Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Nutrición Hospitalaria
versión On-line ISSN 1699-5198versión impresa ISSN 0212-1611
Nutr. Hosp. vol.28 no.3 Madrid may./jun. 2013
https://dx.doi.org/10.3305/nh.2013.28.3.6284
ORIGINAL
Can the exercise mode determine lipid profile improvements in obese patients?
¿El modo de ejercicio puede ser determinante en la mejora del perfil lipídico en pacientes con obesidad?
Blanca Romero Moraleda1, Esther Morencos1, Ana Belén Peinado1, Laura Bermejo2, Carmen Gómez Candela2, Pedro José Benito1; on behalf of the PRONAF Study group.
1Department of Health and Human Performance. School of Physical Activity and Sport Sciences. Technical University of Madrid. Madrid. Spain.
2Nutrition Department. University Hospital La Paz. Madrid. Spain.
The PRONAF Study takes place with the financial support of the Ministerio de Ciencia e Innovación, Convocatoria de Ayudas I+D 2008, Proyectos de Investigación Fundamental No Orientada, del VI Plan de Investigación Nacional 2008-2011 (Contrac: DEP2008-06354-C04-01).
ABSTRACT
Introduction: Unfavorable lipid profile is associated with developed cardiovascular diseases. It is necessary to know the beneficial effects of different mode exercises to improve lipid profile.
Objective: To investigate, in obese men and women, the effect on lipid profile of hypocaloric diet combined with structured exercise programs or recommendations of physical activity.
Methods: Ninety six obese subjects (59 women and 61 men; 18 - 50 years; BMI >30 and < 34.9 kg/m2) were randomised into four supervised treatment groups: strength training (S; n = 24), endurance training (E; n = 26), combined S + E (SE; n = 24), and and received recommendations of physical activity (PA; n = 22). Energy intake, body composition, training variables (VO2peak, strength index, dynamometric strength index) and blood lipid profile were recorded at baseline and after 24 weeks of treatment.
Results: Blood lipid profile improved in all groups. No statistically significant differences in baseline and posttraining values were observed between groups. HDL-Cholesterol showed no changes. A decrease in LDL-Cholesterol values was observed in all groups after the intervention (S: 11.2%, E: 10.8%, SE: 7.9%, PA: 10.8%; p < 0.01). S, E and PA subjects showed decrease in triglycerides (S: 14.9%, E: 15.8%, PA: 15.7%; p < 0.01). Total cholesterol decreased in all groups (S: 8.4%, p < 0.01; E: 8.8%, p < 0.01; SE: 4.9%, p < 0.01; PA: 8.3%, p < 0.05).
Conclusion: All protocols proposed in our study improved blood lipid profile in obese people. There were no significant differences about the effect on the lipid profile between the implementation of a structured training protocol with physical activity professional supervision and follow recommendations of physical activity.
Key words: Lipoprotein. Obese. Strength training. Aerobic training. Combined training.
RESUMEN
Introducción y objetivo: El perfil lipídico desfavorable se asocia con el desarrollo de enfermedades cardiovasculares. Para reducir este factor es necesario estudiar el impacto que los diferentes modos de ejercicio con dieta tienen sobre el perfil lipídico. Por ello, el objetivo de este trabajo fue investigar, en hombres y mujeres obesos, el efecto sobre el perfil lipídico de la dieta hipocalórica combinada con programas de ejercicios estructurados o recomendaciones de actividad física.
Métodos: Noventa y seis participantes con obesidad (edad entre 18-50; IMC > 30 and < 34.9 kg/m2) fueron repartidos en 4 grupos: fuerza (S; n = 24), aeróbico (E; n = 26), combinado de fuerza y aeróbico (SE; n = 24), los cuales entrenaron 3 veces/semana durante 22 semanas, y el grupo de recomendaciones de actividad física (PA; n = 22). A todos se les asigno una dieta equilibrada con un 35% de restricción. Antes y después de la intervención todos los grupos fueron evaluados de los cambios en el perfil lipídico, la composición corporal y la ingesta diaria.
Resultados: El perfil lipídico mejoró en todos los grupos. No se observaron diferencias significativas en los valores basales y tras la intervención entre los grupos. El HDL no mostró cambios. Para los valores de LDL se observó una disminución significativa en todos los grupos (S: 11,2%, E: 10,8%, SE: 7,9%, PA: 10,8%). Los sujetos S, E y PA mostraron una disminución en los triglicéridos (S: 14,9%, E: 15,8%, PA: 15,7%; p < 0,01). El colesterol total disminuyó significativamente en todos los grupos (S: 8,4%, E: 8,8%, SE: 4,9%, PA: 8,3%).
Conclusiones: Todos los protocolos propuestos en nuestro estudio mejoraron el perfil lipídico en personas obesas. No hubo diferencias significativas en cuanto al efecto sobre el perfil lipídico entre la aplicación de un protocolo de entrenamiento estructurado que seguir las recomendaciones de actividad física.
Palabras clave: Lipoproteínas. Obesidad. Entrenamiento con cargas. Entrenamiento aeróbico. Entrenamiento combinado. Ejercicio supervisado. Recomendaciones de actividad física.
Abbreviations
ACSM: American College of Sport Medicine.
BMI: Body mass index.
CF: Cardiovascular fitness.
DEE: Daily energy expenditure.
DXA: Dual-energy x-ray absorptiometry.
E: Endurance training group.
HDL: High density lipoprotein.
HPA: Habitual physical activity.
HR: Heart rate.
HRR: Heart rate reserve.
HULP: University Hospital La Paz.
ICCr: Intraclass correlation coefficient.
LDL: Low density lipoprotein.
PA: Diet and physical activity recommendations group.
PRONAF: Programas de Nutrición y Actividad Física para el tratamiento del sobrepeso y la obesidad.
RM: Repetition maximum.
RPE: Rate of perceived exertion.
S: Strength training group.
SE: Combine training group.
TC: Total cholesterol.
TG: Triglycerides.
VO2: Oxygen uptake.
Introduction
Dyslipidemia is an important comorbidity of obesity associated with a very high incidence of coronary and vascular events.1 Weight loss achieved with diet or exercise has shown a reduction of triglycerides (TG) levels and elevation of high density lipoprotein-cholesterol (HDL) levels.2 These studies concluded that reduction of fat through diet or exercise produces comparable and favorable changes in plasma lipoprotein concentrations.2,3
Prospective epidemiological studies have proved a close link between the lipoprotein profile and cardiovascular morbidity and mortality. Epidemiological evidence suggest that physically active individuals have a 30-50% lower risk of developing type 2 diabetes or cardiovascular disease (CVD) than do sedentary persons. Moreover habitual physical activity (HPA) confers a similar risk reduction for coronary heart disease.4 A Study from Ekblom-Bak et al. (2010) showed that both PA and cardiovascular fitness (CF) are independently associated with lower cardiovascular risk, suggesting that both variables should be modified to improve cardiometabolic health.5
Physical exercise without diet restriction or weight loss has evidenced improvements in blood lipids profiles and to decrease fat mass.6 It is unknown which is the most efficient mode of exercise to improve the response on lipid profile. High intensity strength training showed evidence that improved blood lipid profile.7,8 Endurance tranining also have shown improvements on blood lipid profile without diet restriction.9 We hypothesize that the combination of both modes of exercise can benefit synergists. There are studies that examine the effects of different exercise modes (endurance training, resistance training and both combination) on blood lipid levels in previously sedentary adults engaging exercise intervention.10,11 Findings about effects of different exercise mode on blood lipids did not differ. Many studies have been criticized for methodological flaws or design limitations that make the results somewhat questionable.12 These flaws have included the lack of separate control group and no dietary control and the results from different exercise types have been conflicting. From an ethical point of view, it is important to consider that the health of patients is at stake. This makes necessary to include in a randomized control trial a group that receives the same treatment they may achieve at hospital units. Therefore we propose a control group design following habitual clinical practice guidelines, which includes diet restriction and general physical activity recommendations, looking after lifestyle changes. Although hypocaloric diet alone generally achieves beneficial results, regular exercise add salutary effects and therefore both strategies support counseling by health practitioners.13 Unfortunately, patients tend to dropout from healthcare weight-loss programs relatively quickly, and a continued compliance is a matter of concern in these strategies.14
Therefore our study evaluates the impact of different supervised and well controlled exercise modes with diet restriction on lipid profile. A secondary objective was to check if different structured protocols of exercise combined with diet versus physical activity recommendations with diet could be more effective in improving lipid profile in obese men and women.
Material and methods
Participants
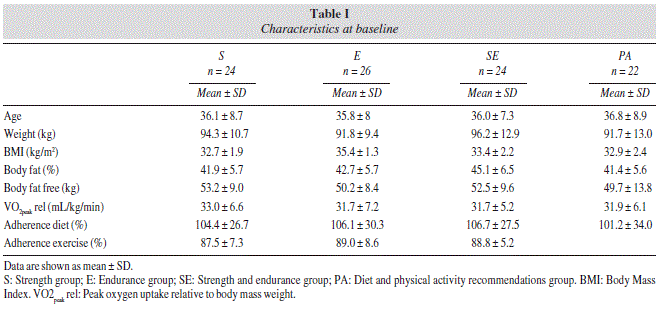
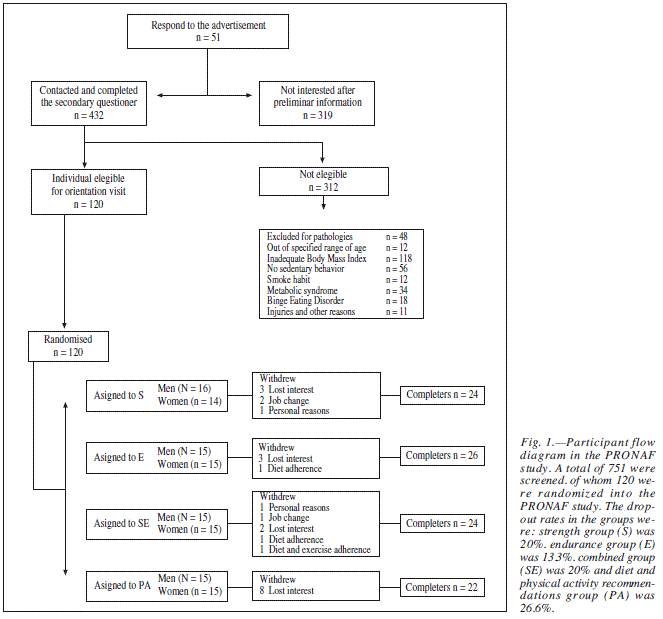
This study was performed as part of the larger study Nutrition and Physical Activity for Obesity (the PRONAF study according to its Spanish initials), the aim of which was to assess the usefulness of different types of physical activity and nutrition programs for the treatment of obesity. Participants were sought via advertisements in newspapers and on the radio, internet and TV. The eligible sample population consisted of 120 (59 women and 61 men) obese subjects (body mass index [BMI] 30-34.9 kg/m2), all middle-aged (range 18-50 years), living in the Region of Madrid, Spain. Characteristics of the participants are summarized in table I. Figure 1 shows the flow diagram of the PRONAF study. All subjects were healthy, normoglycaemic, non-smokers, but led sedentary lifestyles. All female subjects had regular menstrual cycles. The exclusion criteria covered all physical and psychological diseases that may have precluded the performance of the requested strength or endurance training, and the taking of any medication known to influence physical performance or the interpretation of the results. Subjects with a background of systematic strength or endurance training (moderate to high intensity training more than once a week) in the year before the study started were also excluded. In agreement with the guidelines of the Declaration of Helsinki regarding research on human subjects, all participants signed an institutionally approved document of informed consent. All subjects were carefully informed about the possible risks and benefits of the study, which was approved by the Human Research Review Committee of the La Paz University Hospital (HULP) (PI-643).
Study design
Subjects who fulfilled the inclusion criteria and passed a baseline physical examination were stratified by age and sex and randomly assigned to a strength training group (S), endurance training group (E), combined strength + endurance training group (SE) or physical activity recommendations group (PA), according to a randomisation table.
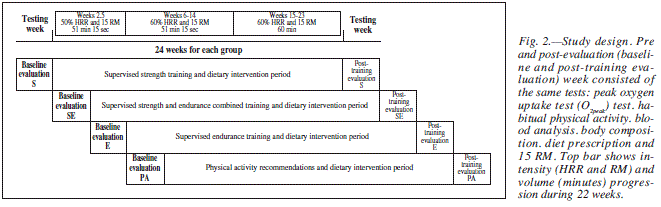
This study was an intervention trial of 24 week duration. The measurements took place in the first week (baseline values) for all subjects before starting training, and after 22 weeks of training, in week 24 (post-training values). Once the first group started the pre-evaluation week, each group started sequentially (fig. 2) maintaining the same periodization.
Exercise training program
The different exercise groups followed the corresponding, supervised training program, which consisted in all cases of training 3 times/wk for 22 weeks. All training sessions were carefully supervised by certified personal trainers. An adherence to training of 90% was demanded. The exercise programs were designed taking into account each subject's muscle strength (MS) and the heart rate reserve (HRR). MS was measured using the 15-repetition maximum (15 RM) testing method,15 in the S and SE groups (both of which involved strength training). The 15 RM for each exercise in each program was recorded twice on different days during the pre-intervention subject strength evaluation period. The intraclass correlation coefficient of reliability for all exercises was ICCr = 0.995 and ICCr = 0.994 for the men and women respectively (groups S and SE subjects together). All the assessments and trainings were carried out with the same machines and free weights (Johnson Health Tech. Iberica, Matrix, Spain). Heart rate reserve (HRR) was also calculated to prescribe exercise intensity plus resting heart rate for E and SE interventions programs.16,17
The intensity of exercise was increased over the study period. In weeks 2-5 exercise was at an intensity of 50% of the 15 RM and HRR, and lasted an overall 51 min and 15 s (twice around the circuit, lasting 7 min 45 s each lap). In weeks 6-14 exercise was performed at an intensity of 60% of 15 RM and HRR, again with a duration of 51 min and 15 s (again, twice around the circuit). Finally, in weeks 15-23, exercise was performed at an intensity of 60% of 15 RM and HRR, with a duration of 64 minutes (three times around the circuit). The recovery period between circuits was set at 5 min. Participants performed 15 repetitions (45 s) of each exercise with a rest period of 15 seconds between them.
Each training session for the strength, endurance + combined strength and endurance training commenced with a 5 min aerobic warm-up, followed by the main session exercises, and concluded with 5 min of cooling down and stretching exercises. In addition, each session was monitored for heart rate (HR) and Rate of Perceived Exertion (RPE) scale. In all sessions the exercise rhythm was controlled by instructions recorded on a compact disk. The cadence for the resistance exercises was fixed at 1:2 (concentric-eccentric phase).
Feedbacks for training loads were done once a month with the RPE to subjectively evaluate each session and determine where the participant considered the intensity to be at, following a similar methodology as used elsewhere.18
Endurance training group (E). The E training involved the use of a treadmill, exercise bike or cross trainer.
Strength training group (S). The S followed a circuit involving the following eight exercises: shoulder press, squat, barbell row, lateral split, bench press, front split, biceps curl, and french press for triceps.
Strength and endurance training group (SE). The SE performed a combination of cycle ergometry, treadmill or cross trainer work, plus weight training with the following exercises intercalated: squat, row machine, bench press and front split.
Diet and physical activity recommendations group (PA). Control participants followed the habitual hospital clinical practice. This means the same dietary intervention as the training groups plus general recommendations in PA from the American College of Sport Medicine (ACSM),19 without being supervised and regulated, only registered with accelerometer for lifestyle changes control, just as real clinical health practitioners at hospital units.
Hypocaloric diet program
Diet prescription was performed for all patients by expert dieticians in the Nutrition Department of HULP. All groups underwent an individualized and hypocaloric diet (between 1,200 and 3,000 kcal). Diet was lowered a 25% from daily energy expenditure (DEE)20 measured using SenseWear Pro ArmbandTM data. Macronutrient distribution consisted of 29-34% of energy from fat, 12-18% from protein, and 50-55% from carbohydrates, according to recommendations.21 A dietitian interviewed each participant at baseline, 3 months, and 6 months and reviewed a 3-day food record diary. All subjects were instructed how to record their dietary intake using a daily log, and given recommended portion sizes and information on possible food swaps. In addition, voluntary group nutrition education sessions were given by the dieticians. The goal was to equip the participants with knowledge and skills necessary to achieve gradual, permanent behavioural changes. An adherence to diet of 90% was elicited and was calculated with 72-hour recall.22
Data collection
The following analyses and measurements were made at baseline and at the end of the study period:
- Blood analysis: All blood samples were taken after 12 h fast between 7:00 and 9:00 a.m. at baseline and post-training intervention (week 1 and week 24). All post-training samples were obtained 72 hours after the last training day to avoid acute effects of training on blood lipids. All blood samples were drawn from the antecubital vein and handled according to standardized laboratory practice at HULP.
Blood lipids and lipoprotein. Serum biochemicals (total cholesterol (TC), low-density lipoprotein (LDL) cholesterol, high-density lipoprotein (HDL) cholesterol, and triglycerides (TG) were determined using enzymatic methods with Olympus reagents by automated spectrophotometry performed on Olympus AU 5400 (Olympus Diagnostica, Hamburg, Germany). Menstrual cycle was controlled by diary to define the follicular and luteal phases when blood samples were taken.23
- Physical fitness variables: Peak oxygen uptake test (VO2peak) was measured using the modified Bruce protocol used elsewhere with overweight and obese population.24,25 The test was conducted on an H/P/ COSMOS 3P 4.0 computerised treadmill (H/P/Cosmos Sports & Medical, Nussdorf-Traunstein, Germany). The volume and composition of expired gas measure were measured using a Jaeger Oxycon Pro gas analyser (Erich Jaeger, Viasys Healthcare, Germany) and continuous 12-lead electrocardiographic monitoring. The exercise test was maintained until exhaustion. VO2peak.was taken to be the mean of the three largest measurements. The dynamometric strength index (DSI) was determined by measuring muscular strength using a Tecsymp Tkk5002 hand and leg dynamometer (Tecsymp, Barcelona, Spain) and a Tecsymp Tkk5401 back dynamometer (Tecsymp, Barcelona, Spain). The DSI value was calculated as the sum of the values obtained with both apparatuses divided by subject body weight.
- Habitual physical activity: Habitual physical activity (PA) was assessed with a SenseWear Pro3 Armband® (Body Media, Pittsburgh) previously validated.26,27 This device is worn on the right upper arm over the triceps muscle and monitors various physiological and movement parameters. Information provided by the manufacturer (www.bodymedia.com) indicates that the accelerometer uses non-invasive biometric sensors to continuously measure physical parameters (heat flux, galvanic skin response, skin temperature, near-body temperature, and two-axis accelerometry) and demographic characteristics (gender, age, height, weight) to estimate energy expenditure utilizing proprietary equations. Daily energy expenditure (DEE) was calculated using the propriety algorithm (Innerview Research Software Version 6.0). Subjects were instructed to wear the monitor continuously for 5 days including weekend days and weekdays following general recommendations28 at baseline and post-training intervention. Data was recorded by 15 min intervals. All subjects were instructed to continue their habitual daily activities as before and were provided with a PA diary to log the type, duration, and intensity of any PA or exercise undertaken during intervention.
- Body composition. Body composition was assessed by dual-energy x-ray absorptiometry DXA (GE Lunar Prodigy; GE Healthcare, Madison, WI, GE Encore 2002, version 6.10.029 software) and was used to measure total body fat (%) and body fat free (kg) mass.
Anthropometric measures included height (stadiometer SECA; range 80-200cm), body mass (BC-420MA. Bio Lógica. Tecnología Médica SL) and body mass index (BMI) calculated as [body weight (kg)/(height (m))2].
- Dietary assessment: All food and beverages consumed by the participants were recorded using a food frequency questionnaire and a "3-day food and drink record", validated for the Spanish population,29 at the beginning and end of the intervention. Participants were instructed to record the weights of food consumed, if possible, and to use household measurements (tablespoons, cups, etc.) when not. The energy and nutritional content of the foods consumed were then calculated using DIAL software (Alce Ingeniería, 2004).
Statistical analysis
SPSS version 15.0 for Windows was used for statistical analyses (SPSS Inc., Chicago, Illinois, USA). Standard statistical methods were used for the calculation of the means and standard deviation. Two way analysis of variance (ANOVA) (group x measurement [baseline-post]) for repeated measures was used to determine any differences between the four groups and differences in baseline values and post-training values in each group assessed. Bonferroni's post-hoc test was employed to locate specific differences. The delta percentage was calculated through the standard formula: change (%) = [(post-test score-pre-test score)/ pre-test score] x 100. The effect of menstrual cycle on lipid profile was assessed by impaired T-test. The effect of ApoE on lipid profile was assessed by univariate analysis of variance (ANOVA). The significance level was set at α = 0.05.
Results
Baseline characteristics
As observed in figure 1, final completers were n = 96 (48 women and 48 men). Adherence criteria for diet and exercise were also took into account to determine final analyzed completers (table I). Baseline characteristics of the participants revealed no significant differences for weight, percentage body fat, body fat free mass and O2peak rel.
Body composition and blood lipids and lipoproteins
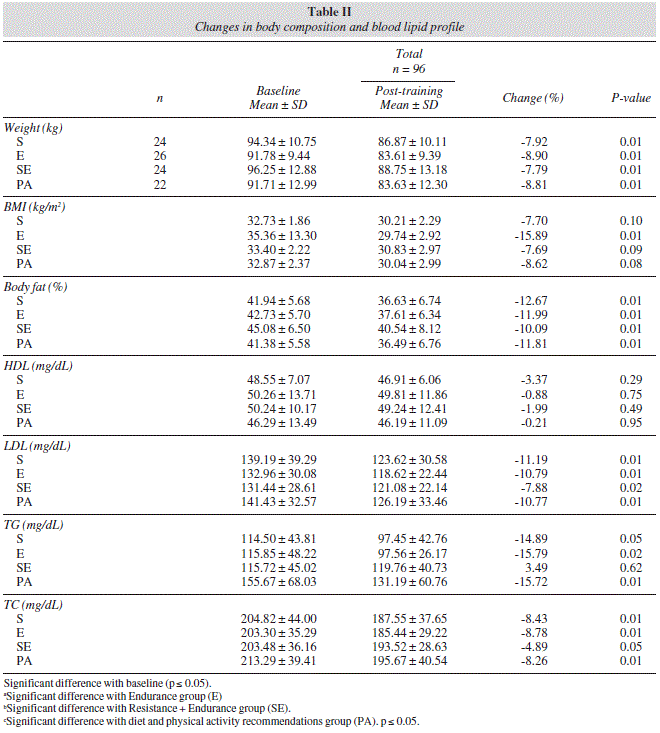
Table II shows changes in body composition and plasma lipid and lipoprotein concentrations in four groups before and after the intervention period (training and diet). There were no statistically significant differences between groups for post-training values.
Body weight decreased between 7.92% and 8.90%. This was accompanied by a reduction of body fat between 10.09% to 12.67% (table II). BMI decreased significantly in E group and showed a trend towards a significant reduction in the rest of groups (table II).
There were no significant changes to HDL levels after intervention. For LDL values a significant decrease was observed for all groups (S: 11.2%, p < 0.01; E: 10.8%, p < 0.01; SE: 7.9%, p < 0.05; PA: 10.8%, p < 0.01). S, E and PA showed a statistically significant decrease in TG (S: 14.9%, E: 15.8%, PA: 15.7%, p < 0.05). TC decreased significantly for all groups (S: 8.4%, p < 0.01; E: 8.8%, p < 0.01; SE: 4.9%, p < 0.05, PA: 8.3%, p < 0.01). The effects size was calculated to check the exercise mode effect in the intervention. The effects size to lipid profile variables was: HDL: 0.006, LDL: 0.010, TG: 0.067, CT: 0.019.
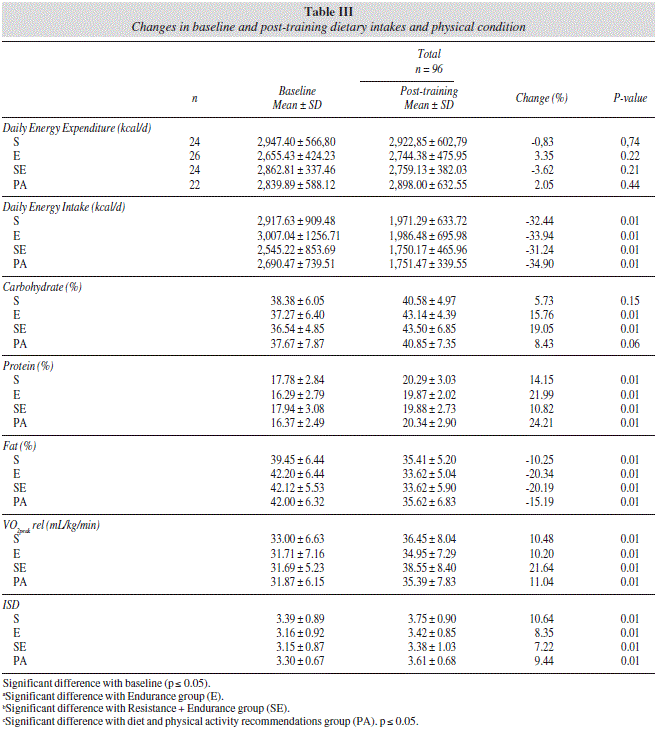
Dietary analyses
A summary of macronutrient and energy intakes at baseline and at week 24 is shown in table III. All groups significantly reduced their energy intake: S group -946 ± 716, E group -1,220 ± 1,149, SE group -795 ± 853, PA group: 939 ± 748 kcal, with no significant differences between groups. Statistical analysis of daily nutrient intake for each of the four groups revealed no significant differences in carbohydrate, protein and lipid percentage at baseline. After the intervention period, none of the macronutrient showed differences between groups. There were no differences between groups neither at baseline nor at post-training for daily energy expenditure.
Physical condition
VO2peak significantly increased in all group: S group 3.4 ± 3T, E group 3.2 ± 5.5, SE group 6.8 ± 5.5, PA group: 3.5 ± 4.1 mL/kg/min (p < 0.01). DSI increased also in four groups (S: 10.6%; E: 8.3%; SE: 7.2%; PA: 9.4%; p < 0.01).
Confounding variables
There were no differences in lipid profile values between luteal and non-luteal (follicular) phase at baseline and post-training measurements (data not shown). Regarding ApoE groups, there were no differences in serum lipids and lipoprotein concentrations at baseline (data not shown).
Discussion
The main finding of the present study was that structured exercise programs and physical activity recommended program with hypocaloric diet are effective as treatment to improve the blood lipid profile. All participants engaged in the program showed greater reductions in LDL, TG and TC, with no differences seen among these groups.
The treatment or strategies used to improve the lipid profile have attempted to encourage improvements in the cardiometabolic health, the literature suggests that healthy diet, weight loss, exercise and physical activity are key to prevent and treat the development of these diseases.30 As in our intervention, diet restriction achieves a weight loss with fat mass loss that improves lipid function.31
In the present work, no significance changes were observed in HDL. High density lipoproteins has been reported to increase,32 decrease,33,34 or remain stable35 with weight loss. The discrepancy in results seems to be due to the divergent effects of weight loss. Several studies show that reduce fat intake in diet results in a decrease in HDL, even when weight loss occurs in both short36 and long-term37 studies. The work of Pelkman et al. (2004) reported that weight loss HDL decrease and when weight maintenance HDL may increase.31 This study add to increase HDL is necessary a moderate fat intake.31 Clinical trial to evaluate effects on lipid profile with exercise intervention show modest or no changes to HDL.10,38,39 Our results are in agreement with these studies. Therefore, no change in HDL may be due to reduction in fat intake.
In our study, LDL decrease significantly for all groups without differences between groups. After the 22 weeks of intervention, LDL values achieved decrease to values considered no atherogenic. When exercise is accompanied by a loss body fat, LDL decrease.40 The work of Pronk et al. (1995) and Greene et al. (2012) reported a decrease in LDL concentration after acute exercise.41,42 Improvements in lipid profile due to physical training may be dependent on loss on body fat.43 However, Hurley et al. (1988) reported reductions in concentrations LDL that were independent of changes in body composition.7
The work of Kelley (2009) on the effects of exercise on lipoprotein concentrations seen with changes in body mass has reported that reductions in lipoprotein-lipid concentrations occurred more frequently when exercise was combined with body fat loss but could occur without change in body mass.44 Therefore, in agreement with our results, studies prove that a combined exercise with diet program demonstrated higher efficacy on LDL levels.45
The results of the present study exhibit a favorable response of TG levels in all groups except SE. After intervention S, E and PA groups decreased significantly TG concentrations. SE group shows no change maintaining healthy values to TG. PA group obtains a decrease in TG to healthy values. Many studies show in their results the favorable response of TG concentration with exercise program.6,9,46,47 Regular exercise is known to increase amounts of lipoprotein lipase (LPL) in adipose and muscle tissue. Diet restriction has also shown good treatment to decrease TG concentrations2,48 Andersen et al. (1995) confirm previous findings that weight loss is associated with significant improvements in serum lipids and lipoproteins.2,49 Thus, an 11% reduction in body weight achieved a 22.7% reduction in TG.50 These results are agreement with our study where the participants obtain an average reduction of 10.7% in TG concentrations with an 8.3% of weight loss. Reviewing studies that compared weight loss achieved with diet or with exercise, Wood et al. (1991) observed that fat mass loss get significant reductions in TG.2
In our study, after intervention, TC levels were reduced in all groups up to references values. Studies with similar protocols to our study49,50 found no differences between groups, but also achieved significant changes in all groups. Previous studies have shown that plasma TC levels were directly related to total fat intake.51 Therefore, reduction in blood TC seems to be attributable to a great manner to dietary advice due to improvements in fat intake.52 Although there are works where showed improvements in TC with an exercise program without diet restriction during 8 weeks, therefore also exercise program alone can have a positive impact on the TC.47 However, the works of Lemura et al. (2000), Sillanpaa et al. (2009) and Stensvold (2010) when compared the effects on lipid profile and syndrome metabolic variables of different exercise modes were no found differences between groups after exercise intervention.9,10,38 In our study, fat intake is reduce 15% average. This decrease results in a significant improvement to TC in all participants.
When exercise is combined with diet restriction studies report greater improvements in the plasma lipid profile in response to the combination of diet and exercise than diet alone.49,53,54 It is also reported that the addition of exercise with diet restriction does not obtain significant improves.45,49,50 These observations reflect that lipid profile improvements may be dependent of fat mass loss. Therefore it is important to give of clearly establishing an independent role for exercise in the treatment of obesity and related comorbidities. In our study not found additional improvements on lipid profile when added different modes of exercise in agreement with the results of previous studies.45,49,50 We also assumed that, as other studies suggested previously,55 supervised training protocols may have not achieved enough intensity in order to obtain significant improvements versus diet and unsupervised regular physical activity recommendations, since the risk of injury in this population did not allow non-progressive increases in intensity.
Findings from accelerometer-measured daily HPA indicated that there were not significant changes in any group in their daily HPA (non-training activity) after 6 months of intervention. No differences between groups were found, including training sessions (data not shown). Even though PA group may have tried to engage in different activities following the ACSM recommendations received, it was not enough to increase their habitual physical activity significantly. On the other hand, training groups did not result in a more active lifestyle outside training intervention. As the flow diagram shows, the PA group showed up with the highest dropouts percentage (26.6%). Recent studies try to investigate predictive variables for weight loss programs abandons, meaning that is a big matter of concern.14 Our results showed that supervised exercise did not obtain any additive effects to diet restriction and physical activity recommendations on lipid profile, but it seems that was helpful in sustaining adherence in order to finish the intervention program. Hospital units tend to supervise with often feedback the dietary modifications, but poor counseling in the exercise recommendation is done.
A point of interest of the present study is that include the randomized-controlled design, the long supervised training period and the lifestyle. PRonaf study to include a group that follows the principles of hospital clinical practice for lifestyle changes (diet and physical activity recommendations) when treating patients for weight loss management.
To maintain the training principle of progression and adaptation was essential in the design of our study due to the population target, in order to avoid injuries and abandons during the intervention. This may have turned into a limitation because we could not achieve a higher intensity, probably needed to obtain further improvements through exercise.
Conclusion
In conclusion, the present results show that strategies combining supervised physical exercise or physical recomendation and a hypocaloric diet can provide benefits in terms of body composition and improvements on lipid profile. This study show that an intervention program of endurance, strength or combined supervised training protocol with diet restriction did not achieved further improvements on lipid profile than diet restriction and usual physical activity recommendations developed in clinical practice in obese men and women. Future research is required in order to investigate if higher intensity of any supervised training protocol mode can add improvements to dietary modification.
Acknowledgements
We acknowledge and thank all of the subjects who participated in the study. We thank PhD Víctor Díaz Molina for valuable discussion.
Conflict of interest statement
The authors have no conflicts of interest.
References
1. NHLBI. Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III): ATP III Update 2004: Implications of Recent Clinical Trials for the ATP III Guidelines 2004. [ Links ]
2. Wood PD, Stefanick ML, Dreon DM, Frey-Hewitt B, Garay SC, Williams PT, Superko HR, Fortmann SP, Albers JJ, Vranizan KM. Changes in plasma lipids and lipoproteins in overweight men during weight loss through dieting as compared with exercise. New England Journal of Medicine 1988; 319: 1173-9. [ Links ]
3. Rossner S, Bjorvell H. Early and late effects of weight loss on lipoprotein metabolism in severe obesity. Atherosclerosis 1987; 64: 125-30. [ Links ]
4. Bassuk SS, Manson JAE. Epidemiological evidence for the role of physical activity in reducing risk of type 2 diabetes and cardiovascular disease. Journal of Applied Physiology 2005; 99: 1193-204. [ Links ]
5. Ekblom-Bak E, Hellenius ML, Ekblom Ö, Engstrom LM, Ekblom B. Independent associations of physical activity and cardiovascular fitness with cardiovascular risk in adults. European Journal of Cardiovascular Prevention & Rehabilitation 2010; 17: 175-80. [ Links ]
6. Park SK, Park JH, Kwon YC, Kim HS, Yoon MS, Park HT. The effect of combined aerobic and resistance exercise training on abdominal fat in obese middle-aged women. J Physiol Anthropol Appl Human Sci 2003; 22: 129-35. [ Links ]
7. Hurley BF, Hagberg JM, Goldberg AP, Seals DR, Ehsani AA, Brennan RE, Holloszy JO. Resistive training can reduce coronary risk factors without altering VO2max or percent body fat. Med Sci Sports Exerc 1988; 20: 150-4. [ Links ]
8. Joseph LJ, Davey SL, Evans WJ, Campbell WW. Differential effect of resistance training on the body composition and lipoprotein-lipid profile in older men and women. Metabolism 1999; 48: 1474-80. [ Links ]
9. LeMura LM, von Duvillard SP, Andreacci J, Klebez JM, Chelland SA, Russo J. Lipid and lipoprotein profiles, cardiovascular fitness, body composition, and diet during and after resistance, aerobic and combination training in young women. Eur J Appl Physiol 2000; 82: 451-8. [ Links ]
10. Stensvold D, Tjonna AE, Skaug EA, Aspenes S, Stolen T, Wisloff U, Slordahl SA. Strength training versus aerobic interval training to modify risk factors of the metabolic syndrome. J Appl Physiol 2010. [ Links ]
11. Boardley D, Fahlman M, Topp R, Morgan AL, McNevin N. The impact of exercise training on blood lipids in older adults. The American journal of geriatric cardiology 2007; 16: 30-5. [ Links ]
12. Tambalis KD, Panagiotakos DB, Kavouras SA, Sidossis LS. Responses of Blood Lipids to Aerobic, Resistance, and Combined Aerobic With Resistance Exercise Training: A Systematic Review of Current Evidence. Angiology 2009; 60: 614-32. [ Links ]
13. Nawaz H, Katz DL. American College of Preventive Medicine practice policy statement: weight management counseling of overweight adults. American Journal of Preventive Medicine 2001; 21: 73-8. [ Links ]
14. Bautista-Castano I, Molina-Cabrillana J, Montoya-Alonso JA, Serra-Majem L. Variables predictive of adherence to diet and physical activity recommendations in the treatment of obesity and overweight, in a group of Spanish subjects. Int J Obes Relat Metab Disord 2004; 28: 697-705. [ Links ]
15. Morgan B, Woodruff SJ, Tiidus PM. Aerobic energy expenditure during recreational weight training in females y males. J Sports Sci & Med (electronic journal) 2003; 2: 117-22. [ Links ]
16. Da Cunha FA, Farinatti Pde T, Midgley AW. Methodological and practical application issues in exercise prescription using the heart rate reserve and oxygen uptake reserve methods. J Sci Med Sport 2011; 14: 46-57. [ Links ]
17. Karvonen MJ, Kentala E, Mustala O. The effects of training on heart rate; a longitudinal study. Ann Med Exp Biol Fenn 1957; 35: 307-15. [ Links ]
18. Ball Geoff D, Crespo Noe C, Cruz Martha L, Goran Michael I, Salem George J, Shaibi Gabriel Q, Weigensberg Marc J. Effects of Resistance Training on Insulin Sensitivity in Overweight Latino Adolescent Males. Medicine & Science in Sports & Exercise 2006; 38: 1208-15. [ Links ]
19. Donnelly JE, Blair SN, Jakicic JM, Manore MM, Rankin JW, Smith BK. American College of Sports Medicine Position Stand. Appropriate physical activity intervention strategies for weight loss and prevention of weight regain for adults. Med Sci Sports Exerc 2009; 41: 459-71. [ Links ]
20. National Institutes of Health. Clinical guidelines on the identification, evaluation, and treatment of overweight and obesity in adults: executive summary. Expert Panel on the Identification, Evaluation, and Treatment of Overweight in Adults. The American journal of clinical nutrition 1998; 68: 899-917. [ Links ]
21. Dapcich V, Salvador Castell G, Ribas Barba L, Pérez Rodrigo C, Aranceta Bartrina J. Guías alimentarias (Dietary guidelines, Food Guides). Representations 2002; 102: 483-9. [ Links ]
22. Schroder H, Covas MI, Marrugat J, Vila J, Pena A, Alcantara M, Masia R. Use of a three-day estimated food record, a 72-hour recall and a food-frequency questionnaire for dietary assessment in a Mediterranean Spanish population. Clinical Nutrition 2001; 20: 429-37. [ Links ]
23. Muesing RA, Forman MR, Graubard BI, Beecher GR, Lanza E, McAdam PA, Campbell WS, Olson BR. Cyclic changes in lipoprotein and apolipoprotein levels during the menstrual cycle in healthy premenopausal women on a controlled diet. J Clin Endocrinol Metab 1996; 81: 3599-603. [ Links ]
24. Hunter GR, Byrne NM, Sirikul B, Fernández JR, Zuckerman PA, Darnell BE, Gower BA. Resistance training conserves fat-free mass and resting energy expenditure following weight loss. Obesity 2008; 16: 1045-51. [ Links ]
25. Ghroubi S, Elleuch H, Chikh T, Kaffel N, Abid M, Elleuch M. Physical training combined with dietary measures in the treatment of adult obesity. A comparison of two protocols. Annals of Physical and Rehabilitation Medicine 2009; 52: 394-413. [ Links ]
26. Malavolti M, Pietrobelli A, Dugoni M, Poli M, Romagnoli E, De Cristofaro P, Battistini NC. A new device for measuring resting energy expenditure (REE) in healthy subjects. Nutr Metab Cardiovasc Dis 2007; 17: 338-43. [ Links ]
27. Papazoglou D, Augello G, Tagliaferri M, Savia G, Marzullo P, Maltezos E, Liuzzi A. Evaluation of a multisensor armband in estimating energy expenditure in obese individuals. Obesity (Silver Spring) 2006; 14: 2217-23. [ Links ]
28. Murphy SL. Review of physical activity measurement using accelerometers in older adults: considerations for research design and conduct. Prev Med 2009; 48: 108-14. [ Links ]
29. Ortega RM RA, López-Sobaler AM. Questionniares for dietetic studies and the assessment of nutritional status. Madrid; 2003. [ Links ]
30. Churilla JR, Ph.D., M.P.H., RCEP. The metabolic syndrome: The Crucial Role of Exercise Prescription and Diet. ACSM'S Health & Fitness Journal 2009; 13: 20-6. [ Links ]
31. Pelkman CL, Fishell VK, Maddox DH, Pearson TA, Mauger DT, Kris-Etherton PM. Effects of moderate-fat (from monounsaturated fat) and low-fat weight-loss diets on the serum lipid profile in overweight and obese men and women. The American journal of clinical nutrition 2004; 79: 204-12. [ Links ]
32. Hughes TA, Gwynne JT, Switzer BR, Herbst C, White G. Effects of caloric restriction and weight loss on glycemic control, insulin release and resistance, and atherosclerotic risk in obese patients with type II diabetes mellitus. The American Journal of Medicine 1984; 77: 7-17. [ Links ]
33. Wing RR, Jeffery RW, Burton LR, Thorson C, Kuller LH, Folsom AR. Change in waist-hip ratio with weight loss and its association with change in cardiovascular risk factors. The American Journal of Clinical Nutrition 1992; 55: 1086-92. [ Links ]
34. Fujioka S, Matsuzawa Y, Tokunaga K, Kawamoto T, Kobatake T, Keno Y, Kotani K, Yoshida S, Tarui S. Improvement of glucose and lipid metabolism associated with selective reduction of intra-abdominal visceral fat in premenopausal women with visceral fat obesity. International Journal of Obesity 1991; 15: 853. [ Links ]
35. Richter WO, Schwandt P. Benefit from hypocaloric diet in obese men depends on the extent of weight-loss regarding cholesterol, and on a simultaneous change in body fat distribution regarding insulin sensitivity and glucose tolerance. Metabolism 1992;41:1035-9. [ Links ]
36. Noakes M, Clifton PM. Changes in plasma lipids and other cardiovascular risk factors during 3 energy-restricted diets differing in total fat and fatty acid composition. The American Journal of Clinical Nutrition 2000; 71: 706-12. [ Links ]
37. Kasim-Karakas SE, Almario RU, Mueller WM, Peerson J. Changes in plasma lipoproteins during low-fat, high-carbohydrate diets: effects of energy intake. The American journal of Clinical Nutrition 2000; 71: 1439-47. [ Links ]
38. Sillanpää E, Häkkinen A, Punnonen K, Häkkinen K, Laaksonen D. Effects of strength and endurance training on metabolic risk factors in healthy 40-65-year-old men. Scandinavian Journal of Medicine & Science in Sports 2009; 19: 885-95. [ Links ]
39. Sillanpää E, Laaksonen DE, Häkkinen A, Karavirta L, Jensen B, Kraemer WJ, Nyman K, Häkkinen K. Body composition, fitness, and metabolic health during strength and endurance training and their combination in middle-aged and older women. European Journal of Applied Physiology 2009; 106: 285-96. [ Links ]
40. Grandjean PW, Crouse SF, Rohack JJ. Influence of cholesterol status on blood lipid and lipoprotein enzyme responses to aerobic exercise. Journal of Applied Physiology 2000; 89: 472-80. [ Links ]
41. Pronk N, Crouse S, O´Brien B, Rohack J. Acute effects of walking on serum lipids and lipoproteins in women. Journal of Sports Medicine and Physical Fitness 1995; 35: 50-8. [ Links ]
42. Greene NP, Martin SE, Crouse SF. Acute Exercise and Training Alter Blood Lipid and Lipoprotein Profiles Differently in Overweight and Obese Men and Women. Obesity 2012. [ Links ]
43. Leon AS, Sánchez OA. Response of blood lipids to exercise training alone or combined with dietary intervention. Med Sci Sports Exerc 2001; 33: S502-15; discussion S28-9. [ Links ]
44. Kelley GA, Kelley KS. Impact of progressive resistance training on lipids and lipoproteins in adults: a meta-analysis of randomized controlled trials. Preventive Medicine 2009; 48: 9-19. [ Links ]
45. Janssen I, Fortier A, Hudson R, Ross R. Effects of an energy-restrictive diet with or without exercise on abdominal fat, intermuscular fat, and metabolic risk factors in obese women. Diabetes Care 2002; 25: 431-8. [ Links ]
46. Sigal RJ, Kenny GP, Boule NG, et al. Effects of aerobic training, resistance training, or both on glycemic control in type 2 diabetes: a randomized trial. Ann Intern Med 2007; 147: 357-69. [ Links ]
47. Arora E, Shenoy S, Sandhu JS. Effects of resistance training on metabolic profi le of adults with type 2 diabetes. Indian J Med Res 2009; 129: 515-9. [ Links ]
48. Markovic TP, Campbell LV, Balasubramanian S, Jenkins AB, Fleury AC, Simons LA, Chisholm DJ. Beneficial effect on average lipid levels from energy restriction and fat loss in obese individuals with or without type 2 diabetes. Diabetes Care 1998; 21: 695-700. [ Links ]
49. Wood PD, Stefanick ML, Williams PT, Haskell WL. The effects on plasma lipoproteins of a prudent weight-reducing diet, with or without exercise, in overweight men and women. N Engl J Med 1991; 325: 461-6. [ Links ]
50. Andersen RE, Wadden TA, Bartlett SJ, Vogt RA, Weinstock RS. Relation of weight loss to changes in serum lipids and lipoproteins in obese women. Am J Clin Nutr 1995; 62: 350-7. [ Links ]
51. Millen BE, Franz MM, Quatromoni PA, Gagnon DR, Sonnenberg LM, Ordovas JM, Wilson PW, Schaefer EJ, Cupples LA. Diet and plasma lipids in women. I. Macronutrients and plasma total and low-density lipoprotein cholesterol in women: the Framingham nutrition studies. J Clin Epidemiol 1996; 49: 657-63. [ Links ]
52. Tang JL, Armitage JM, Lancaster T, Silagy CA, Fowler GH, Neil HA. Systematic review of dietary intervention trials to lower blood total cholesterol in free-living subjects. BMJ 1998; 316: 1213-20. [ Links ]
53. Nieman DC, Haig JL, Fairchild KS, De Guia ED, Dizon GP, Register U. Reducing-diet and exercise-training effects on serum lipids and lipoproteins in mildly obese women. The American Journal of Clinical Nutrition 1990; 52: 640-5. [ Links ]
54. Stefanick ML, Mackey S, Sheehan M, Ellsworth N, Haskell WL, Wood PD. Effects of diet and exercise in men and post-menopausal women with low levels of HDL cholesterol and high levels of LDL cholesterol. New England Journal of Medicine 1998; 339: 12-20. [ Links ]
55. Kraus WE, Houmard JA, Duscha BD, et al. Effects of the amount and intensity of exercise on plasma lipoproteins. N Engl J Med 2002; 347: 1483-92. [ Links ]
![]() Correspondence:
Correspondence:
Blanca Romero Moraleda.
Department of Health and Human Performance.
Technical University of Madrid.
C/ Martín Fierro, 7.
28040 Madrid, Spain.
E-mail: blanca.romero.moraleda@upm.es
Recibido: 29-X-2012.
1.a Revisión: 19-XI-2012.
Aceptado: 29-XI-2012.