Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Nefrología (Madrid)
versión On-line ISSN 1989-2284versión impresa ISSN 0211-6995
Nefrología (Madr.) vol.31 no.2 Cantabria 2011
Relationship between leptin and all–cause and cardiovascular mortality in chronic hemodialysis patients
Relación entre leptina y mortalidad global y cardiovascular en pacientes en hemodiálisis
J. J. Díez1, M. Bossola2, M. J. Fernández–Reyes3, E. Di Stasio2, L. Tazza2, G. Luciani2, R. Codoceo4, P. Iglesias1, A. Rodríguez3, E. González5, R. Selgas5
1Servicio de Endocrinología. Hospital Ramón y Cajal. Madrid.
2Servizio Emodialisi. Università Cattolica del Sacro Cuore. Roma (Italia)
3Servicio de Nefrología. Hospital General de Segovia.
4Servicio de Bioquímica. Hospital La Paz. Madrid.
5Servicio de Nefrología. Hospital La Paz. Madrid.
This study has been partially supported by grants by Baxter /Extramural Grant Program, 2008), ISCIII by support to RS (PS 09/00641) and Rio Hortega Grant (2009) for EG. Several authors are integrated in REDinREN (RETICS 06/0016 from ISCIII), supported by FEDER Euroean Funds.
ABSTRACT
Background: We aimed to evaluate the relationship between serum leptin and the leptin/body mass index (BMI) ratio with prevalent cardiovascular disease (CVD), and their influence on all–cause and CVD–related mortality in patients on hemodialysis (HD).
Methods: 118 stable HD patients (50 women, median [interquartile range] age, 65.1 [54.7–72.2] years) were studied. All patients had baseline measurement of serum leptin concentrations. Relationships between leptin and all–cause and CVD mortality were studied by means of survival analysis and Cox regression analysis.
Results: The leptin/BMI ratio was similar in patients with and without CVD at baseline (0.65 [0.29–2.23] vs. 0.68 [0.29–1.49] ng·m2/ml·kg, respectively, NS). Multiple logistic regression analysis showed that there was not an independent association between leptin/BMI ratio and prevalent CVD. During the follow–up time, 52 (44.1%) patients died. CVD was the cause of death in 27 out of 52 (51.9%) deceased patients. Survival analysis and Cox proportional multivariate regression analysis showed that there were no significant relationships between leptin levels or the leptin/BMI ratio and all–cause and CVD–related mortality.
Conclusion: These results do not support that, in stable HD patients, serum leptin concentrations and the leptin/BMI ratio are related with prevalent CVD. Leptin/BMI ratio seems not to be a risk factor for mortality in these patients.
Key words: Leptin, End–stage renal disease, Hemodialysis, Mortality, Cardiovascular disease.
RESUMEN
Introducción: El objetivo del presente estudio ha sido evaluar la relación entre la leptina sérica y el cociente leptina/índice de masa corporal (IMC) con la enfermedad cardiovascular (ECV) prevalente y su influencia en la mortalidad global y en la mortalidad por ECV en pacientes en hemodiálisis (HD).
Métodos: Se estudiaron 118 pacientes estables en HD (50 mujeres, edad mediana [recorrido intercuartílico], 65,1 [54,7–72,2] años). En todos los pacientes se cuantificó la concentración basal de leptina. La relación entre leptina y la mortalidad se evaluó mediante análisis de supervivencia y análisis de regresión de Cox.
Resultados: El cociente leptina/IMC fue similar en pacientes con y sin ECV prevalente (0,65 [0,29–2,23] frente a 0,68 [0,29–1,49] ng·m2/ml·kg, respectivamente, NS). El análisis de regresión logística mostró que no existía una asociación independiente entre el cociente leptina/IMC y la enfermedad cardiovascular prevalente. Durante el seguimiento 52 pacientes fallecieron (44,1%). La ECV fue causa de muerte en 27 de 52 pacientes fallecidos (51,9%). El análisis de supervivencia y el análisis multivariante de Cox mostraron que no hubo relación significativa entre los niveles de leptina o el cociente leptina/IMC y la mortalidad global o por causa de ECV.
Conclusión: Estos resultados no apoyan la hipótesis de que, en pacientes estables en HD, las concentraciones de leptina y el cociente leptina/IMC estén relacionados con la ECV prevalente. Más aún, el cociente leptina/IMC no parece ser un factor de riesgo de mortalidad en estos pacientes.
Palabras clave: Leptina, Enfermedad renal terminal, Hemodiálisis, Mortalidad, Enfermedad cardiovascular.
Introduction
Cardiovascular disease (CVD) is the major cause of death in chronic hemodialysis (HD) patients1. According to two of the largest end–stage renal disease (ESRD) registries, the US Renal Data System (USRDS) and the European Registry of patients on renal replacement therapy (ERA–EDTA), the estimated risk for cardiac events such as myocardial infarction is 3.5–50 times higher among HD patients than in the general population2,3. In the last two decades, it has become evident that the increased frequency of CVD observed in HD patients is secondary to the combination of many traditional and novel and uremia–related risk factors4.
Leptin promotes atherosclerosis, thrombosis, and vascular smooth muscle cell proliferation and migration5. In addition, clinical studies have demonstrated a significant correlation between leptin levels and hypertension, hyperlipidemia, perturbed fibrinolysis and chronic inflammation6,7. Leptin levels are significantly higher in HD patients than in healthy subjects8,9. Thus, it has been suggested that such high leptin levels may contribute to the increased cardiovascular risk of HD patients. The present study aimed at evaluating the relationship between serum leptin concentrations and the leptin/body mass index (BMI) ratio with prevalent CVD and their influence on all–cause and CVD–related mortality in ESRD patients undergoing maintenance HD.
Methods
Patients and design
This is a prospective observational study in which we measured leptin levels and the leptin/BMI ratio and correlated with prevalent CVD and all–cause and CVD–related mortality. The study was performed in the Hemodialysis Units from the Università Cattolica del Sacro Cuore of Rome (Italy) and from the Hospital General of Segovia (Spain). Italian patients (n = 53) were recruited in March 2004 (cohort 1). Spanish patients were recruited in two phases. A group of 38 patients were studied between 1998 and 2002 (cohort 2), and a second group of 27 patients were evaluated in April 2008 (cohort 3). Incident patients considered eligible and included in the study were evaluated after at least 6 months of hemodialytic treatment. Patients were followed until census date (December 31st, 2009), renal transplantation or death. Median time of follow–up was 24.7 (interquartile range 15.7–68.0) months. As expected, time of follow–up in cohort 3 (20.1 [20.1–20.4] months) was significantly (P = 0.002) lower than those found in cohorts 1 (46.5 [15.0–76.3] months) and 2 (40.2 [12.3–73.7] months). The study was approved by the local ethics committees and written informed consent was obtained from all patients before enrollment in the study.
Hemodialysis
All patients were receiving conventional 4–hour hemodialysis, three times a week. The blood flow ranged from 250 to 400 ml/min with a dialysis rate flow of 500 ml/min. All patients were treated with high–permeability membranes. Most patients were takingrecombinant human erythropoietin and antihypertensive medications.
Cardiovascular disease definition
Patients were stratified according to the presence of cardiovascular diseases. Patients defined as having prevalent CVD included those with a documented history of angina pectoris, myocardial infarction, stroke, coronary revascularization procedures, transient cerebral ischemia, peripheral artery surgery, and peripheral vascular disease.
Laboratory analyses
Serum leptin concentrations were measured by using a polyclonal antibody RIA raised in rabbits against highly purified recombinant human leptin (Linco Research, St Louis, MO, USA). The sensitivity for this leptin assay was 0.5 ng/ml, and the coefficients of variation intra– and interassay were 4.8% and 3.5%, respectively. The normal values of serum leptin concentrations in a group of healthy subjects, aged 28–70 years, was 10.0 (5.6–27.8) ng/ml. Other laboratory measurements were performed through certified methods in the Departments of Clinical Chemistry of the Università Cattolica del Sacro Cuore of Rome (Italy) and the Hospital General of Segovia (Spain).
Statistical analysis
For mean comparisons the Kruskal–Wallis non parametric test was used for non–normally distributed continues variables and analysis of variance was used for normally distributed variables. Correlations were calculated with the Spearman correlation coefficient. Survival time was estimated by the Kaplan–Meier method, with the log–rank test used to compare arms. Multivariate Cox regression models were used to assess the independent effects of several quantitative and qualitative variables on the risk of death. Statistical significance was set at the level of P <0.05.
Results
A total of 118 patients (50 women) were included in this study (table 1). Italian patients showed higher levels of leptin/BMI in comparison with Spanish patients.
Table 1. Demographic, clinical and laboratory characteristic of the three cohorts of studied patients 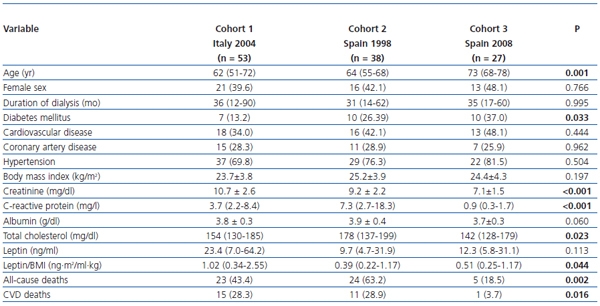
Data are presented as number of patients (percentage), median (interquartile range) or mean ± SD.
Leptin and leptin/BMI ratio
The demographic, clinical and laboratory characteristics of patients stratified according to the leptin/BMI ratio are reported in table 2. In univariate analysis, the leptin/BMI ratio was positively correlated to total cholesterol (rho = 0.298, P = 0.001) and creatinine (rho = 0.183, P = 0.047). The multiple regression analysis confirmed that creatinine and total cholesterol were independent factors associated with the leptin/BMI ratio (R2 = 0.146, P = 0.015).
Table 2. Demographic, clinical and laboratory characteristic of the patients included in the study
stratified according to the leptin/BMI ratio 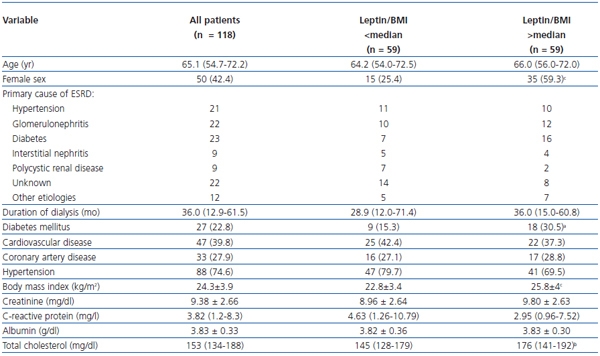
Data are presented as number of patients (percentage), median (interquartile range) or mean ± SD.
a P <0.05; b P <0.001; c P <0.001.
Leptin/BMI and prevalent cardiovascular disease
Forty seven (39.8%) and 33 (27.9%) patients had prevalent CVD and coronary disease, respectively, at baseline. The prevalence of CVD and of ischemic heart disease at baseline was similar between patients with leptin/BMI under the median value and above the median value. The results of the multivariate logistic regression analysis showed that there was not an independent association between leptin/BMI ratio and CVD (table 3). Moreover, this multivariate analysis did not show a significant association between cohort and prevalent CVD.
Table 3. Multivariate logistic regression analysis showing the influence of several qualitative and
quantitative variables on the presence of CVD at the time of inclusion in the study 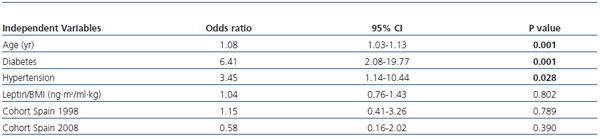
Data are odds ratios and 95% confidence intervals.
Leptin/BMI and survival
During the follow–up period, 52 (44.1%) patients died. CVD was the cause of death in 27 out of 52 (51.9%) deceased patients. The median serum leptin concentrations and the median leptin/BMI ratio was similar in patients who survived (18.0; 7.0–39.1 ng/ml, and 0.73; 0.29–1.50 ng·m2/ml·kg, respectively) and in those who died (12.0; 4.9–44.8 ng/ml, P = 0.524, and 0.49; 0.26–1.94 ng·m2/ml·kg; P = 0.529, respectively). No differences were found in serum leptin concentrations and leptin/BMI ratio between patients who died by CVD (9.6; 4.8–57.9 ng/ml, and 0.42; 0.25–1.99 ng·m2/ml·kg, respectively) and those who did not (16.1; 6.1–39.0 ng/ml, P = 0.863, and 0.68; 0.28–1.49 ng·m2/ml·kg, P=0.985, respectively). We repeated this analysis in the three cohorts of studied patients and found that there were no significant relationships between leptin or leptin/BMI and mortality (data not shown).
Kaplan–Meier analysis (figure 1) showed that patients with leptin levels above the median values showed mean survival times for all–cause mortality and for CVD–related mortality which did not significantly differ from those found in patients with leptin concentrations under the median value. In a similar way, patients with leptin/BMI above the median values showed a mean survival times for all–cause mortality and for CVD–related mortality similar to those found in patients with leptin/BMI under the median value.
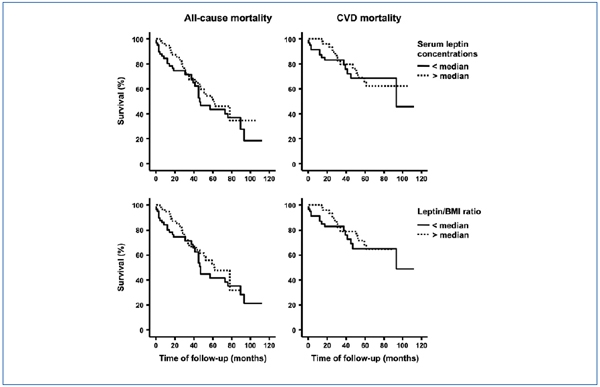
Figure 1. Kaplan–Meier survival analysis for all–cause mortality (left panels) and CVD–related mortality
(right panels) in 118 HD patients stratified according to the serum leptin concentrations (upper panels)
and the leptin/BMI ratio (lower panels).
We performed a Cox proportional hazards regression multivariate analysis including the covariate cohort because of the significant differences found among cohorts in leptin/BMI levels and mortality rates (table 1). This analysis showed that leptin/BMI was not an independent risk factor for all–cause and CVD–related mortality (table 4).
Table 4. Multivariate Cox regression analysis for all–cause and cardiovascular disease–related mortality 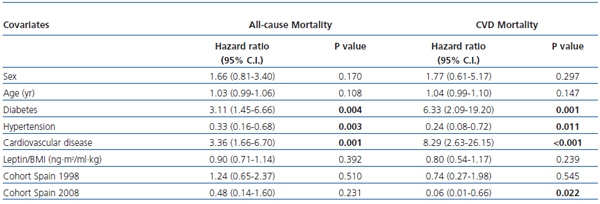
For categorical variables, reference hazard ratio (HR = 1) for female sex, absence of diabetes,
hypertension and cardiovascular disease, and cohort Italy 2004.
Discussion
Our results show that there is no significant relationship between serum leptin or the leptin/BMI ratio and the presence of CVD or coronary artery disease at baseline. Moreover, our follow–up data also show a lack of relationship between serum leptin or the leptin/BMI ratio and all–cause and cardiovascular mortality. Our cross–sectional data are in agreement with our previous study that showed that leptin/BMI was not related to prevalent CVD10. On the contrary, our longitudinal study results clearly differ from those reported by Scholze et al.11. Discrepancies may be accounted for by differences in the genetic background of studied patients, confounding influences of covariates, and different laboratory procedures. Furthermore, we used leptin values corrected by BMI because leptin is a sensor of body fat mass and a correlation between leptin levels and BMI has been demonstrated both in healthy subjects12 and in uremic patients10.
Leptin increases sympathetic activity13, promotes vascular smooth muscle cells proliferation, increases oxidative stress and has prothrombotic activity5. Furthermore, leptin has been related to coronary artery calcification in type 2 diabetic patients14 and with several CV risk factors and vascular dysfunction in humans15. The WOSCOP study16 have reported an association between leptin and the risk of CVD. However, a recent meta–analysis showed only a moderate association between leptin levels and the risk of coronary heart disease, which is largely dependent of BMI17. In agreement with this we could not demonstrate any significant association of leptin/BMI with CVD mortality.
A possible explanation to the lack of relationship between leptin and mortality in HD patients might be found in the reverse epidemiology of the cardiovascular risk factors which is frequently observed in patients with ESRD18. Numerous reports have suggested that an increase in BMI is correlated with an increased survival in HD patients19,20. In fact, recent European guidelines consider that BMI under 23 kg/m2 is suggestive of malnutrition in HD patients21. In this context, high leptin levels can be considered as a marker of overnutrition and, therefore, associated to a favourable prognosis. Another possible explanation is that the actions of leptin on the cardiovascular system are not always detrimental. For example, although elevated leptin levels have been associated with poor vascular compliance in adolescents15 and impaired coronary vasoreactivity in otherwise healthy young obese subjects22, some evidences suggest that leptin may have both vasoconstrictor and vasodilator effects through endothelium–dependent mechanisms23. Lastly, it is also conceivable that the prognostic value of serum leptin is different in the general population than in dialysis patients, as it has been suggested for adiponectin24.
Our study has some strengths and limitations. First, this is the first multicenter study assessing leptin values in HD patients and is also the first study that analyzes corrected leptin values in relation to all–cause and CVD–related mortality in these patients. A limitation of our study comes from the fact that classification of cardiovascular disease was made on the basis of clinically manifest event, and therefore the true prevalence of atherosclerotic disease may be underestimated. We could not correct leptin concentrations by fat mass, although we did correct for BMI values. Another limitation is its low statistical power and the fact that we studied three cohorts of patients at three different baseline times and, therefore, with different times of follow–up and mortality rates, and also with differences in some clinical and biochemical characteristics. In fact, in comparison with cohorts 1 and 2, cohort 3 patients had older age, higher proportion of diabetes and lower levels of creatinine and cholesterol. All–cause and CVD mortality was lower in cohort 3, a fact in direct relationship with the lower follow–up period in this cohort. However, laboratory procedures for leptin measurement and clinical protocols for patients follow–up were the same. Furthermore, survival analysis performed in the three cohorts of patients separately did not show any significant relationship between leptin/BMI and all–cause or cardiovascular mortality.
The clinical corollary of our study would be stopping the qualification of leptin as a cardiovascular risk factor in HD patients. In a similar way, leptin has been considered by some authors as a causal factor of cachexia in uremic patients. However, previous data from our group showed not only a correlation between leptin and BMI, but also a direct relationship between this hormone and albumin, transferrin and cholesterol10,25,26. Besides, our patients with anorexia exhibited low, rather than high, serum leptin levels26.
In conclusion, in this population of stable HD patients, obtained results do not support the hypothesis that serum leptin and leptin/BMI ratio are related with prevalent CVD and are risk factors for all–cause and cardiovascular mortality.
Conflict of interest statements
R. Selgas has received a non–restricted grant by Baxter to support part of this investigation.
Acknowledgements
The authors are specially indebted to the nurses of our dialysis units for their collaboration.
Referencias Bibliográficas
1. Rucker D, Tonelli M. Cardiovascular risk and management in chronic kidney disease. Nat Rev Nephrol 2009;5:287–96. [ Links ]
2. Brunner FP, Selwood NH on behalf of the EDTA Registry Committee. Profile of patients on RRT in Europe and death rates due to major causes of death groups. Kidney Int 1992;42:S4–15. [ Links ]
3. Locatelli F, Manzoni C, Del Vecchio L, Di Filippo S. Changes in United States Renal Data System, 2006. [ Links ]
4. Herzog CA. Sudden cardiac death and acute myocardial infarction in dialysis patients: perspectives of a cardiologist. Semin Nephrol 2005;25:363–6. [ Links ]
5. Bodary PF, Gu S, Shen Y, Hasty AH, Buckler JM, Eitzman DT. Recombinant leptin promotes atherosclerosis and thrombosis in apolipoprotein E–deficient mice. Arterioscler Thromb Vasc Biol 2005;25:e119–22. [ Links ]
6. Nakamura Y, Ueshima H, Okuda N, Murakami Y, Miura K, Kita Y, et al. Relation of serum leptin to blood pressure of Japanese in Japan and Japanese–Americans in Hawaii. Hypertension 2009;54:1416–22. [ Links ]
7. Bullo M, García–Lorda P, Megias I, Salas–Salvadó J. Systemic inflammation, adipose tissue, tumor necrosis factor and leptin expression. Obes Res 2003;11:525–31. [ Links ]
8. Heimbürger O, Lönnqvist F, Danielsson A, Nordenström J, Stenvinkel P. Serum immunoreactive leptin concentration and its relation to the body fat content in chronic renal failure. J Am Soc Nephrol 1997;8:1423–30. [ Links ]
9. Bossola M, Muscaritoli M, Valenza V, Panocchia N, Tazza L, Cascino A, et al. Anorexia and serum leptin levels in hemodialysis patients. Nephron Clin Pract 2004;97:c76–82. [ Links ]
10. Díez JJ, Iglesias P, Fernández–Reyes MJ, Aguilera A, Bajo MA, Álvarez–Fidalgo P, et al. Serum concentrations of leptin, adiponectin and resistin, and their relationship with cardiovascular disease in patients with end–stage renal disease. Clin Endocrinol 2005;62:242–9. [ Links ]
11. Scholze A, Rattensperger D, Zidek W, Tepel M. Low serum leptin predicts mortality in patients with chronic kidney disease stage 5. Obesity (Silver Spring) 2007;15:1617–22. [ Links ]
12. Considine RV, Sinha MK, Heiman ML, Kriauciunas A, Stephens TW, Nyce MR, et al. Serum immunoreactive–leptin concentrations in normal–weight and obese humans. N Engl J Med 1996;334:292–5. [ Links ]
13. Eikelis N, Lambert G, Wiesner G, Kaye D, Schlaich M, Morris M, et al. Extra–adipocyte leptin release in human obesity and its relation to sympathoadrenal function. Am J Physiol Endocrinol Metab 2004;286:E744–52. [ Links ]
14. Reilly MP, Iqbal N, Schutta M, Wolfe ML, Scally M, Localio AR, et al. Plasma leptin levels are associated with coronary atherosclerosis in type 2 diabetes. J Clin Endocrinol Metab 2004;89:3872–8. [ Links ]
15. Singhal A, Farooqi IS, Cole TJ, O'Rahilly S, Fewtrell M, Kattenhorn M, et al. Influence of leptin on arterial distensibility: a novel link between obesity and cardiovascular disease? Circulation 2002;106:1919–24. [ Links ]
16. Wallace AM, McMahon AD, Packard CJ, Kelly A, Shepherd J, Gaw A, et al. Plasma leptin and the risk of cardiovascular disease in the west of Scotland coronary prevention study (WOSCOPS). Circulation 2001;104:3052–6. [ Links ]
17. Sattar N, Wannamethee G, Sarwar N, Chernova J, Lawlor DA, Kelly A, et al. Leptin and coronary heart disease. Prospective study and systematic review. J Am Coll Cardiol 2009;53:167–75. [ Links ]
18. Kalantar–Zadeh K, Block G, Humphreys MH, Kopple JD. Reverse epidemiology of cardiovascular risk factors in maintenance dialysis patients. Kidney Int 2003;63:793–808. [ Links ]
19. Abbott KC, Glanton CW, Trespalacios FC, Oliver DK, Ortiz MI, Agodoa LY, et al. Body mass index, dialysis modality, and survival: analysis of the United States Renal Data System Dialysis Morbidity and Mortality Wave II Study. Kidney Int 2004;65:597–605. [ Links ]
20. Liu Y, Coresh J, Eustace JA, Longenecker JC, Jaar B, Fink NE, et al. Association between cholesterol level and mortality in dialysis patients: role of inflammation and malnutrition. JAMA 2004;291:451–9. [ Links ]
21. Fouque D, Vennegoor M, Ter Wee P, Wanner C, Basci A, Canaud B, et al. EBPG Guideline on nutrition. Nephrol Dial Transplant 2007;Suppl. 2:ii45–87. [ Links ]
22. Sundell J, Huupponen R, Raitakari OT, Nuutila P, Knuuti J. High serum leptin is associated with attenuated coronary vasoreactivity. Obes Res 2003;11: 776–82. [ Links ]
23. Vecchione S, Maffei A, Colella S, Aretini A, Poulet R, Frati G, et al. Leptin effect on endothelial nitric oxide is mediated through Akt–endothelial nitric oxide synthase phosphorylation pathway. Diabetes 2002;51:168–73. [ Links ]
24. Díez JJ, Estrada, Bajo MA, Fernández–Reyes MJ, Grande C, Del Peso G, et al. High stable serum adiponectin levels are associated with a better outcome in prevalent dialysis patients. Am J Nephrol 2009;30:244–52. [ Links ]
25. Iglesias P, Díez JJ, Fernández–Reyes MJ, Bajo MA, Aguilera A, Méndez J, et al. Effects of short–term recombinant human growth hormone therapy on plasma leptin concentrations in dialysis patients. Nephrol Dial Transplant 2002;17:260–4. [ Links ]
26. Aquilera A, Bajo MA, Rebollo F, Díez JJ, Díaz C, Paiva A, et al. Leptin as a marker of nutrition and cardiovascular risk in peritoneal dialysis patients. Adv Perit Dial 2002;18:212–7. [ Links ]
![]() Correspondence:
Correspondence:
Juan José Díez Gómez,
Servicio de Endocrinología,
Hospital Ramón y Cajal, Madrid,
E–mail: jdiez.hrc@salud.madrid.org
Sent for review: 5 Sep. 2010
Accepted: 13 Dic. 2010














