Highlights
Nanobots are novel inventions utilized to deliver drugs to the patients with less side effects and toxicity. These bots prove to be very useful and fast acting in delivery of drugs, as well as treatment of some diaeases. Nanobots are in extensive research for the incorporation into the human body to treat any lethal disease by delivering specific drugs and also held some part of work in neural regeneration and cure of some particular diseases.
The main contribution of this review is to determine the lack of information and research that are available in the use of nanobots as drug delivery and also about the types and design utilized for the development of nanobots.
Hence, this review could be the base for future research, to identify various types of nano and microbots. So, it can be a noticeable alternative for the delivery of drugs with faster onset of action and cure.
Introduction
Drug delivery systems (DDSs) are methods for delivering medicinal substances, including topical, injectable, and oral methods. DDSs address complex biotechnology-based treatments by incorporating various drug formulations and delivery systems1,2. Nanotechnology, which manipulates matter at the atomic and molecular level, has enormous potential across various industries, including electronics, construction, and healthcare3,4. The medical sciences have made significant scientific strides due to nanotechnology, with nano oncology emerging as a result of its application in oncology5,6.
Nanotechnology is used to create diagnostic systems, and nanomedicine is the branch of medicine that uses it to diagnose and treat illnesses. Nanoscale material measurement is made possible by technologies like electrochemiluminescence, and cellular imaging using nanoprobes can be used for medical diagnosis7,8. Nanotechnology also aids in the development of pharmaceuticals, enhances their dispersion throughout the body, and targets specific therapeutic sites. The creation of nanobots, controlled nanoscale devices, was prompted by the rapid expansion of robot technology’s use in health and medical science9. Nanobots are accurate, versatile, and flexible, consisting of sensors and motors that undergo conformational changes to release a chemical that acts against trouble-causing intruders. The development of nanobot drug delivery systems is the result of developments in bioinformatics, robotics, nano structuring, medicine, and computing10.
Nanobots come in various forms, including reciprocate, microbivore, surgical, and cellular repair nanobots. Carbon is the most suitable element for nanobots due to its strength and inertness, and atomic force microscopy (AFM) and scanning electron microscopy (SEM) are used to create a haptic and visual interface for studying their molecular structure. However, the creation and control of these nanobots pose the biggest development challenges11,12.
Objectives of Nanobots13:
Variable physical-chemical characteristics.
High volume and surface area to enable controlled distribution of a large medication payload.
Simple surface functionalization using biomolecules and targeted ligands.
Ongoing observation of the body and feedback.
To change physiological capacities, heal tissues, and clean blood vessels and airways.
To function as an independent on-site surgeon within the human anatomy.
Able to maintain a smooth bloodstream circulation while carrying a larger payload than a tiny molecule.
Mechanism of propulsion of the nanorobot.
Smaller dimensions lead to improved binding and accumulation at particular locations.
Keep a close eye on the patient’s health and be able to spot cancer and other illnesses early on.
Ideal characteristics of Nanobots14:
Nanorobots need to be between 0.5 and 3 microns in size, with 10 nm components.
Greater than mentioned nanobot sizes will obstruct capillary flow.
Because of its passive diamond exterior, it will shield itself from immune system attacks.
It will use carrier wave frequencies of 1-100 MHz to encode messages to acoustic signals in order to communicate with the doctor.
It may replicate itself several times to replace deteriorated units; this is known as self-replication.
Advantages of Nanobots14:
The primary benefits of the bots are their durability and speed.
Reducing unwanted side effects by keeping the drug inactive in regions where therapy is not required.
Because it’s a less invasive approach, little care is needed.
One of the key benefits of nanorobots is their longevity; theoretically, they can continue to function for several years, decades, or even centuries.
One advantage of nanorobots is their ability to eradicate pathogens that enter the body and spread silently.
Disadvantages of Nanobots14:
Upon introducing distinct nanorobots to treat varying ailments, the body may develop clusters inside them.
Installation cost is quite high.
Since every nanorobot is unique in its attributes and features, so too are their effects, and as a result, they could all be extremely detrimental to human health.
Foreign nanobots have the potential to do significant harm to our bodies, just as our own nanobacteria can. Therefore, caution must be used to get over this obstacle.
If nanobots multiply themselves, a dangerous variant of the bots might be produced.
Inability to communicate with a biological system.
Types of robots
Microbots:
Microbots have shown a great deal of promise for carrying out a number of crucial tasks, including medication delivery, cell manipulation, microassembly, and biosensing via manual methods. For instance, magnetotactic bacteria were exhausted under gradients of direct current (DC) magnetic fields to establish tailored delivery. Numerous studies have demonstrated how microbots can use magnetic fields to move target materials like chemicals and cells. Furthermore, a novel system called micro-electromechanical systems has been used to create micro grippers for microrobots; this technology may be used to increase the functionality of microrobots. Motion control is achieved by premeditating the kinematic models of established microrobots. Using separate motion plans, the biological and non-biological microrobots have been guided into the human body for the treatment of various diseases15,16,17.
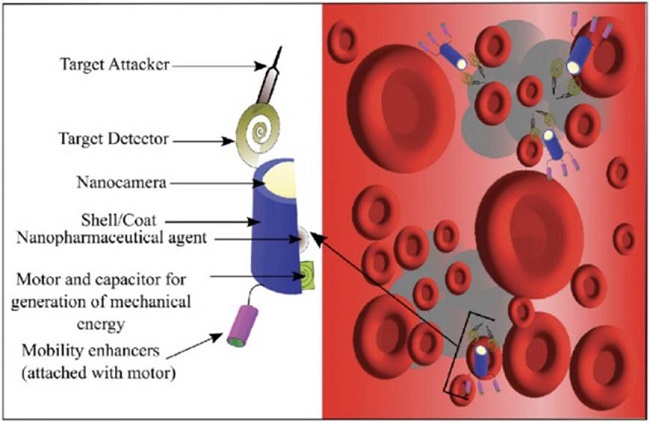
Pharmacyte:Pharmacyte is a medicinal nanorobot that has a 1-2 µm diameter and a 1-3 µm medication retention capacity in its reservoirs, as shown in Figure 2. Mechanical systems are used to organize and regulate categorization pumps.
Chemotactic sensors or molecular markers, which guarantee the accuracy of the targeting mechanism, are used to distribute them. The limited settings-intestinal fluid, blood, and cytosol-are where power, glucose, and oxygen are extracted for the on-panel supply. After completing the work, nanorobots can be removed or recovered using centrifuge nanopheresis15,18.
Microchips:
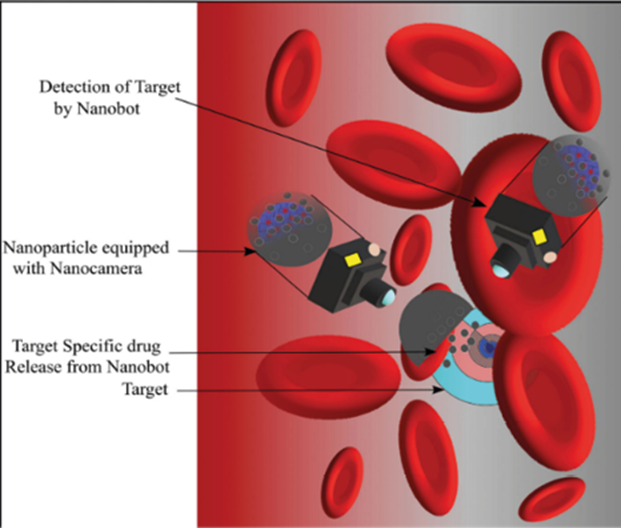
Human molecules overlap microchips carried by nanorobots; when the molecules identify an illness, the chip transmits electrical impulses. For instance, the unique sensor nanobots can detect illnesses and verify blood contents by being injected under the skin. Additionally, they can be used to report blood sugar levels. The advantages include being easy to use and having a low yield charge. Nanorobots utilized for blood vessel imaging and diagnostics are depicted in Figure 3 15,19.
Respirocyte:
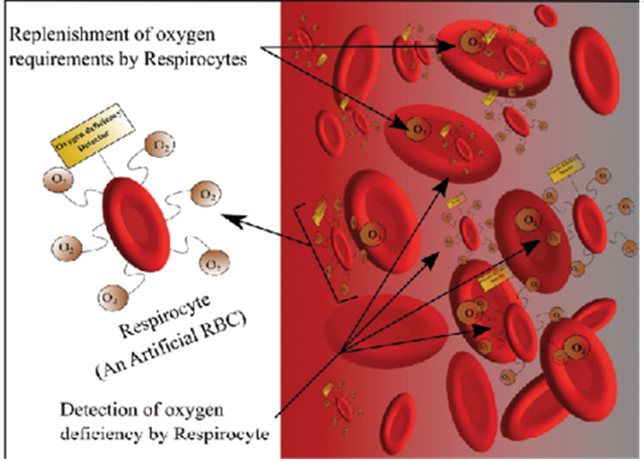
Respirocytes are microscopic mechanical devices known as nanomachines, which function at the molecular level. The diameter of each respirocyte ranges from 0.2 to 2 microns. Respirocytes have three chambers: an oxygen vessel that holds oxygen gas, a carbon dioxide vessel that stores carbon dioxide, and a water ballet that aids in buoyancy. Respirocytes serve as artificial red blood cells, transporting oxygen and carbon dioxide molecules through the blood cells. For the purpose of controlling the intake and release of carbon dioxide and oxygen, there are dedicated rotors. Additionally, a rotor permits glucose to enter the apparatus, where it combines with oxygen from the internal storage tank to provide energy needed for respirocyte function.This particular kind of nanorobot functions as an oxygen-carrying artificial red blood cell. The endogenous serum glucose provides the power. Compared to red blood cells (RBCs), these artificial cells can supply tissues with 2366 times more oxygen per unit volume and direct acidity. Figure 4 shows a respirocyte15,20.
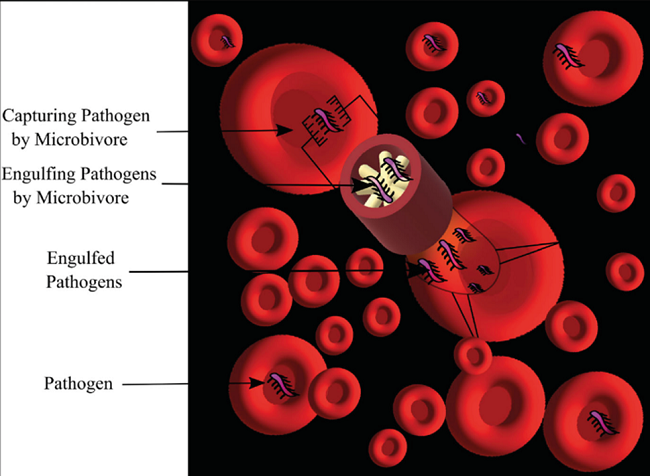
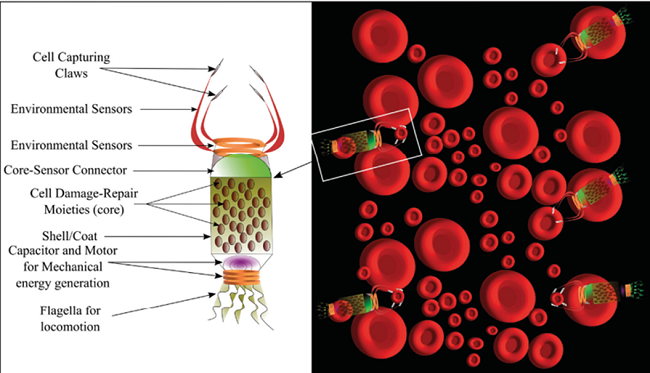
Microbivores:
With a diameter of 2.0 µm on the minor axis and 3.4 µm along the main axis, this flat, spheroidal device is intended for use in nanomedicine. The Figure 5 nanorobot can be used continuously at 200 pW; it is used to break downmicroorganisms that have become trapped. Its phagocytic ability, which is over 80 times more proficient than that of other macrophages in terms of (volume/sec digested) per unit volume of a phagocytic agent, is another distinctive feature15,21.
Clottocytes:
Clottocytes, also known as artificial mechanical platelets, possess a unique biological property that allows for “instant” haemostasis. Additionally, platelets are roughly two micrometres in size, coarsely spherical blood cells without a nucleus, that adhere to a bleeding site and aid in clotting blood vessels to halt bleeding. They also carry materials that help the coagulation process along15.
Chromallocyte:
In order to correct genetic abnormalities and other long-term impairments to the genetic composition, chromallocytes would replace entire chromosomes in individual cells, hence eliminating aging. A repair machine for a cell will first evaluate its state by examining its constituent materials, functions, and interactions on a molecular and structural level. These machines are able to completely revamp a cell. Figure 6 depicts a diamondoid-shaped nanorobot (cell-repair)15.
Design of Nanobots
Design strategy is crucial in micro- and nanorobotics research, with many types created by shrinking macroscale mechanical structures to micro- or nanoscale scales. Bionics, biosynthetic, and mechanical structure modelling are used to create micro/nanomachines, which can be used as prototypes for nanoscale motors. The study of microscopic or nanoscopic signal transmission in biomedicine is growing, with mechanical devices based on molecules or nanocomponents helping. Biomimetics, or imitation of biology, is growing in popularity in the design of micro and nanorobots. Biohybrid or bio syncretic robots, combining living and non-living systems, have gained attention for their practical uses. Two types of microrobots have been proposed, with sperms incorporating microtubules for mobility and helical structures for motility and immotility22-26.
Composition of Nanobots14:
Biochip:
In order to enable the necessary manufacturing technology for nanorobots for common medical applications, such as surgical instrumentation, diagnosis, and drug delivery, it is best to combine the use of nanoelectronics, photography, and new biomaterials. Therefore, practical nanorobots should be integrated as nanoelectronics devices, which will allow teleoperation and advanced capabilities for medical instrumentation.
Bacteria based:
This method suggests using biological microorganisms, such as bacteria called Escherichia coli. Therefore, the model’s means of propulsion is a flagellum. Despite its restricted uses, electromagnetic fields are typically used to control the mobility of this type of biologically integrated equipment.
Nubots:
Nubot is an abbreviation for “nucleic acid robot”. They are organic molecular machines at the nanoscale. Biological circuit gates are based on DNA materials, which have been engineered as molecular machines to allow in-vitro drug delivery for the targeted region.
Positional nano assembly:
Positional nano assembly is a process whereby nano-elements are guided into ordered patterns by means of external forces. It may entail picking up, moving, and positioning molecules one at a time using computer-controlled robot arms the size of molecules.
Established in 2000 by Robert Freitas and Ralph Werkle, the Nano Factory Collaboration is a targeted, continuous endeavor aimed at creating positionally-controlled diamond mechanosynthesis and a diamondoid nano factory, which would enable the construction of diamondoid medical nanorobots.
Method
A non-systematic assessment of scientific literature on the development of Nanobots was conducted, emphasizing the role of nanotechnology and the significance of its working as a drug delivery carrier. In order to achieve this, a bibliographic search was conducted mostly in the last ten years using several databases, including ScienceDirect and PubMed, though in certain instances, earlier references had to be consulted. Combining keywords such as Nanobots and Nano drug delivery or treatment of various diseases with nanobots systems (nanoparticle, nanotechnology, nanocarrier) led to the beginning of the search. Among all the papers discovered, those relevant to the review’s subject were chosen as an inclusion or exclusion criterion.
The next step involved focusing on targeting strategies of Nanobots, navigation of nanobots, mechanism and biocompatibility. At last the article ended with the conclusion of application and future perspective of nanobots.
Result and discussion
Targeting strategies of nanobots:
Passive targeted drug delivery:
The primary mechanism of action for passively targeted drug delivery systems (PTDDSs) in cancer therapy is the enhanced photochemical reaction (EPR) effect, which is primarily dependent on the distinct pathophysiological features of tumors, the properties of nanomaterials, and the parameters of blood circulation (for example, circulation time, phagocytosis, etc.). To achieve the therapeutic effect, researchers create a variety of passive drug carriers based on the physiological and nanomaterial parameters that influence the EPR effect27. Tumours typically exhibit the four primary pathophysiological traits listed below: 1) Profound angiogenesis; 2) impaired lymphatic drainage/recovery system; 3) markedly elevated permeability mediator production; 4) aberrant vasculature with poorly aligned endothelial cells, impaired angiotensin II receptor function, impaired lymphatic system, and absence of smooth muscle layer. Scientists leverage these distinct characteristics of tumours to enhance the effectiveness of targeted drug delivery systems (TDDSs). In addition, surface charge, surface wettability, size, and shape are important factors in drug distribution28.
Active targeted drug delivery:
The basis of active targeted drug delivery systems (ATDDs) is the interaction of cognate receptors with attached high-affinity ligands, which have the ability to bind to target cell receptors in a specific way. ATDDSs have employed a wide range of ligands, including the previously listed macromolecules (like peptides) and small molecules (like Ferulic Acid). Therefore, they may also be thought of as targeting nanocarriers mediated by ligands. ATDDSs can increase the bioavailability of chemotherapy drugs while lowering off-target effects. More significantly, when exposed to endogenous stimuli (such as pH, hypoxia, etc.) and exogenous stimuli (such as ultrasound, light, heat, and magnetic field), some advanced active drug carriers can achieve environment-responsive drug delivery or controllable drug delivery and deliver nanocarriers to the target sites29. The benefits listed above demonstrate that ATDDSs have garnered a lot of interest and have been the subject of in-depth research. Nanomaterial-based TDDSs are a newly developed cancer treatment technique. Because of their focused alterations, high surface-to-volume ratios, attractive drug release profiles, and nanoscale sizes, nanocarriers have the potential to deliver medications more steadily and controllably into target tumour tissues. Nonetheless, there are still certain issues and difficulties with the conventional targeted medication delivery methods. First off, the shapes, sizes, surface characteristics, and charges of biological fluids (like blood serum) and components (like proteins) that are present in the bioenvironment may interact with nanocarriers. This could have a negative impact on the effectiveness of nanomedicine. The second difficulty is accurately assessing the toxicity of nanocarriers due to numerous factors that impede characterization, including material, size, shape, porosity, surface area, surface charge, and hydrophobicity. Real-time detection of their position and dissemination presents the third challenge.
Further impeding their clinical translation are biostability, clearance rate of nanocarriers, and body tolerance. More crucially, due of their passive diffusion and short-range recognition (<0.5 nm) targeting strategies, the generated PTDDSs and ATDDSs only exhibit a low tumour targeting efficiency (0.7 %, median) and lack the ability to propel themselves28,29.
Navigation and propulsion of nanobots:
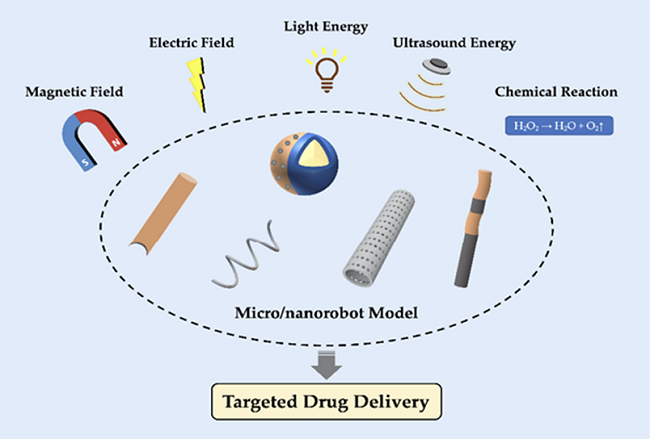
Exogenous power driven Nanobots:
The drug delivery robot system must overcome Brownian motion in order to achieve autonomous mobility in complicated bodily fluids because it is only micro- or nanoscale in size. In order to propel the particles, an external power source is typically utilized to regulate and synchronize the actions of the micro/nanorobot (as shown in Figure 7). In drug carriers, external powers such as heat, light, acoustic waves, magnetic fields, and electric fields are commonly employed. Multiple driving modes are frequently combined during the actual design phase to create micro/nanorobots that can perform a variety of tasks30.
Optical navigation/propulsion:
Optical tracking is the initial and most widely applied technique. It needs exact system calibration to use optical tracking for nanorobot localization. Optical tracking systems have several benefits, including excellent spatial resolution, low cost, low equipment complexity, and real-time observation. Nonetheless, it is difficult to localize microrobots in three dimensions using optical tracking. Noise or other contaminants in the workspace can easily distort the image. This indicates that transparent surroundings are the only ones in which optical tracking devices can be employed. These systems have shown encouraging results in the localization of nanorobots31,32.
Magnetic field propulsion:
The medical imaging method known as magnetic resonance imaging (MRI) was created in the 1980s. MRI is a non-invasive method of obtaining images from biological tissue. It may produce images with great spatial resolution that are incredibly delicate and precise. When combined with electromagnetic waves of radiofrequency (RF) and magnetic field gradients, a massive magnet produces a very strong magnetic field. Because MR imaging makes it easy to distinguish between healthy and malignant tissue, it is frequently employed in neurosurgery. Functional magnetic resonance imaging, or fMRI, has shown promise in studying brain function over the past ten years. Recently, MRI has been utilized to track and locate nanorobots in vivo31,33.
X-Ray navigation:
An established imaging method is X-ray. Numerous methods, including as radiography, fluoroscopy, and CT scans, are based on X-ray imaging. The most effective method for localizing nanorobots among these approaches appears to be fluoroscopy. In research involving animals, it has been utilized for visual serving and needle orientation control. X-ray fluoroscopy has several benefits, including high sensitivity, real-time imaging, excellent material density discrimination, and deep penetration of the human body. Radiation that causes cancer is the primary drawback of X-rays31,34.
Ultrasound energy propulsion/navigation:
The basis of ultrasonic imaging is how sound waves interact with human tissue. Ultrasound imaging is frequently utilized in medical contexts due to its non-invasive nature. It is inexpensive, offers real-time imaging, and has little negative health impacts. Nanorobot manipulation is possible via ultrasound imaging. Ultrasound imaging has several difficulties and drawbacks, such as its inability to move through the air and its propensity to disperse easily from bones. Ultrasound imaging technologies have a very small scanning area and can only go a few centimeters into the human body. Ultrasound pictures have poor resolution and are hardly useful for controlling or visually serving nanorobots. Some researchers employed this technique for localization31,35.
Light energy propulsion:
For in vitro research, this technology is recommended due to its speed and ability to give real-time imaging. While it offers very high sensitivity and resolution in 2D, 3D nanorobot localization can be difficult. This approach typically necessitates additional surface modification of nanorobots and has a restricted depth of penetration into the human anatomy. Nanorobots have been tracking in vivo using this technique for a long time through adjusting the propagation direction, intensity, polarization, and frequency of light31.
Electric field propulsion:
Magnetic and electric energy can be combined and separated depending on certain circumstances. An external high-frequency electric field is used by a Janus colloidal system to enable autonomous mobility and cargo pickup in a common electric field-driven micro/nanorobot. Under an electric field, a rotating nanomotor device made of carbon nanotubes has demonstrated extremely high-speed movement and quick response. When an electric field and light energy are combined, nanoparticles can move in all directions. Using a combination of direct current and alternating current electric fields, a Pt-based bimetallic nanorod motor was able to achieve movement in various directions and at varying speeds. Electric field regulation enables precise pickup and release of Au nano porous templates. These investigations establish a strong basis for drug administration in vivo by confirming the superior capabilities of micro/nanorobots for extracting, delivering, and releasing cargos in vitro. In practical applications, electric field intensity is increased since electric energy penetration is not as great as magnetic field penetration30,36.
Endogenous power driven Nanobots:
The primary source of the endogenous power that drives nanorobots to propel themselves forward is a chemical or biological response. Usually asymmetric, these micro/nanorobots are covered in catalysts to continuously harvest chemical energy from their surroundings. Redox reaction-based nanorobot driving force conversion from chemical energy is a widely used concept. In particular, the most often employed technique was a hydrogen peroxide breakdown process. Because of its weak chemical link, hydrogen peroxide is quickly broken down into oxygen and water by a variety of catalysts, including metals, enzymes, and an alkaline environment. Additionally, it was extensively used in nanorobots as vesicular polymers, Janus particles, bimetallic nanorods, and other forms30. A comparison profile of exogenous and endogenous power of nanobots is given in Table 1.
Table 1. Comparison of exogenous and endogenous power driven Nanobots30.
| Type | Energy | Penetration | Move Ability | Persistence | Safety |
|---|---|---|---|---|---|
| Exogenous Power | Magnetic fields | Good, can work under a relative weak magnetic field. | Precise 3D-navigation in fluids under rotating magnetic fields. | Good, nanorobots can keep moving with the guidance of external forces. | The magnetic field used is within safe range; metal materials will bring potential harm to human body. |
| Electric energy | Relatively weak, need to increase the electric field intensity. | Directio-nal move-ment under the combination of electric energy and other energy. | Strong electric field intensity may affect human body; metal materials will bring potential harm to human body. | ||
| Light energy | The transmittance of different light (visible light, UV, NIR, etc.) is different. | Usually function as a trigger for other reactions, can achieve directional move-ment. | Ultra-violet light is harmful, other lights are basically safe. | ||
| Ultrasound energy | Good, with a strong penetration ability. | Usually combined with magnetic field, can achieve directional movement. | Ultra-sound might cause oxidative stress in cells (affect normal cells); metal materials will bring potential harm to human body. | ||
| Endogenous power | Chemical energy | Not applicable | With the movement ability, but still need to be positioned by external forces (such as magnetic attraction). | Not so good; chemical energy can run out and the micro/nanorobots’ ability to move can also be compromised when this happens gradually. | The safety of fuel needs to be considered, H2O2 is noxious, glucose and urea are nontoxic fuels. |
Mechanism of nanobots
Proximity sensors on nanorobots detect obstructions in their path, both moving and stationary. Nanorobots follow a predefined route, navigating by determining the shortest path between barriers every 0.5 mm. Nanorobots wait 0.0025 milliseconds to choose a different course to navigate if impediments are in their way. Once at the desired location, nanorobots use both internal and external resources to deliver medications. The human excretory system will be used to eliminate them from the body after the goal has been reached. They may also be eliminated in specific circumstances by active scavenger systems, sometimes referred to as nano terminators13.
These installed nanobots can be eleminated from the body by some of the techniques given below:
1.Natural excretion: Numerous nanobots are made to naturally exit the body through the excretory systems, including feces or urine. The ability of the nanobots to enter the digestive system or kidneys must frequently be maintained for this procedure to work efficiently13.
2.Biodegradation: Certain nanobots are constructed from materials that the body’s biological processes can degrade. These substances are broken down into innocuous metabolites by enzymes and other biochemical processes13.
3.Targeted removal: More sophisticated nanobots may have capabilities that enable them to be targeted and extracted from the body. External triggers or systems that cause the nanobots to self-destruct or gather by specialized equipment could be involved in this technique13.
4.Medical intervention: Medical methods may be required in some circumstances to remove nanobots, particularly if they are not made to break down or expel themselves naturally. Endoscopic or surgical methods may be used for this technique.13.
Biocompatibility of Nanobots
Researchers are still unsure of the exact interaction between nanorobots and living systems due to the complexity of interactions between biological matter and materials used to build nanorobots. This has led to a lack of adequate identification of potential risks and hazards. Experiments have shown that particles made of the same material may behave differently due to surface coating variations. Other factors to consider include material, size, form, surface coating, actuation and sensing mechanisms, and the working environment. Efforts have been made to identify potential targets in the human body, such as the lung, liver, heart, and brain, and key entrance channels31.
Challenges of Nanobots
Nanobots, a form of robot with multipurpose functions, have been beneficial in nanotechnology, robotics, biomedicine, and electromechanical science. They can move rapidly in biological environments, haul cargo, and perform in vivo functions. However, they face challenges like changing physical laws and combining multiple operations. Smart nanobots require signal detection, acquisition, processing, and transmission. Biomedical nanobots are needed for soft tissues, physiological changes, and unexpected biological events. However, nanoparticles’ ability to transcend biological boundaries raises ethical concerns, including access to knowledge, fundamental rights, and responsible use of nanomedicine3.
Application of Nanobots in medicine
Cancer detection and treatment:
If caught in its early stages, cancer is treatable. Due to the ability of its nano sensors to identify tumour-producing cells early on, nanorobots aid in the early diagnosis of cancer. Chemotherapy has side effects because the anticancer drug delivery systems that are now in use lack inherent navigation for tissue penetration, lengthy circulation times, targeting, and localized drug delivery. Because nanorobots are self-navigating, they may target specific cells, penetrate tissues, and provide the greatest amount of therapeutic benefit37.
Nanobots in surgery:
Surgeons are always searching for new approaches to treat patients, aiming to do minimally invasive techniques during surgery since patients recover more quickly from less trauma. There are often few post-operative problems because the procedures are minimally invasive. The early detection, diagnosis, prevention, or treatment of disease at the cellular or subcellular level will be the main goals of using nanorobots in surgery. Surgical nanorobots let surgeons execute procedures precisely at the nano, micro, and macro scales. Surgical nanorobots are inserted into the human body’s circulatory system via micro syringes and catheters. Surgical nanorobots function inside the human body as a semi-autonomous on-site surgeon and are programmed or directed by a human surgeon38.
Nanobots in Gene therapy:
Gene therapy has the possibility to heal or cure genetic problems by altering an individual’s genes. In order to treat an illness, gene therapy involves either inserting a new or altered gene into the body to replace the disease-causing gene with a healthy copy, inactivating the disease-causing gene that isn’t working correctly, or both. Gene delivery is facilitated by nanorobots, which administer gene therapy to specific target areas. Through blood circulation, nanorobots are able to reach target areas, infiltrate tumour cells, and turn off aberrant genes. Because of their small size and efficient interaction with biomolecules at the cell surface, nanorobots are able to transfer genetic materials to target cells with great ease39.
Nanobots in cell therapy:
Human cells can be renewed with the aid of regenerative medicine to return to normal function. Stem cells are used in regenerative medicine treatments to promote tissue growth. Magnetically directed nanorobots transport stem cells to specific body locations by acting as a carrier system. These uses show that nanorobots are important for targeted medication delivery and will help with cell therapy and regenerative medicine in the future40.
Nanobots in Dentistry:
Nanorobotic dentifrices are being used in dental preparations to break down harmful bacteria and provide analgesics. These nanorobots penetrate sublingual surfaces and use motility processes to inject a colloidal suspension into the patient’s gingival region. They can penetrate the pulp, regulate nerve perception, and aid in quick and painless tissue healing and tooth alignment. The analgesic produced by nanorobots is a quick-acting, reversible aesthetic with no adverse effects41.
Nanobots in wound healing:
Chronic wound healing is challenging due to poor healing mechanisms or insufficient coagulation factor levels. Nanorobots can help promote efficient wound healing by delivering growth factors and clotting factors to the wound site. These nanorobots, loaded with tissue plasminogen activator, can break up blood clots by accelerating thrombolysis by increasing interaction between the tissue plasminogen activator and blood clot interface42.
Future perspectives of Nanobots
More focused research studies are needed to enhance the usefulness and dependability of small-scale robot systems, which are still quite novel and in their infancy. It has been demonstrated that a number of intelligent medical micro and nanorobots hold promise for precision cancer treatment and drug delivery that targets tumours. In this work, we examined recent advancements in drug delivery using micro-and nanorobots as well as other medicinal applications. Although micro/nanorobots can be used in deep tissue for real-time control and monitoring as well as complex task performance, there are a number of obstacles to overcome. These include the inability to control nanorobots, difficulties in locating and removing them from the body, communication issues, ease of preparation, increased drug delivery efficiency, unwanted immune reactions, and biocompatibility and biodegradability. Coordinated efforts should be made to standardize the procedures and parameters that are used to describe the actions of the devices, such as the treatment of diseases, the delivery of medications and cells with precision, and the employment of diagnostic agents15. Recent studies of nanobots in drug delivery is given in Table 2.
Table 2. Recent studies of Nanobots in pharmaceutical drug delivery.
| Animal models | Nanobots used | Loaded drug | Targeted region | Key findings | References |
|---|---|---|---|---|---|
| Mice | Gold Nanobots | Doxorubicin | Tumour | Enhanced drug delivery Tumour shrinkage. | (43) |
| Rats | Magnetic Nanobots | Paclitaxel | Blood Stream | Improved efficacy and drug circulation. | (44) |
| Rabbits | Polymer Nanobots | Methotrexate | Joints | Reduced inflation & Targeted therapy. | (45) |
| Guinea pigs | Lipid Nanobots | Insulin | Gastrointestinal | Controlled insulin release & Controlled efficacy. | (46) |
| Monkey | DNA Nanobots | Anti - Viral drug. | Brain | Enhanced brain drug delivery. | (47) |
Conclusion
Micro/nanorobots are a revolutionary advancement in pharmaceutical drug delivery, offering personalized medicine and reducing side effects. These nanoscale machines can optimize treatment regimens, reduce side effects, and enhance therapeutic outcomes. They can cross biological barriers, enabling treatments for conditions previously considered inaccessible. They enable multimodal therapies, delivering multiple drugs or agents simultaneously, potentially enhancing treatment efficacy and reducing drug resistance. Encouraging investment in nanorobotics research, fostering collaborations, and establishing safety and efficacy standards through regulatory bodies are crucial steps towards a healthier future in medicine.