My SciELO
Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Nutrición Hospitalaria
On-line version ISSN 1699-5198Print version ISSN 0212-1611
Nutr. Hosp. vol.29 n.1 Madrid Jan. 2014
https://dx.doi.org/10.3305/nh.2014.29.1.7068
ORIGINAL / Síndrome metabólico-Diabetes
Hypocaloric high-protein diet improves clinical and biochemical markers in patients with nonalcoholic fatty liver disease (NAFLD)
La dieta hipocalórica rica en proteínas mejora los marcadores clínicos y bioquímicos en pacientes con hepatopatía grasa no alcohólica (HPGNA)
Sebastião Mauro Bezerra Duarte1, Joel Faintuch2, José Tadeu Stefano1, Maria Beatriz Sobral de Oliveira1, Daniel Ferraz de Campos Mazo1, Fabiola Rabelo1, Denise Vanni1, Monize Aydar Nogueira1, Flair José Carrilho1 and Claudia Pinto Marques Souza de Oliveira1
1 University of São Paulo School of Medicine. Department of Gastroenterology. Clinical Division. Hepatology Branch (LIM-07).
2 University of São Paulo School of Medicine. Department of Gastroenterology. Brazil.
ABSTRACT
Objective: To investigate the role of hypocaloric high-protein diet, a prospective clinical study was conducted in NAFLD patients.
Research methods and procedures: Pre-versus post-interventional data were analyzed in 48 stable NAFLD patients (submitted to a hypocaloric high-protein diet during 75 days. Variables included anthropometrics (body mass index/ BMI and waist circumference/WC), whole-body and segmental bioimpedance analysis and biochemical tests. Diet compliance was assessed by interviews every two weeks.
Results: BMI, WC and body fat mass remained relatively stable (-1.3%, -1.8% and -2.5% respectively, no significance). HDL- cholesterol increased (P < 0.05) whereas total, LDL and VLDL cholesterol, triglycerides, aspartate aminotransferase/AST, gamma glutamyltransferase/GGT, alkaline phosphatase/AP, fasting blood glucose and glycated hemoglobin/ HbA1c decreased (P < 0.05). When patients were stratified according to increase (22/48, 45.8%) and decrease (21/48, 43.8%) of BMI, association between weight decrease and liver benefit could be elicited in such circumstances for ALT, AP and AST/ALT ratio. No change could be demonstrated in patients who gained weight. Multivariate assessment confirmed that waist circumference, ferritin, triacylglycerol, and markers of glucose homeostasis were the most relevant associated with liver enzymes.
Discussion: Ours results are consistent with the literature of calorie restriction in the management of NAFLD. Changes in lifestyle and weight loss are recommended for NAFLD patients. European guidelines also support this recommendation.
Conclusion: This is the first study that demonstrated that a high protein, hypocaloric diet were associated with improvement of lipid profile, glucose homeostasis and liver enzymes in NAFLD independent on BMI decrease or body fat mass reduction.
Key words: NAFLD. Hypocaloric diet. Hyperproteic diet. Liver enzymes. Weight loss.
RESUMEN
Objetivo: Para investigar el papel de la dieta hipocalórica rica en proteínas, se realizó un estudio clínico prospectivo en pacientes con HPGNA.
Métodos de investigación y procedimientos: Se analizaron los datos antes y después de la intervención en 48 pacientes con HPGNA estable, sometidos a una dieta hipocalórica y rica en proteínas durante 75 días. Las variables incluían medidas antropométricas (índice de masa corporal (IMC) y circunferencia de la cintura (CC)), análisis de bioimpedancia corporal completa y segmentaria y pruebas bioquímicas. El cumplimiento de la dieta se evaluó mediante entrevistas quincenales.
Resultados: El IMC, la CC y la masa grasa corporal permanecieron relativamente estables (-1,3 %, -1,8 % y -2,5%, respectivamente, sin significación). Las HDL-colesterol aumentaron (P < 0,05) mientras que el colesterol total, las LDL y las VLDL, los triglicéridos, la aspartato aminotransferasa (AST), la gamma glutamiltransferasa (GGT), la fosfatasa alcalina (FA), la glucemia en ayunas y la hemoglobina glucosilada (HbA1c) disminuyeron (P < 0,05). Cuando se estratificó a los pacientes en función del aumento (22/48, 45,8 %) o descenso (21/48, 43,8 %) del IMC, se pudo observar una asociación entre la pérdida de peso y el beneficio hepático reflejado en la ALT, la FA y el cociente AST/ALT. No se pudo demostrar ningún cambio en los pacientes que ganaron peso. La evaluación multivariada confirmó que la circunferencia de la cintura, la ferritina, el triacilglicerol y los marcadores de la homeostasis de la glucosa eran los que más relevantemente se asociaban con las enzimas hepáticas.
Discusión: Nuestros resultados son consistentes con la bibliografía relativa a la restricción calórica en el manejo de la HPGNA. Los cambios en el estilo de vida y la pérdida de peso se recomiendan en los pacientes con HPGNA. Las guías europeas también apoyan esta recomendación.
Conclusión: Éste es el primer estudio que demuestra que una dieta rica en proteínas se asocia con la mejora del perfil lipídico, la homeostasis de la glucosa y las enzimas hepáticas en la HPGNA independientemente del descenso del IMC o la reducción de la masa grasa corporal.
Palabras clave: HPGNA. Dieta hipocalórica. Dieta hiperproteica. Enzimas hepáticas. Pérdida de peso.
Abbreviations
ALT: Alanine aminotransferase.
AP: Alkaline phosphatase.
AST: Aspartate aminotransferase.
BFM: Body fat mass.
BMI: Body mass index.
ECW: Extracellular water.
GGT: Gamma glutamyltransferase.
HbA1c: Glycated hemoglobin A1c.
HCC: Hepatocellular carcinoma.
HDL-C: High-density lipoprotein cholesterol.
HOMA-IR: Homeostasis model assessment - insulin resistance.
LBLL: Segmental left leg lean body mass.
LBRL: Segmental right leg lean body mass.
LBT: Segmental trunk lean body mass.
LDL-C: Low-density lipoprotein cholesterol.
NAFLD: Nonalcoholic fatty liver disease.
NASH: Non-alcoholic steatohepatitis.
TG: Triacylglycerol.
TSH: Thyroid-stimulating hormone.
VLDL: Very low-density lipoprotein cholesterol.
WC: Waist circumference.
Introduction
Non-alcoholic fatty liver disease (NAFLD) is a spectrum of liver diseases associated with the accumulation of fat in the liver that affects individuals without history of alcohol abuse.1 This clinical and pathologic condition could evolve from simple deposits of fat (steatosis) to non-alcoholic steatohepatitis (NASH), possibly followed by cirrhosis and hepatocellular carcinoma (HCC).1,2 NAFLD is currently the most prevalent liver disease, especially in western countries.3 Most patients are obese and diabetic4 and up to 88% may be affected in some circumstances as nutritional factors, use of certain drugs, metabolic causes, infections and exposure to toxins.5
Excessive intake of carbohydrate and fat including soft drinks, fatty diets and cholesterol-rich nutrients has been shown to be responsible both for development of metabolic syndrome6 and accumulation of excess fatty acids in the liver,7 thus representing important predictors of NAFLD.8 The consumption of sucrose-sweetened beverages has been shown to be responsible for increased accumulation of visceral fat. Maersk et al. analyzed for 6 months the consumption of such beverages in comparison with skim milk, diet cola and water, confirming that those drinks increased the risk of cardiovascular and metabolic diseases.9
There is no consensus on the characteristics of a good nutritional strategy for NAFLD,10 despite the emphasis on lifestyle change, which in principle means weight loss in combination with exercise and diet. This approach tends to improve liver enzymes and improves insulin resistance.11 In a recent experience, an 8% reduction in body weight was followed by significant benefit for aminotransferases and lipid deposits liver.12
Studies addressing specific macronutrients showed conflicting results, some groups emphasizing that certain micronutrients as well could be relevant for the development of NAFLD.13,14
Limited evidence suggests that a high protein diet could be effective for the treatment of NAFLD because of increase in energy expenditure and hepatic lipid oxidation, as liver catabolism of ingested amino acids is an energy -intense process.15 There are few studies showing the relationship between protein intake and NAFLD.16 In obese and sedentary women, short-term protein supplementation was advantageous for hepatic steatosis and lipid profile.17 Soy protein has also been successfully employed in this context, however the functional properties of soybeans were the focus, not the nitrogen intake.13
In the current study, short term prescription of a hypocaloric, high protein diet to a NAFLD population was conducted. It was hypothesized that even in the absence of significant weight loss, favorable clinical results would occur as a consequence of the more robust nitrogen input.
Patients and methods
Study design and subjects
This was a prospective clinical study with one population, one intervention (dietary protocol), and two scheduled observation periods (baseline and end of the study). End point was significant reduction of liver enzymes, notably AST, ALT and AST/ALT ratio. Seventy three patients accompanied for at least one year at the Hepatology Outpatient Unit were recruited. Criteria of inclusion were overweight and obese males and females with biopsyproven NAFLD, with or without comorbidities, and written informed consent. Criteria of exclusion were other liver abnormalities such as alcoholic liver disease, viral hepatitis (B, C, D), primary biliary cirrhosis, sclerosing cholangitis, hemochromatosis, Wilson's disease, alpha1-antitrypsin deficiency and drug induced liver disease, consumptive diseases (cancer, tuberculosis, HIV/ AIDS), bariatric surgery, use of pharmacologic agents for obesity or NAFLD, participation in any other trial in the last 6 months, and refusal to participate in the study.
The investigation was approved by the Institutional Ethical Committee. Preliminary screening included hepatobiliary ultrasonography, viral serology, autoantibody titers, serum iron, ferritin and transferrin saturation, ceruloplasmin, copper levels and alpha 1-antitrypsin. Identification of metabolic syndrome components followed the recommendations of the Adult Treatment Panel III Report, namely fasting glucose ≥ 110 mg/dL, triglycerides ≥ 150 mg/dL, high density lipoprotein (HDL) < 40 mg/dL in men or < 50 mg/dL in women; and ≥ 130 mmHg systolic or ≥ 85 mmHg diastolic pressure, as well as abdominal obesity, identified as waist circumference > 102 cm in men and > 88 cm in women. Other comorbidities were characterized according to hospital files or current treatment.
Randomization and stratification
No randomization was adopted in this single -group protocol. In order to further explore the associations between enzymatic changes and BMI, patients were stratified according to BMI in weight-stable (< 0.2% change), weight gaining and weight losing subjects.
Dietary methods
A standard 24 h food recall complemented by a food frequency questionnaire was performed by an experienced dietitian, adjusted for Brazilian foods and portions and for weekend changes, at baseline and study completion. Macronutrients (total energy, protein, carbohydrates, and lipids including saturated, monounsaturated and polyunsaturated fatty acids as well as cholesterol), plus dietary fibers, were calculated with the Avanutri 4.0 software (Avanutri, Rio de Janeiro, Brazil).
Diet composition
Prescribed diet consisted of 1.200 kcal/day for females and 1.400 kcal/day for males during 75 days, corresponding to an energy deficit of approximately 500 kcal from usual food consumption. Fat represented 25% of the total caloric value, with saturated fat 7% (28% of fat), monounsaturated 10% (40% of fat), and polyunsaturated 8% (32% of fat). The proportion of protein (mixed animal and vegetable) was 35%, and carbohydrates made up the remainder (40% of total calories, 50% whole grains, 25 grams of sugar), with 20g fiber/day. The diet was adapted from current highprotein prescriptions, which have been demonstrated as safe and well tolerated for up to one year.19,20
Compliance was monitored by means of individual consultations with an experienced dietitian, every two weeks, checking menu plans, portion sizes, leftover items and unprescribed meals, drinks or snacks. Subjects suspected of poor adherence or giving conflicting answers were excluded from the protocol.
Anthropometrics and body composition
Anthropometric variables represented by weight, height, body mass index/BMI were determined by stadiometer (Biospace model BSM370, GangnamGu, Seoul, Korea) and waist circumference/WC was determined between the iliac crest and the lower costal margin using inelastic plastic tape 0.7 cm wide.21 Body water (total, intracellular water/ICW, extracellular/ ECW), body fat mass/BFM, segmental trunk lean body mass/LBT, segmental right leg lean body mass/LBRL, and segmental left leg lean body mass/LBLL, were measured by bioimpedance analysis (Biospace-model InBody 720®, GangnamGu, Seoul, Korea).
Laboratory assays
Total cholesterol, high-density lipoprotein cholesterol/HDL, low-density lipoprotein cholesterol/LDL, very low-density liprotein cholesterol/VLDL, triacylglycerol/TG), alanine aminotransferase/ALT, aspartate aminotransferase/AST, gamma glutamyltransferase/GGT, fasting blood glucose, Glycated hemoglobin A1c/HbA1c, alkaline phosphatase/AP, total bilirubin, total protein, prothrombin time, ferritin, iron, blood urea, creatinine, sodium, potassium, thyroid stimulating hormone/TSH a and insulin were measured by automated methods in a certified academic laboratory. The homeostasis model assessment-insulin resistance index/ HOMA-IR was calculated as well, according to the equation HOMA-IR = (Fasting Plasma Insulin x Fasting Plasma Glucose)/22.5.22
Sample size calculation and statistical analysis
It was anticipated an effect size of 30% derived from the increase of daily protein intake, aiming for a statistical power of 80% with an alpha error of 0.05. For a net increase of 30% in dietary protein after the intervention, the minimum sample size was 45. Values are expressed as mean ± standard deviation (SD). Student's t test and Wilcoxon test were used according to normality assessment (Kolmogorov-Smirnov). Univariate and multivariate (multiple logistic) regression analysis were selected to address enzymatic changes according to clinical and biochemical patterns. As a large set of variables was compared in this investigation, the Benjamini and Hochberg correction was employed to control for spurious findings. Statistical Analysis Systems, version 9.1.3 (SAS Institute, Cary, NC) was used, and a P-value of less than 0.05 indicated a significant difference.
Results
Clinical findings
Forty eight patients (65.8%, 48/73) completed the study period and will be here analyzed. Age was 58.8 ± 10.9 years, 85.4% were females and BMI was 31.8 ± 4.7 kg/m2. Type 2 diabetes mellitus was present in 52.8%, 79.2% exhibited dyslipidemia and 66.7% suffered from arterial hypertension. Tolerance of the diet was adequate, no patient being excluded on account of diarrhea, constipation, abdominal distention, or dehydration. Excluded patients exhibited minor clinical differences without significance (age 54.8 ± 11.0 years, P = 0.115, 68.0% females, P = 0.203, BMI 31.3 ± 5.8 kg/m2, P = 0.472). Reasons for discontinuation were carbohydrate (sweet) addiction with suspected breach of the diet (12.3%, 9/73), and failure to attend scheduled consultations (21.9%, 16/73).
Anthropometric measurements and body composition
Anthropometric variables and body composition findings can be appreciated in table I. No significant changes were detected, even though a tendency (P = 0.08) toward reduction of waist circumference measurements could be perceived.
Biochemical analysis-gross findings
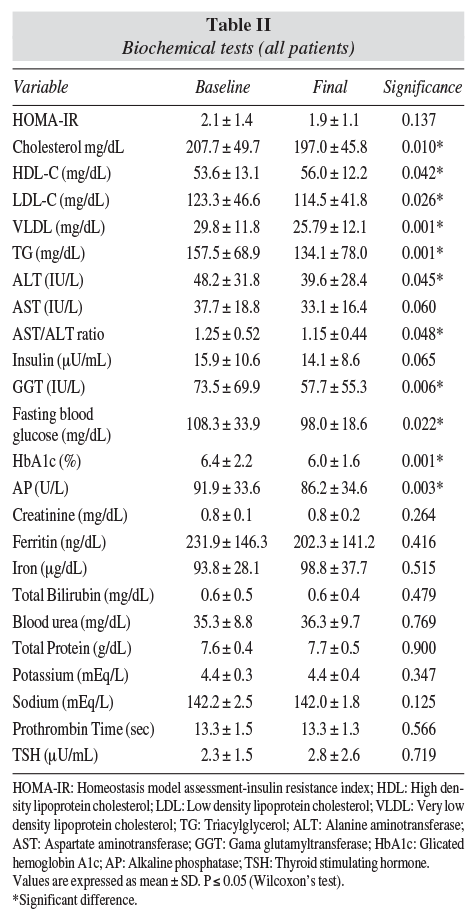
Serum markers of metabolic syndrome and liver steatosis underwent robust positive changes after seventy five days of the nutrition intervention. Significant improvement was confirmed for all lipid fractions, and all liver enzymes except for AST, which nevertheless displayed a tendency (P = 0.060).
Two glucose indexes improved as well, namely fasting blood glucose and HbA1c. No difference was registered for HOMA-IR, insulin, creatinine, ferritin, iron, total bilirubin, urea, total protein, potassium, sodium, prothombin time and thyroid-stimulating hormone (table II).
Biochemical analysis-stratified population
Population was rather evenly divided between those with positive gain (22/48, 45.8%) and those who lost weight during the observation period (21/48, 43.8%). Few patients remained with stable BMI (< 0.2% change, 5/48, 10.4%) and no statistical confirmation was detected for any outcome. In this sense, such patients are not depicted here. Weight fluctuations were mild in all circumstances and did not reach statistical significance for weight gaining or losing cohorts either, namely 32.2 ± 4.3 vs. 33.0 ± 4.1 kg/m2, P = 0.518 and 31.3 ± 5.2 vs. 29.7 ± 5.3 kg/m2, P = 0.310 (tables III and IV).
As pointed out in table IV, association between weight decrease and liver benefit could be elicited in such circumstances for ALT, AP and AST/ALT ratio. No change could be demonstrated in patients who gained weight.
Univariate and multivariate regression analysis
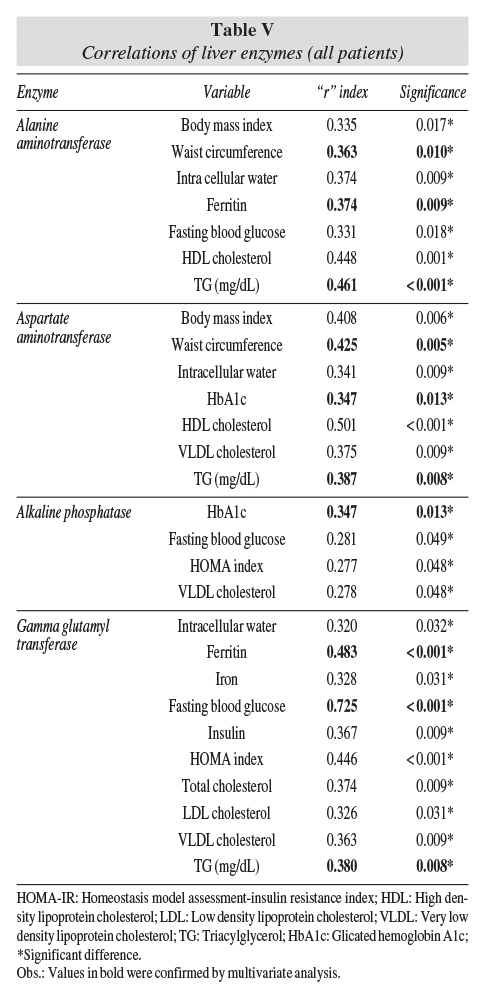
Several nutritional and biochemical variables correlated with liver improvement (table V). Multivariate assessment confirmed that waist circumference, ferritin, triacylglycerol, and markers of glucose homeostasis were the most relevant associated with liver enzymes.
Discussion
The present study demonstrated that a high protein, low calorie diet was associated with improvement of lipid profile, glucose homeostasis and liver enzymes. These findings are consistent with the well established principle of calorie restriction in the management of metabolic syndrome components and liver histology.10,23
Recent guidelines endorsed by the American Association for the Study of Liver Diseases, American College of Gastroenterology and American Gastroenterological Association recommend lifestyle intervention for weight reduction for adults and children with NASH.24 European guidelines also support this recommendation.25 Such agreement notwithstanding, our results move one step farther, in the sense that even in the absence of significant weight loss, the high protein diet already displayed advantages. A recent systematic reviews conclude that weight loss is important in order to reduce hepatic damage. It is highlighted that in such circumstances, liver fat may diminish by 10-80% and aminotransferases by 21-60%. Reduction of as little as 3-5% of body weight attenuates hepatic steatosis, but when the targets are inflammation and progression of NASH, as much as 10% weight loss may be needed.26
Also in the Swedish Obesity Study, the largest and longest cohort study of bariatric surgery in the world, weight loss was strongly associated with an advantageous hepatic profile. Serum ALT and AST diminished at 2- and 10- year follow up, and the reduction in ALT levels was proportional to the degree of weight loss.27 Degree of weight loss has been emphasized by many as critical, and results in the current protocol were somewhat disappointing, given the prescribed 500 kcal restriction (-1.3% decrease of BMI after 75 days). This outcome is nevertheless consistent with the fact that dieting patients often fail to reach targeted intakes.28 One interesting finding is that HDL cholesterol, a negative marker of carbohydrate intake, significantly increased in the weight losing population however not in those gaining weight. There are reasons to believe that compliance with carbohydrate restriction therefore occurred and was relevant for weight loss.28 One should recognize that nearly all lifestyle studies in fatty liver disease report major weight decrease, rendering comparison with the current protocol difficult. Nevertheless, even during use of sever calorie restriction, patients who fail to respond or even gain weight may be observed.
A representative investigation with several therapeutic arms was conducted by St George et al.29 Diet was not defined, except regarding avoidance of saturated fats and processed food, and increase in omega 3 and fiber sources. Energy reduction target was similar to ours (400-570 kcal/day). In contrast all groups exercised more than 200 minutes/week. The group with the most intensive follow-up exhibited a 1.0 kg/m2 mean reduction in BMI, whereas those undergoing low-intensity management lost 0.7 kg/m2. Such values are consistent with the mean 0.4 kg/m2 change in the current study, given the fact that exercise was not here adopted.29
There are few reports comparing different diets for NASH treatment. In the largest randomized trial, with 170 overweight adults, 6 months of a low fat or low carbohydrate diet produced equivalent reductions in intrahepatic fat, ALT, visceral adiposity, total weight and insulin sensitivity.30 A 3-month study similarly found that low carbohydrate and low fat diets reduced ALT to a similar degree.31 Soy protein has been advocated as ideal for NAFLD patients on the basis of animal investigations, nevertheless clinical experience is scant 13 in most of these protocols weight loss tends to be prominent.
A recent study showed that whey protein supplementation (60 g/day) during 30 days, without energy restriction, decreased intrahepatic lipids. BMI remained stable however body fat mass diminished. Additional metabolic advantages were identified for plasma lipids, however not for glucose homeostasis markers.17
Two somewhat different short term protocols with stable body weight also improved hepatic fat content. Protein intake was not changed however a low glycemic index, low fat and low saturated fat prescription was adopted in one center, a monounsaturated-enriched regimen in the other. Again liver enzymes were not part of the protocol.32
In the elegant protocol of de Luis et al.33 142 nondiabetic obese subjects were managed by a normalprotein, low calorie diet during three months. Specifically in the subpopulation exhibiting NAFLD, both BMI and liver enzymes (ALT and AST) significantly decreased, along with improvements in glucose homeostasis. The current investigation is in general agreement with such outcome. Remarkable differences are of course the lack of significant weight loss in our experience, and the adopted diet. In the mentioned series, the proportion at baseline and after 3 months of carbohydrates, lipids and protein was approximately 40.7%-38.0%-20.5% and 39.6%-38.7%-21.5%. Current carbohydrates were similar however protein was substantially elevated, with corresponding lipid reduction, probably offsetting the lack of weight loss with regard to NAFLD alleviation.33
This is the first study with conventional dietary protein and moderate energy restriction to indicate that amelioration of enzymatic profile in NAFLD is not dependent on BMI decrease or body fat mass reduction. One should emphasize that calorie restriction was moderate and did not aim ketogenesis, as experimentally ketogenic protocols tend to stimulate development or recurrence of NAFLD as well as systemic glucose intolerance in mice.34
In the current study, we observed lower levels of enzymes and serum lipids after the nutritional intervention. Stratification according to weight changes revealed influence of minor fluctuations of BMI on changes of ALT, AP and AST/ALT ratio, however not of AST and GGT.
One confounding variable was simultaneous decrease in carbohydrate intake. Although moderate (40% of total calories) and well controlled during frequent follow-up consultations, it might have interfered with clinical outcome. Unfortunately available protocols use marked carbohydrate restriction, along with high fat followed by substantial weight loss, therefore precluding direct comparison. One study with moderate carbohydrate decrease (40-45%), however still relatively rich in fat (35-40%) and low in protein (15-20%), recruiting NASH patients, confirmed improved liver histology after one year.35
The absence of a control group was a limitation in our protocol and the small sample due the number of dropouts occurred during the 75 days of the experience despite the relatively moderate regimen. Indeed obese patients have diminished mobility and are relatively resistant to prolonged diets. Even with frequent text messaging adherence of just 60-69% has been achieved in other experiences, in the same range as here observed.36 A recent German article underscores as many as 18 reasons why even sensible and well-monitored diets fail.37 This was not a randomized trial, and liver histology was not assessed. The major strength of our proposal is the simple nature of the diet, which does not require expensive ingredients or hard to come by supplements, thus it can be prescribed in any environment.
In conclusion, this is the first study demonstrating the value of moderate calorie restriction, non ketogenic and not weight-loss inducing, coupled with substantially increased conventional protein, in the management of NAFLD. Benefit for liver enzymes, fasting glucose and lipid profile could be demonstrated. Randomized trials with long-term follow-up, including intrahepatic lipids and liver histology, should provide more insight on pathophysiologic mechanisms as well as prognostic implications of such approach, particularly for weight-loss resistant patients.
References
1. Cave M, Deaciuc I, Mendez C, et al. Nonalcoholic fatty liver disease: predisposing factors and the role of nutrition. J Nutr Biochem 2007; 18: 184-95. [ Links ]
2. Haukeland JW, Konopski Z, Linnestad P, et al. Abnormal glucose tolerance is a predictor of steatohepatitis and fibrosis in patients with non-alcoholic fatty liver disease. Scand J Gastroenterol 2005; 40: 1469-77. [ Links ]
3. Browning JD, Szczepaniak LS, Dobbins R, et al. Prevalence of hepatic steatosis in an urban population in the United States: impact of ethnicity. Hepatology 2004; 40: 1387-95. [ Links ]
4. Kang H, Greenson JK, Omo JT, et al. Metabolic syndrome is associated with greater histologic severity, higher carbohydrate, and lower fat diet in patients with NAFLD. Am J Gastroenterol 2006; 101: 2247-53. [ Links ]
5. Angulo P. Obesity and nonalcoholic fatty liver disease. Nutr Rev 2007; 65: S57-63. [ Links ]
6. Zivkovic AM, German JB, Sanyal AJ. Comparative review of diets for the metabolic syndrome: implications for nonalcoholic fatty liver disease. Am J Clin Nutr 2007; 86: 285-300. [ Links ]
7. Utzschneider KM, Kahn SE. Review: The role of insulin resistance in nonalcoholic fatty liver disease. J Clin Endocrinol Metab 2006; 91: 4753-61. [ Links ]
8. Yasutake K, Kohjima M, Nakashima M, Kotoh K, Nakamuta M, Enjoji M. Nutrition therapy for liver diseases based on the status of nutritional intake. Gastroenterol Res Pract 2012; 2012: 859697. [ Links ]
9. Maersk M, Belza A, Stødkilde-Jørgensen H, et al. Sucrose-sweetened beverages increase fat storage in the liver, muscle, and visceral fat depot: a 6-mo randomized intervention study. Am J Clin Nutr 2012; 95: 283-9. [ Links ]
10. Catalano D, Trovato GM, Martines GF, Randazzo M, Tonzuso A. Bright liver, body composition and insulin resistance changes with nutritional intervention: a follow-up study. Liver Int 2008; 28: 1280-7. [ Links ]
11. Dixon JB, Bhathal PS, O'Brien PE. Weight loss and non-alcoholic fatty liver disease: falls in gamma-glutamyl transferase concentrations are associated with histologic improvement. Obes Surg 2006; 16: 1278-86. [ Links ]
12. Scaglioni F, Marino M, Ciccia S, et al. Short-term multidisciplinary non-pharmacological intervention is effective in reducing liver fat content assessed non-invasively in patients with nonalcoholic fatty liver disease (NAFLD). Clin Res Hepatol Gastroenterol 2012. [ Links ]
13. Oliveira LP, de Jesus RP, Freire TO, Oliveira CP, Castro Lyra A, Lyra LG. Possible molecular mechanisms soy-mediated in preventing and treating nonalcoholic fatty liver disease. Nutr Hosp 2012; 27: 991-8. [ Links ]
14. Vilar L, Oliveira CP, Faintuch J, et al. High-fat diet: a trigger of non-alcoholic steatohepatitis? Preliminary findings in obese subjects. Nutrition 2008; 24: 1097-102. [ Links ]
15. de Wit NJ, Afman LA, Mensink M, Müller M. Phenotyping the effect of diet on non-alcoholic fatty liver disease. J Hepatol 2012; 57: 1370-3. [ Links ]
16. McCarthy EM, Rinella ME. The role of diet and nutrient composition in nonalcoholic Fatty liver disease. J Acad Nutr Diet 2012; 112: 401-9. [ Links ]
17. Bortolotti M, Maiolo E, Corazza M, et al. Effects of a whey protein supplementation on intrahepatocellular lipids in obese female patients. Clin Nutr 2011; 30: 494-8. [ Links ]
18. Grundy SM, Hansen B, Smith SC, et al. Clinical management of metabolic syndrome: report of the American Heart Association/National Heart, Lung, and Blood Institute/American Diabetes Association conference on scientific issues related to management. Arterioscler Thromb Vasc Biol 2004; 24: e19-24. [ Links ]
19. Clifton PM, Keogh JB, Noakes M. Long-term effects of a highprotein weight-loss diet. Am J Clin Nutr 2008; 87: 23-9. [ Links ]
20. Wycherley TP, Buckley JD, Noakes M, Clifton PM, Brinkworth GD. Comparison of the effects of weight loss from a highprotein versus standard-protein energy-restricted diet on strength and aerobic capacity in overweight and obese men. Eur J Nutr 2013; 52: 317-25. [ Links ]
21. Lean ME, Han TS, Morrison CE. Waist circumference as a measure for indicating need for weight management. BMJ 1995; 311: 158-61. [ Links ]
22. Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985; 28: 412-9. [ Links ]
23. Schwarz J, Tomé D, Baars A, Hooiveld GJ, Müller M. Dietary protein affects gene expression and prevents lipid accumulation in the liver in mice. PLoS One 2012; 7: e47303. [ Links ]
24. Chalasani N, Younossi Z, Lavine JE, et al. The diagnosis and management of non-alcoholic fatty liver disease: practice guideline by the American Gastroenterological Association, American Association for the Study of Liver Diseases, and American College of Gastroenterology. Gastroenterology 2012; 142: 1592-609. [ Links ]
25. Ratziu V, Bellentani S, Cortez-Pinto H, Day C, Marchesini G. A position statement on NAFLD/NASH based on the EASL 2009 special conference. J Hepatol 2010; 53: 372-84. [ Links ]
26. Peng L, Wang J, Li F. Weight reduction for non-alcoholic fatty liver disease. Cochrane Database Syst Rev. 2011: CD003619. [ Links ]
27. Burza MA, Romeo S, Kotronen A, et al. Long-Term Effect of Bariatric Surgery on Liver Enzymes in the Swedish Obese Subjects (SOS) Study. PLoS One 2013; 8: e60495. [ Links ]
28. Sacks FM, Bray GA, Carey VJ, et al. Comparison of weight-loss diets with different compositions of fat, protein, and carbohydrates. N Engl J Med 2009; 360: 859-73. [ Links ]
29. St George A, Bauman A, Johnston A, Farrell G, Chey T, George J. Effect of a lifestyle intervention in patients with abnormal liver enzymes and metabolic risk factors. J Gastroenterol Hepatol 2009; 24: 399-407. [ Links ]
30. Haufe S, Engeli S, Kast P, et al. Randomized comparison of reduced fat and reduced carbohydrate hypocaloric diets on intrahepatic fat in overweight and obese human subjects. Hepatology 2011; 53: 1504-14. [ Links ]
31. de Luis DA, Aller R, Izaola O, Gonzalez Sagrado M, Conde R. Effect of two different hypocaloric diets in transaminases and insulin resistance in nonalcoholic fatty liver disease and obese patients. Nutr Hosp 2010; 25: 730-5. [ Links ]
32. Utzschneider KM, Bayer-Carter JL, Arbuckle MD, Tidwell JM, Richards TL, Craft S. Beneficial effect of a weight-stable, low-fat/low-saturated fat/low-glycaemic index diet to reduce liver fat in older subjects. Br J Nutr 2013; 109: 1096-104. [ Links ]
33. de Luis DA, Aller R, Izaola O, Sagrado MG, Conde R, Gonzalez JM. Effect of a hypocaloric diet in transaminases in nonalcoholic fatty liver disease and obese patients, relation with insulin resistance. Diabetes Res Clin Pract 2008; 79: 74-8. [ Links ]
34. Schugar RC, Crawford PA. Low-carbohydrate ketogenic diets, glucose homeostasis, and nonalcoholic fatty liver disease. Curr Opin Clin Nutr Metab Care 2012; 15: 374-80. [ Links ]
35 Huang MA, Greenson JK, Chao C, et al. One-year intense nutritional counseling results in histological improvement in patients with non-alcoholic steatohepatitis: a pilot study. Am J Gastroenterol 2005; 100: 1072-81. [ Links ]
36. Shapiro JR, Koro T, Doran N, et al. Text4Diet: a randomized controlled study using text messaging for weight loss behaviors. Prev Med 2012; 55: 412-7. [ Links ]
37. Ellrott T. 18 reasons why diet fail. MMW Fortschr Med 2013; 155: 49-52. [ Links ]
![]() Correspondence:
Correspondence:
Claudia Pinto Marques de Souza de Oliveira.
Department of Gastroenterology.
University of Sao Paulo. School of Medicine.
Av. Dr. Eneas de Carvalho Aguiar, 255.Instituto Central, #9159.
05403-000 Sao Paulo. Brazil.
E-mail: cpm@usp.br
Recibido: 26-VIII-2013.
1.a Revision: 23-X-2013.
Aceptado: 5-XI-2013.












 Curriculum ScienTI
Curriculum ScienTI