Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Nutrición Hospitalaria
versión On-line ISSN 1699-5198versión impresa ISSN 0212-1611
Nutr. Hosp. vol.24 no.3 Madrid may./jun. 2009
Effect of non-steroidal anti-inflammatory drug etoricoxib on the hematological parameters and enzymes of colon and kidney
Efecto del fármaco antiinflamatorio no esteroideo etoricoxib sobre los parámetros hematológicos y las enzimas del colon y el riñón
N. Behal, S. Singh Kanwar, P. Sharma and S. N. Sanyal
Department of Biophysics. Panjab University. Chandigarh. India.
ABSTRACT
The present study was designed to investigate the effects of a selective COX-2 inhibitor, etoricoxib in rats on the hematological and toxicity parameters in colon and kidney at two different doses of the drug, one within the therapeutic anti-inflammatory range as based on the reported ED50 value (Eto-1) while the other at ten times higher (Eto-2), relative to the toxicity studies which have not been reported so far. The results showed that the control and the drug treated animals achieved similar linear growth rate and also showed no major alterations in the histological parameters in the liver and kidney tissue. The animals treated with lower dose of etoricoxib showed an overall decrease in total leukocytes counts as well as in the number of neutrophils, lymphocytes, monocytes and eosinophills while the higher dose of the drug produced a highly significant increase in all the cell counts. However, the drug treatment at both the dose level produced significant fall in the activities of alkaline phosphatase, sucrase, lactase and maltase in the kidney but increased the activity of alkaline phosphatase in colon. The treatment of etoricoxib did not produce any change in the nitric oxide and citrulline levels in kidney while an increase was noted in the colonic tissue. It was concluded that etoricoxib is a relatively safe drug at its anti-inflammatory ED50 dose in rats when the hematological parameters and the structural and functional characteristics of kidney and colonic tissues were studied.
Key words: Non-steroidal anti-inflammatory drug etoricoxib. Leukocyte cell counts. Enzymes of rat colon and kidney.
RESUMEN
El presente estudio se diseñó para investigar los efectos de un inhibidor selectivo de la COX-2, etoricoxib, sobre los parámetros hematológicos y de toxicidad en colon y riñón de rata, con dos dosis distintas del fármaco, una dentro del rango terapéutico sobre la base del valor ED50 notificado (Eto-1) mientras que la otra fue diez veces superior (Eto-2), relativa a los estudios de toxicidad que aún no han sido publicados. Los resultados mostraron que los animales control y los tratados consiguieron tasas de crecimiento linear similares y no mostraron alteraciones importantes en los parámetros histológicos del hígado o riñón. Los animales tratados con la dosis inferior de etoricoxib mostraron una disminución global del recuento de neutrófilos, linfocitos, monocitos y eosinófilos, mientras que la dosis superior del fármaco produjo un aumento significativo de todos los recuentos celulares. Sin embargo, el tratamiento con el fármaco a ambas dosis produjo una caída significativa de las actividades de la fosfatasa alcalina, sucrasa, lactasa y maltasa del riñón y una actividad aumentada de la fosfatasa alcalina del colon. El tratamiento con etoricoxib no produjo ningún cambio en las concentraciones de óxido nítrico ni de citrulina en el riñón pero sí se observó un aumento en el tejido colónico. Se concluyó que el etoricoxib es un fármaco relativamente seguro a su dosis ED50 antiinflamatoria en ratas cuando se estudiaron los parámetros hematológicos y las características estructurales y funcionales de los tejidos renal y colónico.
Palabras clave: Fármaco antiinflamatorio no esteroideo etoricoxib. Recuentos leucocitarios. Enzimas del colon y riñón de rata.
Introduction
Non-steroidal anti-inflammatory drugs (NSAIDs) have come to play an important role over the years in pharmacological management of arthritis and pain.1 NSAIDs are effective in controlling the joint pain and swelling in rheumatoid arthritis and have also shown in recent times to prevent the formation of cancer in different tissues.2 However, NSAIDs are associated in humans with toxicities such as gastro-intestinal ulcer and bleeding.3 The anti-inflammatory action of NSAIDs can be explained by their capability to inhibit the synthesis of prostaglandins, particularly to inhibit the cyclooxygenase (COX) enzymes.4 COX is demonstrated to be existing as three distinct isoforms, COX-1, COX-2 and COX-3.5 COX-1 is expressed constitutively in human kidney and brain, while its expression is being induced in many tissues during inflammation, normal wound healing and neoplasia.6 Therefore, it was proposed that the selective COX-2 inhibitors may become more effective and safe chemopreventive agents than classical NSAIDs which preferentially inhibit COX-1.7 Studies have also shown that the selective COX-2 inhibitors are effective and well tolerated in treatments for rheumatoid arthritis and other inflammatory disorders.8,9
Etoricoxib (5-chloro-6-methyl-3-[4-(methyl sulfonyl) phenyl]-2-3'-bipyridine) is a recent entry into the field of selective COX-2 inhibitors that has been developed for treatment of osteoarthritis, rheumatoid arthritis and pain.10 Etoricoxib, being COX-2 inhibitor, has the therapeutic advantage of decreasing inflammation at the tissue sites particularly in joints, while sparing gastrointestinal mucosa due to continued prostaglandin production via the COX-1 isoform.11 However, COX-2 enzymes are also expressed at multiple nephron sites in the mammalian kidney including the cortical thick ascending limb, macula densa, medullary interstitial cells and the endothelium of arteries and veins as well as glomerular podocytes.12 Thus, it is possible that inhibition of COX-2 enzymes may be associated with alterations in renal functions. Also, the expression of COX-2 is increasingly induced during consecutive stages of cancer. The role of this enzyme in colorectal carcinogenesis is well established by Oshima and Taketo13 showing that COX-2 deficiency partly suppressed the familial adenomatous polyposis and cancers. Thus, the chemopreventive role of COX-2 inhibitors is also a major consideration in their therapeutic dose and being investigated in the present laboratory in experimental colon cancer.14-18
In the present study, an attempt has been made to investigate the effect of the COX-2 selective inhibitor, etoricoxib on rat kidney and colon histoarchitecture and enzyme profiles at two different doses, one at within its therapeutic anti-inflammatory range as based on the reported ED50 value for rats while the other at a ten times higher dose which is expected to relate to the toxicity effects. In view of the immunosuppressive response of the drug, the hematological parameters were also studied in the peripheral blood leukocytes.
Materials and methods
The experiments were performed on male Wistar rats of body weights ranging between 135-150 g, obtained from the central animal house of Panjab University. The animals were housed in plastic cages embedded with rice husk and maintained on standard rodent feed, and also had free access to water. The body weights of the animals were recorded weekly in a single pan animal scale. All of the animal procedures as reported here followed the guidelines approved by the Panjab University Ethical Committee on the use of the experimental animals for biomedical research.
A six week study was designed where the animals were divided into three different groups having 6 animals each. Group 1: The animals served as control receiving the daily dose of 0.5% carboxy methyl cellulose sodium salt as vehicle of the NSAID. Group 2: The animals were given a daily oral administration of Etoricoxib at a concentration of 0.64 mg/kg body wt, called Eto-1. Group 3: The animals were given a daily oral administration of Etoricoxib at a concentration of 6 mg/kg body weight, called Eto-2.
At the end of the six weeks duration, the animals were anesthetized with ether and blood collected from the ocular vein with a glass capillary. The animals were thereof sacrificed with an overdose of ether and tissues collected.
Total leukocytes count (TLC) was done following the methods of Dacie and Lewis19 in blood samples diluted with freshly prepared Turk's solution containing crystal violet stain and using a WBC diluting tube (Thomas White Cell Pipette) with a dilution of 1:20. The cells were then counted in a Neubauer Chamber and using a light microscope. Differential leukocyte count (DLC) was done by the thick smear method of blood followed by Field's stain which contained the stains, Azure A, Methylene blue and Eosin. Eosinophills appeared as bright red, large cells with well defined granules, neutrophils as pale purple pink and small indistinct cells, basophils as deep blue cells with reddish cast while the monocytes were large sized cells with kidney shaped nucleus.
For histopathological studies, small pieces of colon and kidney were taken, washed with ice-cold 0.9% saline and fixed in Bouin's fixative for 24 hours.20 After fixation, the tissues were processed carefully for paraffin wax (58-60º C), embedded in the wax and sections were cut at 5 μM thickness in a microtome. Parrafin sections as taken on an albumin coated glass slides as a continuous ribbon were dewaxed in xylene, down graded (hydrated) in decreasing percentage of alcohols and brought to water, stained with haematoxylin for 20 sec and washed in tap water till the appearance of blue color. The slides were then rinsed in ammonia water, again washed with water and treated with acid water if over stained. The sections were upgraded in alcohol till 70%, stained with 1% alcoholic eosin for 30 sec and differentiated in 90% alcohol (Delafield hematoxylineosin technique). The slides were cleaned in xylene and finally mounted in distyrene plasticizer xylene (DPX).21
The kidney and colonic tissues were dissected, washed and a 10% homogenate of the tissue made in chilled 1 mM tris-50 mM mannitol buffer (pH 7.4). For colon, the intestinal segment starting from the ligament of Triez was dissected and thoroughly flushed with the chilled buffer. The homogenate was centrifuged at 1,000 x g for 10 min at 4º C. The pellet was discarded and the supernatant was used for various biochemical estimations.
Alkaline phosphatase activity was assayed according to the method of Bergmeyer22 where p-nitrophenyl phosphate was used as the substrate which was hydrolyzed by the enzyme to yield p-nitrophenol at an alkaline pH. The yellow color of p-nitrophenol was measured at 410 nm.
The activity of the three disaccharidase enzymes, sucrase, lactase and maltase was determined by measuring the D-glucose liberated from the respective sugar substrate using a glucose oxidase-peroxidase (GODPOD) enzymatic system.23 Nitric oxide (NO) production by nitric oxide synthase was estimated by measuring the nitrite, a stable metabolic product of NO which quickly reacts with oxygen to yield nitrite. Nitrite thus formed, reacts with Griess reagent to form a purple azo dye, the color of which can be read at 540 nm.24 The citrulline assay was based on its reaction with diacetylmonooxime and the absorbance of the color produced was measured at 530 nm.
Results
The weight change profile showed a linear growth in the body weight during a complete six week treatment schedule which was shown in figure 1. No significant change in the body weight was observed between the control and the treated animals. Table I shows the total and the differential leukocyte counts in the different treatment groups during a six weeks treatment regimen. Eto-1 group showed an overall decrease in total leukocyte counts as well as the individual count of neutrophils, lymphocytes, monocytes and eosinophils in comparison to the control group. On the other hand, Eto-2 group showed a fairly significant increase in the leukocyte counts. The neutrophils did not show much change in the cell counts.
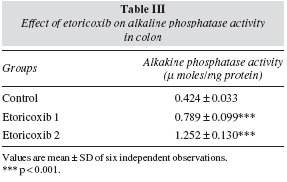
Table II demonstrated the activity of the four different enzymes in the kidney namely, alkaline phosphatase, sucrase, lactase and maltase which shows a highly significant decrease in both the Eto-1 and Eto-2 groups when compared to the controls. However, the alkaline phosphatase activity in colon showed a significant increase in both the treated groups (table III). Table IV demonstrates that there was no significant change in the nitric oxide and citrulline level in kidney in the Eto-1 and Eto-2 groups of the animals as compared to the controls while an increase was registered in the colonic tissues for the same in both the treatment groups.
Histologicaly, the paraffin embedded sections of colon and kidney were critically examined under light microscope following hematoxylin and eosin staining (H/E). The normal histological architecture of colon comprises of mucosal layer containing crypt lined with the various epithelial cells which included absorptive columnar cells, mucin producing goblet cells and neuroendocrine cells (fig. 2). These are embedded in the connective tissues called stroma. Mucosa rests over the sub mucosa below which lines the muscularis mucosa. The H/E results produced no major histoarchitectural changes in etoricoxib treated animals when compared with the control. Figure 3 shows the histoarchitecture of the kidney cortex. The Bowman's capsule structure was prominent. However, no major alteration was seen in both the etoricoxib groups in kidney, when compared to the controls (fig. with Eto-2 not shown). Figure 4 which shows the effect of Eto-1 treatment on the kidney histoarchitecture in the medullary region also revealed no major change in the same.
Discussion
NSAIDs are among the most widely prescribed class of pharmaceutical agents worldwide, having broad clinical utility in treating pain, fever and inflammation.25,26 The anti-inflammatory action of NSAIDs rests in their ability to inhibit the activity of COX enzymes which in turn results in a diminished synthesis of proinflammatory prostaglandins (PGs).27 COX-1 and COX-2, the two isoforms of COX are almost identical in structure but have important differences in substrate and inhibitor selectivity.28 COX-1 synthesizes protective PGs which preserved the integrity of stomach and intestinal lining and maintain normal functioning of kidney as well. It also plays a role in the production of thromboxane A2, causing an aggregation of platelets to prevent inappropriate bleeding.29 On the other hand COX-2 emerged out as an inducible isoform for it could be induced by inflammatory stimulus and by cytokines in migratory and other cells.30 The discovery of COX-2 isoenzyme and the characterization of its role in inflammation fostered the development of a new class of compounds that selectively inhibits COX-2 without affecting the COX-1 dependent PG biosynthesis necessary for physiological functions.31 This new generation of anti-inflammatory drugs has been proven in vitro to selectively inhibit COX-2 activity and used to be as efficacious as the standard NSAIDs (both COX-1 and COX-2 inhibitors) in a number of in vivo models of inflammation (rat carrageenan-induced foot paw edema and rat adjuvant-induced arthritis)32 and hyperalgesia (rat carrageenan-induced hyperalgesia).33 Two selective COX-2 inhibitors in particular (called COXIBs) rofecoxib and celecoxib, have proven to provide significant relief in clinical trials of the signs and symptoms of osteoarthritis and rheumatoid arthritis and in eliminating pain following dental extraction, while reducing the incidence of gastrointestinal ulcers and erosions as seen with standard NSAID therapy.34 Moreover, these eagerly awaited highly selective COX-2 inhibitors are of great interest because they may represent an alternative therapeutic option for the treatment of inflammation in diseases such as cirrhosis with ascites in which renal function is critically dependent on PGs.35 However, in recent times unfortunately both rofecoxib and celecoxib have been reported to produce cardiovascular complications36,37 and therefore necessitates searching for safer drugs. A second generation of selective COX-2 inhibitors such as valdecoxib, lumiracoxib and etoricoxib is currently under evaluation for clinical use,38 and among these, etoricoxib has shown particular promise in chemoprevention in colon cancer in animal studies recently in our laboratory.14-18
The results of the present study wherein two different doses of etoricoxib were used in rats, one at the anti-inflammatory therapeutic dose and the other at ten times higher conclusively show that the drug does not interfere with the normal growth profile of the animal as well as the structural and functional parameters of kidney and colon. However, it was also demonstrated that etoricoxib at both the dose can influence the activities of sucrase, maltase, lactase and alkaline phosphatase in kidney. These enzyme activities are intimately associated with the process of membrane transport in the kidney brush border as shown by us earlier.39 An increase in alkaline phosphatase activity in colon is seen in the present study where it has been reported to have a link with the transport of phosphate and calciumions40 may play a crucial role in signal transduction mechanism in the colonic brush border membrane.41
NO is produced by three nitric oxide synthase (NOs) enzymes. Endothelial NOs and neuronal NOs are both constitutively expressed while inducible NOs expression is enhanced by various inflammatory cytokines.42 The role of NO in carcinogenesis is controversial as NO has both anti-tumor and tumor promotive properties. 43 The principal role of NO in the cellular microenvironment may depend on various factors such as the level of NO production and the genetic make up of the cells.44 There are two pathways of citrulline production. It can be derived from arginine/NOs pathway and/or arginine/arginase pathway.45 NOs pathway being responsible for the production of NO as well as citrulline, may play a major role in cell growth and proliferation. The present study of etoricoxib treatment at two different dose level led to no significant change in NO and citrulline level in the kidney tissues, while in colon only, NO was found to be increased and the citrulline level unaltered. Etoricoxib may help in replenishing the highly protective cells of colon as a little higher level of NO than normal is required for cell differentiation. 46 The results therefore suggest that etoricoxib may modulate the cellular environment in case of colon while no effects are seen in the kidney. Based on the present results it can be concluded that the specific COX-2 inhibitor, etoricoxib is a safe drug for therapeutic use at its ED50 value.
References
1. Vane JR, Bottling RM. The mechanism of action of aspirin. Thromb Res 2003; 110: 255-258. [ Links ]
2. Sengupta S, Lynda A, Sellers et al. Cyclooxygenase-2-selective nonsteroidal anti-inflammatory drugs inhibit hepatocyte growth factor/scatter factor-induced angiogenesis. Cancer Res 2003; 63: 8351-8359. [ Links ]
3. Gretzer B, Maricic N, Respondek M et al. Effects of specific inhibition of cyclo-oxygenase-1 and cyclo-oxygenase-2 in the rat stomach with normal mucosa and after acid challenge. Br J Pharmacol 2001; 132: 1565-1573. [ Links ]
4. Vane JR, Bakhle YS, Botting RM. Cyclooxygenase 1 and 2. Ann Rev Pharmacol Toxicol 1998; 38: 97-120. [ Links ]
5. Willoughby DA, Moore AR, Colville-Nash PR. COX-1, COX-2 and COX-3 and the future treatment of chronic inflammatory disease. Lancet 2000; 355: 646-648. [ Links ]
6. Bombardier, Laine L, Reicin A. Comparison of upper gastrointestinal toxicity of rofecoxib and naproxen in patients with rheumatoid arthritis. New Engl J Med 2000; 343: 1520-1528. [ Links ]
7. Schwartz JI, Chan, CC, Mukhopadhyay S. Cyclooxygenase-2 inhibition by rofecoxib reverses naturally occurring fever in humans. Clin Pharmacol Ther 1999; 65: 653-660. [ Links ]
8. Emery P, Zeidler H, Kvien TK et al. Celecoxib versus diclofenac in long- term management of rheumatoid arthritis: randomized double-blind comparison. Lancet 1999; 354: 2106-2111. [ Links ]
9. Matsumoto J, Melian A, Mandel DR. A randomized, controlled, clinical trial of etoricoxib in the treatment of rheumatoid arthritis. J Rheumatol 2002; 29: 1623-1630. [ Links ]
10. Riendeau D, Percival MD, Brideau C. Etoricoxib: a preclinical profile and comparison with other agents that selectively inhibit cyclooxygenase-2. J Pharmacol Exp Ther 2001; 296: 558-566. [ Links ]
11. Gregory LB, Michael HS, Jeffrey GM et al. Acute renal failure and hyperkalaemia associated with cyclooxygenase-2 inhibitors. Nephrol Dial Transplant 2004; 19: 1149-1153. [ Links ]
12. Harris RC. Cyclooxygenase-2 in the kidney. J Am Soc Nephrol 2000; 11: 2387-2394. [ Links ]
13. Oshima M, Taketo MM. COX inhibitor, suppression of polyposis, and chemoprevention. Nippon Yakurigaku Zasshi 2002; 120 (5): 276-84. [ Links ]
14. Kanwar SS, Vaiphei K, Nehru B et al. Chemopreventive effects of non steroidal anti-inflammatory drugs on 1, 2-dimethylhydrazine-induced colon carcinogenesis in rats. Toxicol Mecha Meth 2007; 17: 1-8. [ Links ]
15. Kanwar SS, Vaiphei K, Nehru B et al. Chemopreventive effects of non-steroidal anti-inflammatory drugs in the membrane lipid composition and fluidity parameters of the 1, 2-dimethylhydrazine induced colon carcinogenesis in rats. Drug Chem Toxicol 2007; 30: 293-30. [ Links ]
16. Kanwar SS, Vaiphei K, Nehru B et al. Antioxidative effects of Non-steroidal anti-inflammatory drugs (NSAIDs) during the initiation stages of experimental colon carcinogenesis in rats. J Env Pathol Toxicol Oncol 2008; 27 (2): 89-100. [ Links ]
17. Mittal N, Kanwar SS, Sanyal SN. The effect of etoricoxib, a cyclooxygenase-2 specific inhibitor on the 1, 2-dimethylhydrazine administered rat intestinal membrane structure and function. Toxicol Mech Meth 2007; 18: 53-62. [ Links ]
18. Mittal N, Kanwar SS, Sanyal SN. Effect of non-steroidal antiinflammatory drugs and the pro-carcinogen 1, 2 dimethylhydrazine on the antioxidant defense system. Int J Toxicol 2007; 27: 169-174. [ Links ]
19. Dacie JV, Lewis K. Practical Haematology. 4th ed. Churchill, London 1968. [ Links ]
20. Pearse AGE. In: Histochemistry, Theoritical and Applied. 3rdEdn, p-660 Vol 1. Churchill, Livingstone, London 1968. [ Links ]
21. Humanson GL. In: Basic procedures - Animal tissue technique, Part-1, pp. 1961; 130-132. [ Links ]
22. Bergemeyer HU. Phosphatase (phosphomonoesterases). Determination in serum with p - nitrophenyl phosphate. In: Methods of Enzymatic Analysis, HU Bergmeyer (Ed), 1963, pp. 783-785. Academic Press, New York. [ Links ]
23. Dahlqvist A. Methods for assay of intestinal disaccharides. Anal. Biochem 1964; 7: 18-25. [ Links ]
24. Boyde TR, Rahmatullah M. Optimization of conditions for the colorimetric determination of citrulline, using diacetyl monoxime. Anal Biochem 1980; 107 (2): 424-31. [ Links ]
25. Flower RJ, Moncada S, Vane JR et al. Analgesic - antipyretics and anti inflammatory agents: drugs employed in treatment of gout. The Pharmacological Basis of Therapeutics: Macmillan. 1992; pp. 674. [ Links ]
26. Payan DG, Katzung BG. NSAID's: Non-opioid analgesics; drugs used in gout, in: Basic and Clinical Pharmacology; Katzung BG (Ed), 1995; pp. 536-559. Appleton and Lange, Norwalk. [ Links ]
27. Vane JR. Inhibition of prostaglandin synthesis as a mechanism of action for aspirin- like drugs. Nat New Biol 1991; 231: 232-235. [ Links ]
28. Jouzeau JY, Terlain B, Abid A et al. Cyclo-oxygenase isoenzymes. How recent findings affect thinking about nonsteroidal anti-inflammatory drugs. Drugs 1997; 53 (4): 563-582. [ Links ]
29. Funk CD, Funk LB, Kennedy ME et al. Human platelet/erythroleukemia cell prostaglandin G/H synthase: cDNA cloning, expression, and gene chromosomal assignment. FASEB J 1991 5: 2304-2312. [ Links ]
30. Raz A, Wyche A, Fagan D et al. The cell biology of fibroblast cyclooxygenase. Adv Exp Med Biol 1989; 259: 1-21. [ Links ]
31. Gilroy DW, Tomlinson A, Willoughby DA. Differential effects of inhibitors of cycloxygenase (cycloxygenase 1 and cycloxygenase 2) in acute inflammation. Eur J Pharmacol 1998; 355: 211-217. [ Links ]
32. Smith CJ, Zhang Y, Koboldt CM. Pharmacological analysis of cycloxygenase-1 in inflammation. Proc Natl Acad Sci (USA) 1998; 95: 13313-13318. [ Links ]
33. Warner TD, Giuliano F, Vojnovic I et al. Nonsteroid drug selectivities for cyclo-oxygenase 1 rather than cyclo-oxygenase 2 are associated with human gastrointestinal toxicity: a full in vitro analysis. Proc Natl Acad Sci (USA) 1999; 967: 563-756. [ Links ]
34. Lanas A. Clinical experience with cyclooxygenase-2 inhibitors. Rheumatol 2002; (Supl. 1): 16-22. [ Links ]
35. López-Parra M, Claria J, Planaguma A et al. Cyclooxygenase-1 derived prostaglandins are involved in the maintenance of renal function in rats with cirrhosis and ascitses. Brit J Pharm 2002; 135: 891-900. [ Links ]
36. Aguilar JFN, Antich IA, Martinez AP et al. Cycloxygenase-2 inhibition in colon experimental carcinogenesis. Rev Esp En En form Dig (Madrid) 2005; 97: 637-647. [ Links ]
37. Subhashini J, Mahipal SVK, Reddana P. Anti-proliferation and apoptotic effects of celecoxib on human chronic myeloid leukemia in vitro. Cancer Lett 2005; 224: 31-43. [ Links ]
38. Esser R, Berry C, Du Z et al. Preclinical pharmacology of lumiracoxib: a novel selective inhibiter of cycloxygenase-2. Br J Pharmacol 2005; 144: 538-550. [ Links ]
39. Sanyal SN, Singh G, Kanwar SS. Thermotropic lipid phase transition and the behavior of hydrolytic enzymes in the cortex brush border membrane. Chem Biodiver 2006; 3: 1102-1115. [ Links ]
40. Shiffle H, Binswanger V. Calcium ATPase and intestinal calcium transport in uremic rats. Am J Physiol 1980; 238: G 424. [ Links ]
41. Lizumi Y, Hirano K, Sugoira M et al. Evidence for the physiological function of alkaline phasohatase. Chem Pharm Bull 1983; 32: 772-775. [ Links ]
42. Nathan C, Xie QW. Regulation of biosynthesis of nitric oxide. J Biol Chem 1994; 269: 13725-13728. [ Links ]
43. Ricciardolo FL, Sterk PJ, Gaston B. Nitric oxide in health and disease of the respiratory system. Physiol Rev 2004; 84: 731-765. [ Links ]
44. Wu G, Morris SM. Arginine metabolism: nitric oxide and beyond. Biochem J 1998; 336: 1-17. [ Links ]
45. Lala PK. Significance of nitric oxide in carcinogenesis, tumor progression and cancer therapy. Cancer Metastasis Rev 1998; 17: 1-6. [ Links ]
46. Stuehr DJ, Marletta MA. Synthesis of nitrite and nitrate in murine macrophage cell lines. Cancer Res 1987; 47 (21): 5590-5594. [ Links ]
![]() Correspondence:
Correspondence:
S. N. Sanyal.
Department of Biophysics.
Panjab University.
160014 Chandigarh - India.
E-mail: sanyalpu@pu.ac.in
Recibido: 15-IX-2008.
Aceptado: 2-II-2009.