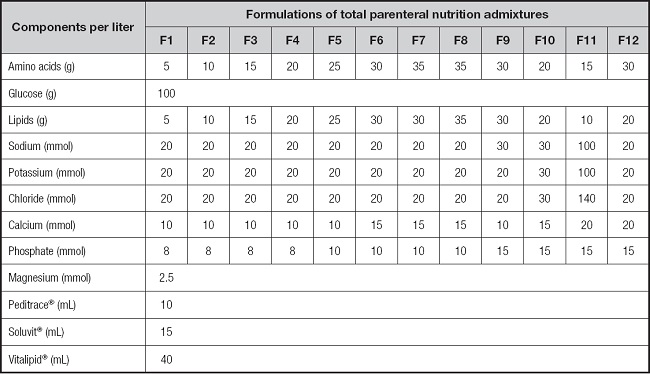
Formulations F1 to F12 (n = 12); and additionally, F1, F7 and F11 were also tested with Lipoplus® 20 % (n = 3) in substitution of SMOF Lipid® 20 % (F1-L, F7-L and F11-L), and with SMOF Lipid® 20 % + Multi-12/K1® Pediatric (F1-M, F7-M and F11-M) 15 mL/L (n = 3) in substitution of Soluvit® + Vitalipid®.
Two different bags of these 12 admixtures were stored at 4 oC for 24 h and two at 4 oC for 96 h. Two samples per bag were tested.
TPNAn F1, F7 and F11 with Lipoplus® 20 % (PN-L), (Braun, soybean-fish oil LCT + MCT) instead of SMOFlipid® 20 % (F1-L, F7-L & F11-L). Two samples per bag were tested after 96 h of conservation.
TPNAn F1, F7 and F11 with SMOF Lipid 20 % and Multi-12/K1® Pediatric (Sandoz-Novartis) (F1-M, F7-M & F11-M), instead of Soluvit® + Vitalipid Infantil®. Two samples per bag were tested after 96 h of conservation.
All bags were checked for color changes, creaming, coalescence or cracking. The samples were obtained directly from bags to sterile syringes after agitationat room temperature, and immediately checked for pH and particle size.
LIPID PARTICLE SIZE
Particle standardized diameter was measured by a laser diffraction technique (Mastersizer 3000® Malvern Panalytical, ISO 13320 certified) in the IESMAT laboratory (www.iesmat.com) with a range of optimal measurement from 0.01 to 100 μm. The technique included dispersion in distilled water with homogenization at 2200 rpm (Hydro EV®, Malvern Panalytical). We analyzed the sample volume required to obtain a laser overshadow between 6 % and 8 %. A refractive index of 1.529 and absorption of 0.01 were applied, following recommendation by ISO13320 for very small particles (17).
PH MEASUREMENT
The pH of every admixture bag was measured with a calibrated pH meter (pH-Meter BASIC 20®, Crison, Barcelona, Spain) equipped with a glass electrode (5011T; Crisol, Barcelona, Spain). The sensitivity value of this technique is 0.01.
DATA ANALYSIS
The particles size distribution was described with the following outcome parameters: 10th percentile (D10), median (D50), 90th percentile (D90), proportion of particle size > 0.6 μm and maximum size. Data were analyzed with mixed linear regression models where each bag was considered as a class to take into account the non-independence of the samples measurements from the same bag. Models included storage time and admixture components as covariates. The data analysis for this paper was generated using SAS/STAT 14.2 software, Version 9.4 of the SAS System by SAS Institute Inc., Cary, NC, USA.
RESULTS
PARTICLE SIZE DATA
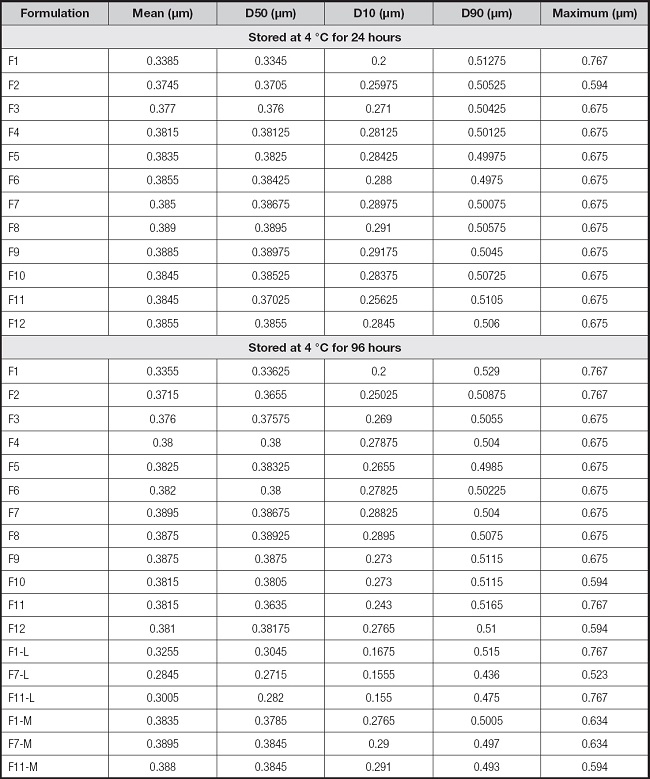
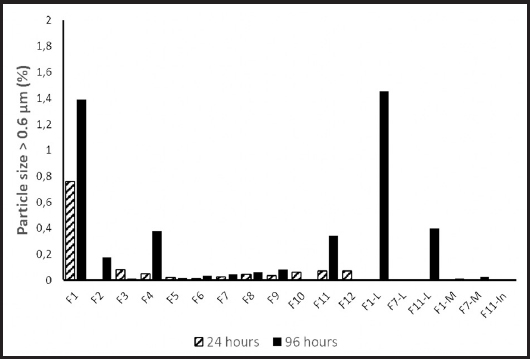
We performed 108 measurements on 54 bags. None of the investigated bags showed color alteration, creaming or coalescence with phase separation. The values obtained in each formulation at 24 and 96 h of conservation are presented in figure 1 and table II. Maximum particle size was < 0.8 μm for all investigated admixtures. The largest particle size detected in any of the mixtures was 0.767 μm (F1 at 24 h and F1, F2 and F11 at 96 h with SMOF®; F1 and F11 at 96 h with Lipoplus®) (Fig. 1). The percentage of particles > 0.6 μm only exceeded 0.5 % in the mixtures with 5 g/L of lipids (F1).

Figure 1. Particles size of all investigated admixtures ((▲) maximum size detected; average of (○) D10, (●) D50 and (○) D90. A. Admixtures stored for 24 h; B. Admixtures stored for 96 h. Standard deviations have a magnitude of 10-3 μm and are not represented.
EFFECT OF STORAGE TIME ON PARTICLE SIZE
Storage for 96 h increased the percentage of particles > 0.6 μm (+0.143 ± 0.07; p = 0.038), but did not impact other parameters (Fig. 2).
EFFECT OF COMPONENTS ON PARTICLE SIZE
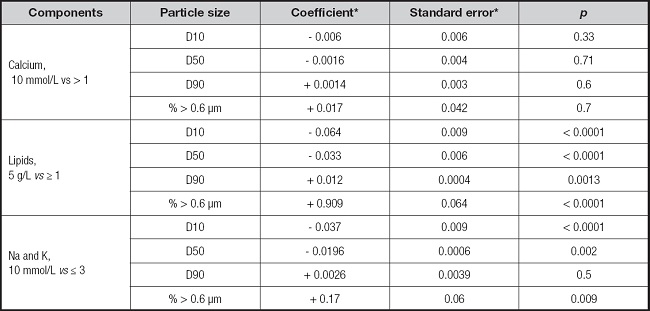
The effect of different components from admixture bags on particle size are reported in table III. Low lipid concentration (5 g/L vs. ≥ 1) significantly increased the proportion of particles > 0.6 μm and D90, although D50 and D10 decreased. No association with calcium composition was observed. Sodium and potassium concentrations of 100 mmol/L slightly increased the proportion of particles > 0.6 μm, and D50 and slightly decreased D10 (Table III).
DISCUSSION
We provided strong evidence that calcium concentrations from 10 to 20 mmol/L and individual monovalent cations up to 10 mmol/L are compatible with individualized admixtures for neonates containing emulsions with fish oil and that these TPNAn remain stable up to 96 h after preparation. Previous studies demonstrate stability of other components of TPNAn (8,18).
The investigated admixtures were designed to meet the requirements of newborns including preterm infants. No relation was found between particle size and calcium content between 10 and 20 mmol/L. Size distribution consistency between different admixtures also supports the stability of our formulations. Only admixtures with a minimum amount of lipids (5 g/L) or maximum cations content (calcium 20 mmol/L, sodium 10 mmol/L and potassium 10 mmol/L) showed wide particle size distribution with a small but significant proportion of particles > 0.6 μm, but also with maximum size < 0.8 μm.
The three additional formulations investigated had a standard composition for the first day of life, complete parenteral nutrition and admixtures with high cations content. No significant differences were observed between SMOFlipid® 20 % and Lipoplus® 20 % formulations at 96 h of storage. The substitution of vitamins source from separate water-soluble and fat-soluble to complete vitamins mixture did not modify particle size.
The differences in pH were inversely related to the concentration of amino acids. The storage time had a limited influence and the glucose content was not analyzed because all the mixtures were dosed with the same amount.
These results support the safe use of fish oil emulsions with concentrations of cations that meet the usual requirements of newborns, including premature infants, as described also in the admixtures for children and adults (6,13,15).
Although the number of samples is limited, it represents the spectrum of common formulations for newborns, specifically in terms of lipids and cation content.
In conclusion, the studied individualized parenteral nutrition admixtures for newborns that contain emulsions with fish oil and meet cation requirements are stable for at least 96 hours.