This is a highly subjective selection of papers that caught my attention published from January 1st 2023 through January 31st, 2024.
Although, for years, it has been known why vitamin D deficiency is clearly associated with the presence of non-bone diseases—also in longitudinal studies—and the appearance of these conditions, the first studies ever reported on supplementation vs placebo did not prove beneficial in these conditions (cancer, diabetes, hypertension, cardiovascular disease, etc.). It quickly became clear to me that many publications were being conducted with low doses, using short regimens, or with “sufficient” populations of 25OHD.
After delving into these articles, interesting topics emerged:
-
Some beneficial effects of supplementation occur “in many” but not “in all” patients due to genetics. Studies have been published (in my opinion with very little impact) showing benefits in subgroups of people with some genetic polymorphisms and not in others. The first paper was published years ago by Baron in England, United Kingdom (1). He published that in subjects with colonic polyps, supplementation with vitamin D did not prevent the appearance of new polyps. However, the next year, when they genetically studied that population, they found that polyps had been prevented in the subgroup that had the rs7968585 polymorphism among individuals with the AA genotype (26 %) in the vitamin D receptor (VDR) gene. It is in this subgroup that the supplementation of vitamin D was able to reduce the appearance of new polyps by 64 % (2).
The first new study on this subject is of that same style. It has a first “negative” part and a second, new “positive” part. This is the AMATERASU study conducted by Urashima et al. (3), which, in 2019, showed that administering 2000 IU/d for 3.5 years vs placebo did not prevent recurrences or improve survival in Japanese subjects with digestive cancers. Now, it is postulated that the key to achieving a certain “extraosseous” benefits is not the dose administered but to what extent 25OHD levels were reached. In the subgroup with < 20 ng/mL at baseline, a mean of 36 ng/mL was reached. In the subgroup with between 20 and 40 ng/mL at baseline, they reached a mean of 45 ng/mL. In the analysis of subgroup study, lower recurrence and higher survival were demonstrated… but in which subgroup? In those who had very little vitamin D (< 20 ng/mL) or in those who had enough for the IOM criterion (> 20 ng/mL)? The answer is that it was prevented in the subgroup that had more 25OHD (HR, 0.44), which are those that reached a mean value > 40 ng/mL. The authors highlighted this finding with caution. At that time, it was not postulated that perhaps 40 mg/mL could be a cut-off value to achieve to prevent or alleviate some non-classic conditions. In this study, polymorphisms of the VDR receptor and the transporter protein were analyzed, and no relationship with benefit was ever found. The new study by this group—whose lead author is now Kanno (4)—studied how many of these subjects were “immunoreactive to the onco-suppressor protein P53”, defining them by having serum antibodies against P53 (+) or an accumulation of that protein in more than 99 % of cancer cells. The hypothesis was that supplementing with vitamin D can increase the anti-cancer immunoreaction only in a subgroup, and results proved it: in the P53 (+) subgroup, relapse or death occurred in 9/56 subjects (16.7 %) vs 14/26 with placebo (53.8 %), while no differences were reported in the P53 (-). In my opinion this manuscript did not achieve the impact it deserves. It is clear that the answer to 25OHD in some non-classical diseases—cancer in this case—may depend on genetics or the presence of certain onco-suppressive proteins.
-
Another re-analysis: supplementing with vitamin D prevented the appearance of autoimmune diseases in the vital study. In 2021, Hanh published that in the 5.3-year follow-up the appearance of an autoimmune disease was confirmed in fewer supplemented patients vs the placebo group (HR, 0.61; 0.43-0.86), concluding that it reduced them by 25 % to 30 %—especially rheumatoid arthritis—and after 2 years of supplementation (5).
What is new about this topic?: Well, 2 years later (at the end of the 7th year), Costenbader published that prevention was no longer significant (patients no longer had supplementation) yet the condition appeared: psoriasis was prevented (HR, 0.61; 0.38-0.98). The other finding is that categorizing them by their BMI, those whose BMI was < 25 kg/m2 maintained prevention (HR, 0.75; 0.59-0.95). Of note, the main limitation of this study, which did not prevent any positive results: participants were allowed the voluntary use of vitamin supplements (6).
Vitamin D supplementation improves outcomes in women with gestational diabetes. A meta-analysis (7) that analyzed 20 RCTs concluded that those supplemented increased their HDL-C levels, decreased their LDL-C and triglyceride levels, and reduced the risk of premature birth, hyperbilirubinemia and neonatal hospitalization. Of note what doses these women (mostly from Iran and China) received. Another topic is the prevention of gestational diabetes, which we have analyzed with other “outcomes” in a review (8).
A work as fascinating as it is complex is “Embryonic vitamin D deficiency programs hematopoietic stem cells to induce type 2 diabetes mellitus”. The authors study in mice the epigenetic mechanisms that influence the risk of intrauterine diabetes associated with vitamin D deficiency. Studies in rodents confirm the relationship among intrauterine vitamin D deficiency, inflammation, hepatic steatosis, excess adiposity and insulin resistance (IR), suggesting it induces epigenetic programming. It has been suggested that the VDR receptor plays a role in programming the immune system during embryogenesis. In these studies, vitamin D-deficient fetal hematopoietic stem cells (HSCs) were transplanted into the uterus of vitamin D-sufficient mice inducing diabetes. These studies show that the deficit epigenetically suppresses Jarid2 expression and activates the Mef2/PGC1 step in HSCs, which results in adipose infiltration into macrophages, which secrete miR106-5p, which in turn promotes IR. Vitamin D-deficient monocytes in the human cord have, these studies state, the above-mentioned changes, causing IR. The authors conclude that this vitamin D deficiency during development has enough epigenetic consequences on the immune system to trigger IR (9).
Vitamin D and Parkinson's disease (PD). Two meta-analyses from 2017 and 2018 confirmed the association between 25OHD deficiency and PD risk and severity (10,11). What is new is the appearance of a meta-analysis on supplementation. It only includes 5 articles (4 on PD and 1 on restless legs syndrome). Some studies showed improvement, especially 1 from Poland where 4000 IU/d were administered only for 3 months to patients with BMI < 25; 5000 IU/d with BMI = 25-30 and 6000 IU/d with BMI > 30. Another study with a 4-month regimen of 10,000 IU/d only seemed to improve balance in young participants, while the remaining studies which administered 1000 IU/d and 1200 IU/d found modest improvements. The authors conclude that screening for vitamin D deficiency and supplementation might be necessary in patients with PD (12).
-
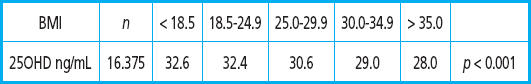
A Vital sub-study showed that the increase in 25OHD observed with 2000 IU/d supplementation was lower in obese and overweight subjects. What was the extent of this 250HD increase based on their respective BMI?
This may partly explain higher or lower responses to health objectives that were found in other megatrials (13).
-
Vitamin D and uterine fibroids, more commonly called leiomyomas, or fibromas. This year (14) Okoro that 75 women with uterine leiomyoma had 25OHD levels of 15.26 ± 4.96 ng/mL while the 75 controls had 25OHD levels of 22.45 ± 6.93 ng/mL (p < 0.001). Additionally, there was a negative correlation between fibroid volume and 25OH Vit D (p < 0.001). In 2013 reports came on the negative correlation between 25OHD and fibroid weight (15) and, in 1997, on the fact that black women had less vitamin D and greater fibroid masses (16).
In 2023 Combs conducted an interesting meta-analysis and concluded that 14 studies confirmed the correlation between 25OHD and the presence of fibroids. A different question is if vitamin D supplementation could reduce the size of leiomyomas? Four out of 5 studies found that compared to placebo, leiomyomas did not grow or shrink. Of note, 2 studies were conducted for as long as 2 months, another 2 for as long as 3 months, and 1 study for as long as 1 year. All participants in all studies were supplemented with 50,000 IU/week, except for the 1-year study in which participants received 50,000 IU/month for 2 months, followed by 2000 IU/day (17).
Finally, a Chinese study conducted after this meta-analysis compared 25 women with fibroids supplemented with a 3-month regimen of 1600 IU/day vs placebo and found a decrease in size with vitamin D. What is striking is that this increase was not very significant (from 10.45 up to 17.14 ng/mL), which are values that stand far away from for the bone health target of 30 ng/mL and even further from the targets suggested for non-classical actions (40 ng/mL) (18).
In conclusion, these are arbitrarily selected studies that, nonetheless, allow us to keep in mind the possibility that vitamin D supplementation can prevent or alleviate multiple non-bone conditions.