BACKGROUND
In recent years, antibiotics have been used to treat disease- causing bacteria. Antimicrobial resistance is a global threat to public health [1, 2, 3].
Enzymatic modification (b-lactamases), reduced permeability (selective blockage), increased membrane transport (efflux pumps), altered binding site (specific receptors) and metabolic bypass are the mechanisms of antibiotic resistance in bacteria [4, 5, 6].
The detection of multidrug resistance is, however, a primary step in effectively controlling antibiotic- resistant bacterial infections that can lead to clinical failure and additional antibiotic resistance [7, 8, 9].
The gram- negative organism of Klebsiella pneumoniae .K. pneumoniae) is one of the most important causes of nosocomial infections in developing countries and one of the eight most important causes in developed countries [10, 11, 12, 13]. Nosocomial infections caused by K. pneumoniae multi- antibiotic resistant have become an increasing public health concern. K. pneumoniae produces various pathogenic virulence factors, including capsular polysaccharide, lipopolysaccharide, fimbriae, siderophores and resistance [14, 15, 16].
Increased global infections by multidrug- resistant bacteria closely associated with limited drug treatments imply the failure of empirical treatments and strengthen the need for antibacterial therapies based on antimicrobial sensitivity tests [1, 17, 18]
Several studies have demonstrated outbreaks in hospitals due to K. pneumoniae resistant to cephalosporins, aminoglycosides and quinolones of third generation [10].
Therefore, this study aimed to investigate the antimicrobial susceptibility profile of Klebsiella pneumoniae isolates collected during the period of January 2016 till December 2017 at Ziaee Hospital in Ardakan.
MATERIALS AND METHODS
From January 2016 to December 2017 all positive cultures of Klebsiella pneumoniae in patients referred to Ziaee Hospital in Ardakan during 2016 and 2017, were evaluated. This study was approved by ethical committee of Yazd University of medical sciences with ID: IR.SSU.MEDICINE.REC.1395.278. It was result of thesis of general physician.
In this cross-sectional study, the samples were collected as census from all positive cultures of K. pneumoniae by a checklist that was previously provided by the researcher and confirmed by qualified specialists.
Samples were obtained in a sequential census of all positive cultures in terms of K. pneumoniae at the designated time period.
Data collection was carried out based on a questionnaire that was previously designed in terms of gender, age, the type of antibiotic (including: imipenem, meropenem, cefepim, ciprofloxacin, amikacin, ceftazidime, ceftriaxon, cefotaxime, ampicilin, cotrimoxazol, cefalotin and clarithromycin) and finally the type of sample (including: urine, blood, ulcer, sputum, pleural fluid, urethral discharge, eye secretions and fecal). Inclusion criteria were including all positive cultures for Klebsiella pneumoniae. The cases in which sample data was incomplete were excluded from study and were as exclusion criteria.
In this study, the disc diffusion sensitivity test was used to evaluate the sensitivity and resistance of K. pneumoniae to antibiotics. In the disc diffusion method, a certain amount of bacteria is set according to the existing standards in terms of the degree of dilution and has already been identified. Special culture media add to the same plates in terms of diameter, depth, etc., and discs the standardized antibiotic filter paper is placed on the plate surface. After the time it takes to grow the microbes, if the antibiotic is able to prevent the growth of the microorganism, it does not grow around the bacterial disk, and the bacteria to the antibody the biotype is more sensitive, these will be no larger growth halo.
Detection and differentiation of sensitive, semi-sensitive and resistant conditions from each other was performed based on the diameter of the around the colony as millimeters and according to the relevant table in laboratory. The data were collected, recorded in SPSS software (version 21) and analyzed by chi-Square test.
RESULTS AND DISCUSSIONS
Of 75 samples, 22 (29.3%) were related to men and 53 (70.7%) were for women. Of these, 60 patients (80%) were admitted and 15 patients (20%) were outpatient.
The samples were including urine specimens that were highest numbers with 64 (85.3%) and then, sputum 4 (5.3%), wound and blood each one 3 (4%) and finally secretions 1 (1.3%).
Klebsiella resistance was 100% for clarithromycin but about amikacin was susceptible in 100% of cases (Table 1).
Table 1. The frequency distribution of Klebsiella pneumoniae susceptibility based on the type of antibiotic
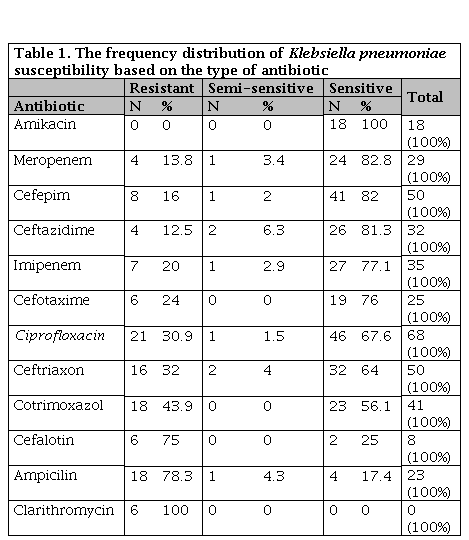
N: Number of sample; * Significance susceptibility based on gender.
It should be noted that for all specimens, all antibiotic disks have not been used. For each sample, according to the type of specimen and physician's opinion, a certain number of disks were inserted. For example, clarithromycin was used in only 6 cases.
In women, the highest sensitivity was observed for amikacin (100%) and meropenem (88.2%). In the case of ciprofloxacin, there was a significant difference between the sexes (p=0.01). In men, resistance to cefalotin, ampicillin and clarithromycin antibiotics was 100%, but in women only resistance to clarithromycin was 100%. Overall, the resistance to all antibiotics examined in men was higher than women (Table 2).
Table 2. The frequency distribution of Klebsiella pneumoniae susceptibility based on gender
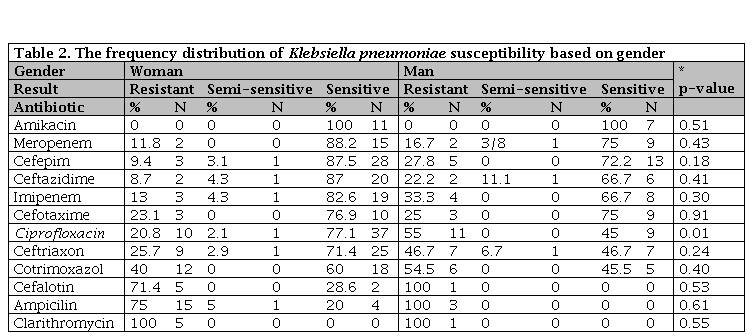
N: Number of sample; * Significance susceptibility based on gender.
In total, the resistance to all antibiotics examined was in the hospitalized patients more than outpatient cases, but this difference was significant only for meropenem (p=0.001) and was probably due to a small number of outpatient cases (Table 3).
Table 3. The frequency distribution of Klebsiella pneumoniae susceptibility based on type of admission
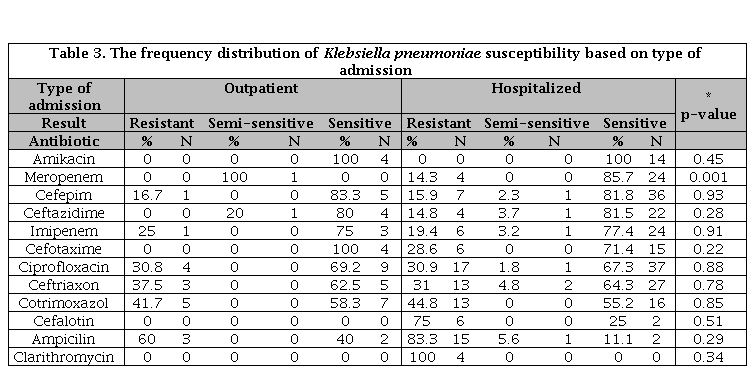
N: Number of sample; * Significance susceptibility based on gender.
Body secretions (Other than ulcers, blood, sputum, urine) were resistant to chlorthromycin and ampicillin (100%), and 100% sensitive to cotrimoxazole, cefotaxime and ceftriaxone. The frequency distribution of Klebsiella pneumoniae sensitivity based on of sample type has been listed in Table 4.
Table 4. The frequency distribution of Klebsiella pneumoniae susceptibility based on type of admission
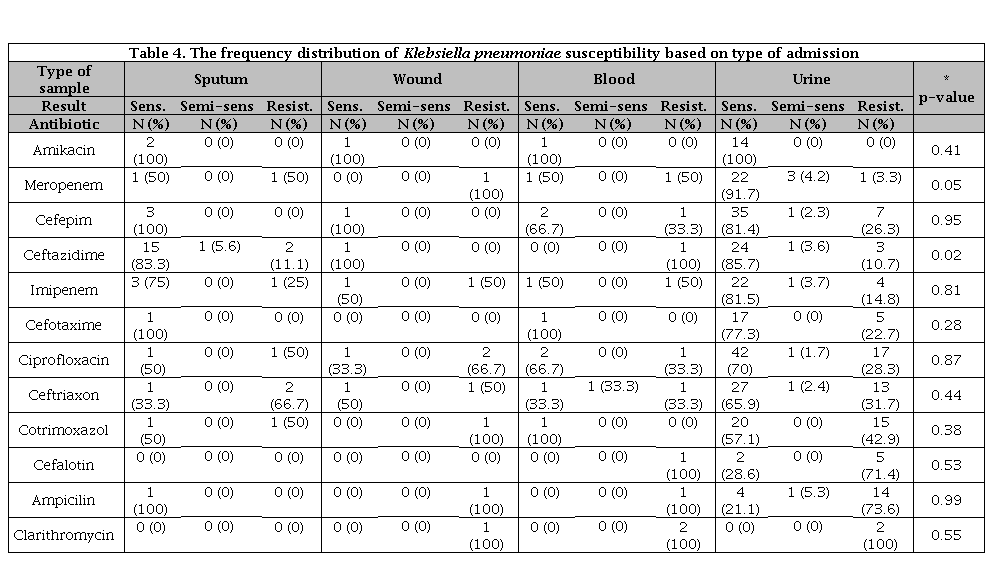
N: Number of sample; * Significance susceptibility based on gender.
A study assessed the 1805 Klebsiella pneumoniae isolates from the Hospital Universitário de Santa Maria, Brazil for antimicrobial susceptibility profile. Resistance to colistin (239.3%), ciprofloxacin (64%), and amikacin (21.4%) was found increased [1]. In the present study, the sensitivity rate was the highest against amikacin. Also the sensitivity rete to ciprofloxacin (67.6%) was more than mentioned study.
Another study showed that among the 168 K. pneumonia isolates studied, 72.0% were resistant to ceftazidime, almost 69% to cefotaxime and 67.2% to amikacin [10]. In our study the sensitivity was 100% against amikacin and there was no any resistance to it.
In a study that was performed on Klebsiella pneumoniae isolates from the hospitals in sari, Mazandaran, the highest resistance against cefotaxime (100%) and ceftazidim (100%) was observed among all isolates, while the highest susceptibility to gentamycin was observed (63%) [19]. But at present study, the highest sensitivity and resistance of Klebsiella pneumonia for antibiotics was related to amikacin and clarithromycin, respectively.
An investigation was conducted to investigate the prevalence of K. pneumoniae isolates and their antibiotic susceptibility pattern. The highest number of K. pneumoniae were resistant to ampicillin (75.6%) followed by, nitrofurontoin and cefuroxime almost 74% and least to chloramphenicol (13%) [20]. In the present study, the highest sensitivity was related to amikacin.
SUMMARY
The results of our study showed that the highest sensitivity of Klebsiella pneumoniae for antibiotics was to amikacin (100%). Also, Klebsiella's resistance to clarithromycin was 100%. The resistance to all antibiotics examined was in the hospitalized patients more than outpatient cases, but this difference was significant only for meropenem. Also, the resistance to all antibiotics examined in men was higher than women.
CONCLUSION
Currently, K. pneumoniae has reached high levels of endemicity in hospitals and is one of the main causes of nosocomial outbreaks. The results on our study show that Klebsiella's resistance to clarithromycin was 100%, but was 100% susceptible to amikacin. It needs to investigate in the more cities in our province for preventing resistant infections by organism.














