Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Nutrición Hospitalaria
versión On-line ISSN 1699-5198versión impresa ISSN 0212-1611
Nutr. Hosp. vol.29 no.5 Madrid may. 2014
https://dx.doi.org/10.3305/nh.2014.29.5.7331
ORIGINAL / Síndrome metabólico
Women with metabolic syndrome improve antrophometric and biochemical parameters with green banana flour consumption
Las mujeres con síndrome metabólico mejoran los parámetros antropométricos y bioquímicos con el consumo de "green banana flour"
Sandra Tavares da Silva1, Carolina Araujo dos Santos2, Yassana Marvila Girondoli3, Luiza Mello de Azeredo4, Luis Fernando de Sousa Moraes5, Josiane Keila Viana Gomes Schitini6, Mario Flävio C. de Lima7, Raquel Cristina Lopes Assis Coelho8 and Josefina Bressan9
1-8 Universidade Federal de Viçosa. Minas Gerais. Brazil.
9 Laboratório de Metabolismo Energético e de Composição Corporal. Universidade Federal de Viçosa. Minas Gerais. Brazil.
ABSTRACT
Background: Consumption of green banana flour (GBF) may promote health benefits, such as, decreased appetite, weight loss, glycemic control, intestinal function and lipid profile improvement, aging delay, cancer and heart disease prevention. The aim of this study was to evaluate the effects of green banana flour consumption on anthropometric and biochemical parameters in overweight women.
Methods: The glycemic index of flour in the study was determined. The effects of consumption of 20 g of green banana flour/day on weight, body mass index (BMI), blood pressure, waist and hip circumference, body composition, hemoglobin, lipid profile, glucose, insulin, insulin resistance, liver function and energy intake were evaluated in 25 overweight women for 45 days.
Results: The glycemic index of the flour under study was classified as low. Reduction (p < 0.05) in systolic blood pressure, hip circumference and fasting glucose levels were found in women who had metabolic syndrome criteria.
Conclusions: Consumption of green banana flour (20 g/day) for 45 days did not promote weight loss or changes in body composition in overweight women. It was noted, however, decreased hip circumference. Significant health parameter improvements were also noted in individuals with metabolic syndrome, which showed a reduction in systolic blood pressure and fasting glucose.
Key words: Musa. Flour. Body composition. Glycemic index. Metabolic syndrome X.
RESUMEN
Introducción: El consumo de "green banana flour" (GBF) puede contribuir para la salud como, por ejemplo, aumento em las sensaciones de saciedad , pérdida de peso , control de la glucemia y mejora de la función intestinal y el perfil lipídico, retraso del envejecimiento y la prevención de el cáncer y de las enfermedades del corazón. El objetivo del estudio fue evaluar los efectos del consumo de GBF sobre el parámetros antropométricos y bioquímicos de las mujeres con exceso de peso.
Métodos: Se determinó el índice glucémico de la harina en el estudio. Los efectos del consumo diario de 20 g de GBF en peso, índice de masa corporal (IMC), presión arterial, circunferencia de la cintura y de la cadera, la composición corporal, los niveles de hemoglobina, glucosa e insulina, resistencia insulínica, en la función del hígado y la ingesta alimentaria se evaluaron en 25 mujeres con exceso de peso durante 45 días.
Resultados: El índice glucémico de harina estudiada fue clasificada como bajo. Fue encontrado reducción (p < 0,05) en la presión arterial sistólica, en la circunferencia de la cadera y en la glicemia en mujeres que tenían los criterios de síndrome X metabólico.
Conclusiones: El consumo de harina de plátano sin madurar durante 45 días no promueven la pérdida de peso o los cambios en la composición corporal en mujeres con sobrepeso. Sin embargo, hubo una reducción de la circunferencia de la cadera. Se observó también una mejoría significativa en importantes factores de salud en mujeres con síndrome X metabólico, tales como una reducción en la presión arterial sistólica y de la glucemia.
Palabras clave: Musa. Harina. Composición corporal. Índice glucémico. Síndrome X metabólico.
Abbreviations
AC: Abdominal circumference.
AU: Uric acid.
AUC: Area under the curve .
BF%: Body fat percentage.
BMI: Body Mass Index.
BMR: Basal metabolic rate.
BP: Blood pressure.
C3: C3 complement.
CP: Ceruloplasmin.
DBP: Diastolic blood pressure.
DEXA: Dual Energy X-Ray Absorptiometry.
EBI: Electrical bioimpedance.
GBF: Green banana flour.
GI: Glycemic index.
GOT: Glutamic oxaloacetic transaminase.
GPT: Glutamic-pyrovate transaminase .
Hb: Hemoglobin.
HC: Hip circumference.
HDL: High density lipoprotein.
HOMA-IR: Homeostasis Model Assessment of Insulin Resistance Index.
IDF: International Diabetes Federation.
IMI: Initial metabolism index.
IPAQ: International Physical Activity Questionnaire.
kg: Kilograms.
LDLc: Low density lipoprotein-cholesterol.
LM: Lean mass. m: Meters.
Max: Maximum.
MG: Minas Gerais.
Min: Minimum.
MS: Metabolic Syndrome.
NCEP-ATP III: Third Report of the National Cholesterol Education Program.
QUICKI: Quantitative Sensitivity Check Index.
RS: Resistant starch .
SBP: Systolic blood pressure.
SD: Standard deviation.
T: Time.
TAG: Triacylglycerol.
TC: Total cholesterol.
TFEQ: Three Factor Eating Questionnaire.
TP: Total protein.
UFV: Federal University of Vigosa.
VAS: Visual Analog Scale.
WHR: Waist-hip ratio.
Introduction
Food consumption in the form of bran and powders is widely spread in the media as complementary foods able to promote health benefits, like the green banana flour (GBF). Some of the possible effects are appetite reduction, weight loss, glycemia control, intestinal function and lipid profile improvement, aging delay, cancer and heart disease prevention.
The GBF gained space in the market for being a promising new weight loss treatment. According to studies, the GBF can be used in preparations like cookies1, juices2, cakes3 and cereal bars4. When added to preparations, the GBF enhances fiber, protein, potassium, iron, magnesium, calcium, phosphorous and sodium levels, besides increasing product yield5. The supposed nutritional benefits attributed to the use of the GBF are associated to elevated total starch level (73.45%), resistant starch (RS) (17.5%) and fiber content (nearly 14.5 %)6.
RS presents important physiologic effects in the prevention of intestinal constipation, diverticulitis and hemorrhoids, besides helping in the excretion of toxic compounds considered carcinogenic. It has also been associated to the control of some Metabolic Syndrome (MS) parameters, such as glycemia, LDLc (low density lipoprotein-cholesterol) cholesterol and triglycerides. RS is not digested in the small intestine, thus, it can serve as substrate for microorganisms growth, acting as potential prebiotic agent7. In healthy individuals, the consumption of RS was directly associated with lower caloric ingestion8.
Considering the necessity of evidencing the effects of GBF on health and the real impacts of its consumption, the purpose of this study was to investigate the implications of the consumption of GBF on food ingestion, weight loss, body composition and biochemical profile of overweight adult women exhibiting or not MS.
Materials and Methods
Subjects
This is an intervention study, in which 40 overweight women aged 20 to 45 years were recruited by the media (fig. 1). The adopted exclusion criteria were the presence of allergy/intolerance to test foods (reported before or during the study), level of physical activity classified as active or very active according to the International Physical Activity Questionnaire (IPAQ), restrictive food behavior, according to the Three Factor Eating Questionnaire (TFEQ), use of medication (except oral contraceptives), tobaccoism, metabolic-chronic diseases, (diabetes mellitus, systemic arterial hypertension, hypothyroidism). Sample calculation was carried out considering an expected loss of 2.5 kg body weight9,10 and 90% statistical power, resulting in 23 volunteers.
MS diagnosis was carried out in accordance with criteria of International Diabetes Federation (IDF)11, which has as compulsory component the waist circumference higher than 80 cm associated to two or more of the following criteria: plasmatic glucose ≥ 100 mg/dL, triglycerides ≥ 150 mg/dL, high density lipoprotein (HDL) < 50 mg/dL, systolic blood pressure ≥ 130 mmHg or diastolic ≥ 85 mmHg.
The study was approved and conducted according to the Ethics Committee in Human Research of the Federal University of Viçosa (process number 147/2011-UFV, MG, Brazil) and participants signed a free and informed consent form.
Consumption of Green Banana Flour
The GBF consumed by participants was obtained in the local market. The portions were distributed weekly in individual plastic bags containing 20 g, identified with the participant name and date for consumption. Participants were instructed to consume the portion of GBF for breakfast, with milk, yogurt, milkshake or fruit juice.
Evaluation of Physical Activity Level
The level of physical activity was quantified by the IPAQ, short version proposed by the World Health Organization, validated for the Brazilian population, according to Matsudo et al.12. Volunteers were asked to keep their physical activity habits during the experimental period.
Evaluation of Food Behavior
The food behavior of volunteers was evaluated by application of the TFEQ, before the beginning of the study. This questionnaire analyzed three food behavior aspects: (i) cognitive restriction scale, which verifies food prohibition to influence weight or body shape; (ii) emotional food scale, which measures the tendency to eat exaggeratedly in reply to negative emotional status, like solitude, anxiety and depression; (iii) uncontrolled eating scale, which verifies the tendency to lose control in presence of hunger or external stimuli. For food restriction, level < 14 was adopted as inclusion criterion. This cut point was adopted aiming to exclude volunteers with very restrictive food behavior, which might influence test food consumption13.
Anthropometry
The anthropometric evaluation was carried out before and after 45 days of intervention with GBF (fig. 1). The body mass in kilograms (kg) was obtained in a digital electronic scale (Tanita®) with 150 kg capacity and 50 g accuracy. Height was registered in meters (m) and obtained using a vertical anthropometer, according to recommendations of Jellife (1968).
The anthropometric evaluation was carried out by Body Mass Index (BMI) calculation. The abdominal circumference was measured at the umbilical level, using a non-extensible tape measure, with the volunteer standing and after complete exhalation. The hip circumference was measured at its largest circumference. Body composition measurements were obtained by electrical bioimpedance (Biodinamics, Model 310) and by Dual Energy X-Ray Absorptiometry (DEXA) (Lunar Densiometry - GE®) aided by Encore 2010 software (version 13.3) conducted by a specialized technician. Evaluations were carried out in the morning with fasting participants after 15 minutes of rest.
Blood pressure (BP) measurement
Blood pressure measurement was obtained using stethoscope and column sphygmomanometer device, both calibrated in accordance with the standards established by the Brazilian Society of Cardiology14 and carried out by a trained professional. Three measurements were carried out, with one minute interval, and the average of the last two considered the blood pressure of the individual. If the diastolic pressure presented differences higher than 4 mmHg, new measurement was carried out until difference lower than this value was obtained.
Biochemical evaluation
Blood collection was carried out at the beginning and end of the experiment (after 45 days of GBF consumption) in individuals with 12 hours fasting at the Health Division of the UFV (MG, Brazil). Determination of plasmatic triglycerides, total cholesterol and fractions and uric acid levels was done by the enzymatic colorimetric method. Blood count was carried out by electrical impedance and total proteins by the biuret method. Glutamic oxaloacetic transaminase (GOT) and glutamic-pyrovate transaminase (GPT) were analyzed by the kinetic method and ceruloplasmin by nefelometry. C3 complement and fasting serum insulin were measured by immunoturbidimetric assay and electrochemiluminescence, respectively. Fasting plasma glucose levels were determined by the glucoseoxidase method. Tests were carried out in the biochemical analyzer Cobas Mira Plus (Roche®).
The Homeostasis Model Assessment of Insulin Resistance Index (HOMA-IR) was calculated according to the formula below15. Value higher than 2.516 was adopted as criterion for presence of insulin resistance.
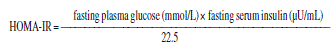
The Quantitative Sensitivity Check Index (QUICKI) was also used, which values inferior or equal to 0.33 suggest insulin resistance17. This value was calculated following the formula below.

Dietetic evaluation
Food consumption was evaluated by food registers filled out on alternate days, including one day of the weekend, at the following moments: one week prior to beginning of study, first, third and sixth weeks of GBF consumption (fig. 1). The registers were revised in the presence of volunteers to guarantee higher information precision. The domestic measures of ingested foods were then converted into grams and the caloric ingestion of macro and micronutrients were analyzed using the DietProâ (version 5.0) software.
Evaluation of Subjective Appetite Sensation
For evaluation of subjective appetite sensation a bipolar visual analogy scale (Visual Analog ScaleVAS) with a 0 to 100 mm, non-milimetered scale adapted for the study was used, according to recommendations of Blundell et al.18. VAS was prepared with questions that evaluated the different subjective components that lead to food ingestion, such as, hunger, gastric plenitude, satiety, craving for food and salty, sweet and greasy foods.
The volunteers were instructed to fill out scales for five different days according to the study protocol: day prior to beginning of consumption (T-1), first day of consumption (T1), at 15 days (T15), at 30 days (T30) and at last day of consumption (T45) (fig. 1). The volunteers filled out the scales at seven different times: immediately before breakfast, every 30 minutes until two hours after breakfast and immediately before lunch and dinner. The volunteers were reminded, by telephone, to fill out of the scale. The quantification was done by the distance between the marked point by the volunteers and the left end of the line18.
By means of the trapezoidal method18 it was possible to calculate the area below the curve produced by each question related to one subjective appetite sensation. Calculation of areas was carried out using the SlideWrite Plus (version 7.0 Demo) software.
Determination of GBF Glycemic Index (GI)
Ten adult volunteers of both sex, mean age of 25.3 years and 24.7% (± 3.8) fat percentage were recruited. Absence of past or familiar history of diabetes or insulin resistance and use of medication that could affect glycemia or the energetic metabolism were considered inclusion criteria. Tests were conducted at the Clinical Analyses Laboratory of UFV (MG, Brazil).
The tested load consisted of 46.3g of GBF (containing 25 g of available carbohydrates) diluted in 250 mL water. The test load was ingested at once by the volunteers. Glucose was chosen as reference food, which portion of 25 g was diluted in 250 mL and ingested for three non-consecutive days. After 10 hours of fast the volunteers went to the University Laboratory to ingest the GBF load or glucose. The total portion was ingested 15 minutes after fast glycemia determination (baseline).
Glycemic alterations were evaluated by capillary blood glucose concentrations determination, using Accucheck Active® device, at baseline (immediately before ingestion), 15, 30, 45, 60, 90 and 120 minutes (after beginning of ingestion). By the trapezoidal method the positive area formed below the glycemic response curve was calculated, using Slide Write (7.0 version demo) software. GBF Glycemic Indexes were expressed as mean percentages of glycemic responses obtained after glucose ingestion. GI was determined by arithmetical average of values obtained by ten volunteers and classified ad according to international standards19.
Statistical analysis
The normality of variables was verified by the Shapiro-Wilk test. The effects of intervention on anthropometric, biochemical parameters and on food ingestion were evaluated by paired t-test, for variables with normal distribution, by Wilcoxon test, for variables that presented not normal distribution and by Friedman test for evolution evaluations along the study period. Statistical analyses were carried out in the SPSS Statistics 17.0 software. Significance level of 5% was adopted.
Results
Sample Characterization
Out of 40 volunteers who began the study, 25 completed the study protocol (fig. 1). Participant's mean age was 34 years, with mean BMI of 27.73 kg/m2, being 12 participants classified as sedentary and 13 as moderately active, according to the IPAC. Regarding food behavior, mean scores of 11 were noticed for cognitive restriction, 8.6 for desinhibition and 6 for uncontrolled eating, which is classified as high, low and medium scores, respectively. According to criteria of the International Diabetes Federation11, 68% of evaluated women (n = 17) presented Metabolic Syndrome.
Anthropometry, Body Composition and Biochemical Parameters
GBF consumption did not cause weight or body composition alteration in the evaluated women. It was observed, however, reduction of the hip circumference and consequent increase of the waist-hip ratio (table I).
In the MS group, besides hip circumference reduction and ratio waist-hip increase, reduction of systolic blood pressure was observed (table II). In the group without MS, no anthropometric and body composition parameter presented significant alteration after the intervention period (not presented data).
Regarding the biochemical parameters, MS group presented reduction (p = 0.032) in glucose levels when compared with the group composed by volunteers without MS (table II). No significant alteration was noticed in the levels of GOT, GPT, lipid profile and in the evaluated inflammatory markers (uric acid, C3-complement and ceruloplasmin).
There was no statistically significant difference for HOMA-IR and QUICKI indexes before and after GBF consumption. However, according to the HOMA-IR calculation, seven volunteers presented insulin resistance and according to QUICKI, six volunteers. Difference was also not found for HOMA-IR and QUICKI indexes, before and after consumption of GBF in the group of volunteers with MS.
It can be suggested, thus, that women with metabolic disorders present more significant response to GBF consumption when compared with women that although overweight, still do not meet the MS criteria.
Dietetic intake
Increase in mean fiber consumption was observed comparing the period previous to beginning of study (Week 0 = 12.72 g) with the last intervention week (Week 6 = 14.16 g) (p = 0.03). Although GBF consumption increased diet fiber levels, mean ingestion value did not reach the daily recommendation of 25 g for women aged 19 to 50 years20.
The caloric ingestion averages of each meal were quantified, to verify whether GBF interfered in the caloric consumption of meals after breakfast, along the day. When comparing Week 0 with Week 1, a significant reduction was observed in the caloric consumption of the afternoon snack (329.51 kcal in Week 0 vs 227.47 kcal in Week 1; p = 0.019) However, this effect was not supported along the study.
There was no difference in the total caloric ingestion of each volunteer in the different study moments (Week 0, Week 1, Week 3 and Week 6), indicating that GBF was not able to alter the diet caloric ingestion during 45 days of study.
Nutrients (carbohydrates, proteins, lipids, cholesterol, calcium, iron and sodium) presented higher interindividual variation as for the mean consumed quantity, with no difference between the different times considered in the study. Low calcium consumption was observed, which remained below the daily recommendation for women aged 19 to 50 years, of 1000 mg21 in all weeks, even with part of the volunteers having consumed the GBF with milk or yogurt.
Mean consumption of full fatty acids, mono and poly-unsaturated was analyzed in order to verify whether volunteers would compensate the consumption of a weight-reducing food, like the GBF, with ingestion of caloric, fat-rich foods. The consumption of these fatty acids was changed in percentage regarding total caloric consumption, before and at the end of study, and compared with recommendations of Third Report of the National Cholesterol Education Program (NCEP-ATPIII): full fatty acids lower than 7%; monounsaturated fatty acids higher than 20%; and polyunsaturated fatty acids higher than 10%22. It was noticed that 100% and 96% of volunteers consumed lower quantity than that recommended by the NCEP-ATPIII for mono and poly-unsaturated fatty acids, respectively, at the two evaluated times (Week 0 and Week 6). The percentage of volunteers who consumed quantities above the recommendations for full fatty acids increased from 60%, at Week 0, to 80% at Week 6. The results demonstrated a possible food compensation attitude by volunteers, with addition of fat-rich foods in the diet and consumption of a weight-reducing food.
Evaluation of Appetite Sensation
The paired analyses between T3 and T1 showed increase in subjective satiety sensation (p = 0.035) and gastric plenitude (p = 0.046) as well as reduction in appetite sensation for salty (p = 0.012), sweet (p = 0.025) and greasy (p = 0.044) foods (fig. 2). In analyses between T1 and T45, only reduction of "desire for salty foods" (p = 0.009) maintained.
The analyses of VAS were repeated, subsequently, excluding results before lunch and dinner, as these were not standardized intervals of time among volunteers, which might interfere in the results. However, significant results were not found. The time intervals between breakfast and lunch and from breakfast to dinner also did not show significant difference along the GBF consumption days.
Determination of Glycemic Index (GI)
The mean area below the glycemic response curve was considerably higher with glucose consumption when compared with the area that represents GBF consumption (fig. 3). Using the proportion between these responses it was determined that the GBF GI is 15.3, being classified as a low GI food (< 55)19.
Discussion
The present study demonstrated that daily consumption of 20 g of GBF contributed for reduction of the hip circumference and consequent waist/hip ratio increase, reduction of systolic blood pressure and plasma glucose only in MS group. These data could be explained by low GI found in the green banana flour.
It has been demonstrated that food fiber provides important health results and contributes to prevention of several diseases. GI of carbohydrates and fiber consumption are relevant dietetic factors that can influence ponderal control and SM components23.
Sharma et al.7, Scholz-Ahrens et al.24 and Nugent25 report beneficial properties of fiber RS, such as protective effects against diabetes, colorectal cancer, cardiovascular diseases and obesity, including potential physiologic effects, as glycemic control and insulin response, increase of satiety and reduction of caloric ingestion. However, these RS physiologic properties and, consequently, possible beneficial effect, can widely vary depending on the study design and differences in source, type and quantity of consumed RS25.
The group of volunteers with MS showed significant reduction (p = 0.032) in glucose levels when compared with the group without MS (table II). A study carried out with diabetic animals or animals receiving high dosage of sucrose showed reduction in serum glucose levels, after being supplemented with native banana RS in different concentrations and forms for four weeks26, corroborating the results of the present study. Nevertheless, the same group of investigators, using supplementation of 24 g/day of native banana RS for four weeks in obese and type 2 diabetes patients, found significant reduction of body weight and BMI when compared with control groups. However, no significant result was observed fasting glycemia, fat percentage and serum levels of triglycerides, cholesterol, LDL and HDL-cholesterol27.
Park et al.28 carried out a study with supplementation of 24g/day RS in healthy individuals with excess weight, observing fast glycemia reduction in individuals when compared with the group control. No alteration was observed in body weight or other anthropometric parameters, serum insulin or C3 complement in the period of 21 days of RS consumption. In our study, GBF addition, which has an important quantity of RS, reduced fast glycemia of volunteers with MS.
RS consumption improved insulin sensitivity of individuals with MS29. In our study, HOMA-IR index, which is correlated with invasive insulin sensitivity tests, did not indicate alteration in insulin resistance, or QUICKI index. Since fast glycemia reduction was observed, it is suggested that the intervention time has been insufficient to demonstrate insulin sensitivity improvement.
Regarding dietetic ingestion of volunteers, significant modifications in food consumption or diet calories reduction were not noticed. Only at the first week of GBF consumption, calories reduction was observed in the afternoon snack. In addition, the evaluation of appetite sensations by VAS showed significant results for appetite reduction only at the first day of GBF consumption. Such results can be explained by the fact that our organism adapts to food habit modifications by physical mechanisms related to energetic compensation30, minimizing the functional effects of a food consumed for a long time.
The motivation of volunteers with the possibility to lose weight and improve some health parameters may still have influenced the food choices in the initial period. However, as time passed, reduction of adherence to treatment occurred along with the attention to daily dietetic consumption31,32.
The increase of satiety sensation found in our study can be due to the elevated fiber content and/or GBF RS. According to Ramos et al.33, GBF can present 10 to 40% of RS, which has the property of promoting satiety34. Studies using VAS demonstrated higher satiety verified by RS35. However, Bodinham et al.8 while testing the effects of RS in healthy adults concluded that nutrients promoted lower caloric consumption, although VAS analysis has not shown hunger reduction and satiety increase results. In our study, there were no differences in caloric consumption, which was also not observed in the results of satiety and hunger evaluated by VAS. Although VAS is a subjective mensuration, according to Blundell et al.18, in appropriate study delineation and analyses, this method is sensitive and reliable to analyze the subjective appetite sensations in free-living conditions. The results obtained by VAS are coherent with the hunger perception mean score found, with absence of food consumption reduction and, consequently, not reduction of weight.
It is worth emphasizing that this study aimed to investigate the isolated effect of the consumption of the GBF in the control of the weight, satiety, physical composition and biochemical profile, considering the food consumption of free life of the volunteers. Nutritional directions were not given during the study and flour consumption was not associated to any food plan and/or caloric restriction.
In these conditions, the results of this study demonstrate that GBF itself was not able to increase satiety, reduce appetite or caloric consumption and, consequently, promote weight loss. It is suggested that diets with caloric restriction are the main factor for continued weight reduction36 along with professional support32 and that diet macronutrient composition, alone, is not able to cause such an effect36. Rocha et al.37 in a study with a similar study experimental design failed to certify functional allegations of weight reduction and increased satiety of "Human Ration". This product is a mix of cereals with consumption stimulated as a meal replacement, which should not be encouraged37. Metaanalysis that evaluated the magnitude of the effect of green tea or its extracts (caffeine and catechins) on body weight and body composition did not find significant results that would substantiate the recommendation of this food/product38.
Moreover, the consumption of a food with functional allegations can induce the individual to a "compensation" idea by consuming more caloric and fat-rich foods and simple carbohydrates. In other words, the individual may tend not to follow the principles of healthy eating due to believing that the studied food can minimize dietary excesses and inadequacies.
The GBF low GI3,12 is of great interest for the control of glycemia, diabetes and other pathologies. In the long run, consumption of foods with high GI can lead to development of insulin resistance and type 2 diabetes mellitus, besides being associated to other non-communicable chronic diseases, such as cardiovascular diseases, obesity and some types of cancer39. Studies have demonstrated that low GI carbohydrates play an important role in the improvement of insulin sensibility, contributing to metabolic processes and body weight control40. For Pereira34, RS contributes to a slower digestion, which brings lower glycemic and, consequently, insulin response. This author still highlights the insoluble and soluble fiber role practiced by RS, respectively, carrying molecules of fat and sugar that will be absorbed more slowly, promoting satiety.
Studies have found expressive quantities of RS in the GBF41, being this, perhaps, the main responsible for low GI. Thakorlal et al.42, while analyzing the RS content of different green banana species, concluded that the level oscillates greatly between the fruit varieties, ripening time and processing.
Borges et al.3, while characterizing the GBF showed that this is a rich source of starch, protein, potassium, phosphorous, magnesium, copper, manganese and zinc, able to substitute other foods since they have high caloric value and higher nutritional content than other types of existing flours in the market. When used in partial substitution to the plain flour for cookies production, it did not alter the sensory characteristics of this food and presented good acceptance in different age groups1-4. This suggests that the GBF can be used as nutritional enrichment raw material of foods, contributing to reduce GI of meals1 representing an alternative for patients who need to exclude plain flour from the diet, such as the celiac disease.
Conclusion
The consumption of green banana flour (20 g/day) for 45 days did not promote alteration of weight or body composition in overweight women. It was observed, however, reduction of the hip circumference and increase of the hip-waist circumference. After classification of volunteers as bearers or not of metabolic syndrome, the group with MS showed reduction of systolic blood pressure and fast glycemia. This last result might be attributed to low glycemic index of the test food.
Thus, women with metabolic syndrome respond better to the product, although it has been observed that the use of GBF alone did not reach the supposed effects on lipid profile improvement, appetite reduction, satiety increase and weight loss. Therefore, the consumption of this food must be associated to directions on healthy eating and a diet plan with caloric restriction, including regular practice of physical activity, so that the health benefits and improvement of other MS parameters are reached.
References
1. Fasolin LH, Almeida GC, Castanho OS, Netto-Oliveira ER. Biscoitos produzidos com farinha de banana: avaliações química, física e sensorial. Ciênc Tecnol Aliment 2007; 27 (3): 524-9. [ Links ]
2. Taipina MS,Cohen VH, Dellmastro NL, Rodas MA, Dellatorre JCM. Aceitabilidade sensorial de suco de manga adicionado de polpa de banana (musasp) verde. Rev Inst Adolfo Lutz 2004; 63 (1): 49-55. [ Links ]
3. Borges AM, Pereira J, Lucena EMP. Caracterização da farinha de banana verde. Ciênc Tecnol Aliment 2009; 29 (2): 333-9. [ Links ]
4. Santos JF. Avaliação das propriedades nutricionais de barra de cereais elaboradas com farinha de banana verde. Dissertação de Mestrado. Universidade de São Paulo - USP, Programa de Pós-Graduação em Ciências dos Alimentos; 2010. [ Links ]
5. Izidoro DR, Scheer AP, Negre MFO, Haminiuk CWI, Sierakowski MR. Avaliação físico-química, colorimétrica e aceitação sensorial de emulsão estabilizada com polpa de banana verde. Rev Inst Adolfo Lutz 2008; 67 (3): 1-67-76. [ Links ]
6. Saifullah R, Abbas FMA, Yeoh SY, Azhar ME. Utilization of green banana flour as a functional ingredient in yellow noodle. Int Food Res J 2009; 16 (4): 373-9. [ Links ]
7. Sharma A, Yadav BS, Ritika BY. Resistant starch: physiological roles and food applications. Food Rev Int 2008; 24 (2): 193-234. [ Links ]
8. Bodinham CL, Frost GS, Robertson MD. Acute ingestion of resistant starch reduces food intake in healthy adults. Br JNutr 2010; 103 (6): 917-22. [ Links ]
9. Saltzman E, Moriguti JC, Das SK, Corrales A, Fuss P, Greenberg AS, Roberts SB. Effects of a cereal rich in soluble fiber on body composition and dietary compliance during consumption of a hypocaloric diet. J Am Coll Nutr 2001; 20 (1): 50-7. [ Links ]
10. Weyne GRS. Determinação do tamanho da amostra em pesquisas experimentais na área de saúde. Arq Méd ABC 2004; 29 (2): 87-90. [ Links ]
11. International Diabetes Federation (IDF). The IDF consensus worldwide definition of the metabolic Syndrome (www.idf.org/webdata/docs/MetSyndrome_FINAL.pdf). [ Links ]
12. Matsudo SMM, Araújo T, Marsudo V, Andrade D, Andrade E, Oliveira LC, Braggion G. Questionário internacional de atividade física (IPAQ): estudo de validade e reprodutibilidade no Brasil. Rev Bras Ativ Fís Saúde 2001; 6 (2): 5-18. [ Links ]
13. Stunkard AJ, Messick S. The Three Factor Eating Questionnaire to measure dietary restraint, disinhibition and hunger. J Psychosom Res 1985; 29 (1): 71-83. [ Links ]
14. Sociedade Brasileira de Cardiologia. VI Diretrizes Brasileiras de Hipertensão. 2010; 95 (1): 1-51. [ Links ]
15. Vogeser M, Konig D, Frey I, Predel HG, Parhofer KG, Berg A. Fasting serum insulin and the homeostasis model of insulin resistance (HOMA-IR) in the monitoring of lifestyle interventions in obese persons. Clin Biochem 2007; 40 (13): 964-8. [ Links ]
16. Bonora E, Targher G, Alberiche M, Bonadonna RC, Saggiani F, Zenere MB, Monauni T, Muggeo M. Homeostasis model assessment closely mirrors the glucose clamp technique in the assessment of insulin sensitivity: studies in subjects with various degrees of glucose tolerance and insulin sensitivity. Diabetes Care 2000; 23 (1): 57-63. [ Links ]
17. Carmina E, Lobo RA. Use of fasting blood to assess the prevalence of insulin resistance in women with polycystic ovary syndrome. Fertil Steril 2004; 82 (3): 661-5. [ Links ]
18. Blundell J, Graaf C, Hulshof T, Jebb S, Livingstone B, Lluch A, Mela D, Salah S, Schuring E, Knaap H, Westerterp M. Appetite control: methodological aspects of the evaluation of foods. Appetite Rev 2010; 11 (3): 251-70. [ Links ]
19. Brouns F, Bjorck I, Frayn KN, Gibbs AL, Lang V, Slama G, Wolever TM. Glycaemic Index Methodology. Nutr Res Rev 2005; 18 (1): 145-71. [ Links ]
20. Washington DC: Dietary Reference Intakes for Calcium and Vitamin D. A Report of the Panel on Macronutrients, Subcommittees on Upper Reference Levels of Nutrients and Interpretation and Uses of Dietary Reference Intakes, Standing Committee on the Scientific Evaluation of Dietary Reference Intakes. "Summary Tables, Dietary Reference Intakes." Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids (Macronutrients). The National Academies Press 2005. (http://www.nap.edu). [ Links ]
21. Washington, DC. Dietary Reference Intakes for Calcium and Vitamin D. Committee to Review Dietary Reference Intakes for Vitamin D and Calcium, Institute of Medicine. "Summary Tables: Dietary Reference Intakes." Dietary Reference Intakes for Calcium and Vitamin D. The National Academies Press 2011. (http://www.nap.edu) [ Links ]
22. The Third Report of the National Cholesterol Education Program (NECP). Expert Panel on Detection: Evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III). JAMA 2001; 285 (19): 2486-97. [ Links ]
23. Sirtori CR, Galli C, Anderson JW, Sirtori E, Arnoldi A. Functional foods for dyslipidaemia and cardiovascular risk prevention. Nutr Res Rev 2009; 22 (2): 244-61. [ Links ]
24. Scholz-Ahrens E, Ade P, Marten B, Weber P, Timm W, Yahya A, Glüer CC, Schrezenmeir J. Prebiotics, probiotics, and synbiotics affect mineral absorption, bone mineral content, and bone structure. J Nutr 2007; 137 (3): 838S-46S. [ Links ]
25. Nugent AP. Health properties of resistant starch. BNF Nutr Bull 2005; 30 (1): 27-54. [ Links ]
26. Ble-Castillo JL, Aparicio-Trápala MA, Olvera-Hernandez V, Rodriguez-Hernandez A, Juarez-Rojop IE, Aguilar-Mariscal H, Vázquez-Sanchez MP, Diaz-Zagoya JC. Abstracts of the 79th EAS Congress. Atherosclerosis Supplements 2011; 12 (1): 1-204. [ Links ]
27. Ble-Castillo JL, Aparicio-Trápala MA, Francisco-Luria MU, Córdova-Uscanga R, Rodríguez-Hernández A, Méndez JD, Díaz-Zagoya JC. Effects of native banana starch supplementation on body weight and insulin sensitivity in obese type 2 diabetics. Int J Environ Res Public Health 2010; 7 (5): 1953-62. [ Links ]
28. Park OJ, Kang NE, Chang MJ, Kim WK. Resistant starch supplementation influences blood lipid concentrations and glucose control in overweight subjects. J Nutr Sci Vitaminol 2004; 50 (2): 93-9. [ Links ]
29. Johnston KL, Thomas EL, Bell JD, Frost GS, Robertson MD. Resistant starch improves insulin sensitivity in metabolic syndrome. Diabet Med 2010; 27 (4): 391-7. [ Links ]
30. Mourao DM, Bressan J. Influência de alimentos líquidos e sólidos no controle do apetite. Rev Nutr 2009; 22 (4): 537-47. [ Links ]
31. Knäuper B, Cheema S, Rabiau M, Borten O. Self-set dieting rules: Adherence and prediction of weight loss success. Appetite 2005; 44 (3): 283-8. [ Links ]
32. Micali FG, Japur CC, Penaforte FRO, Diez-Garcia RW. Weight and body composition variations in overweight women along outpatient nutritional treatment. Nutr Hosp 2014; 29 (3): 526-30. [ Links ]
33. Ramos DP, Leonel M, Leonel S. Amido resistente em farinhas de banana verde. Alim Nutr 2009; 20 (3): 479-83. [ Links ]
34. Pereira KD. Amido resistente, a última geração no controle de energia e digestão saudável. Ciênc Tecnol Aliment 2007; 27 (S): 88-92. [ Links ]
35. Willis HJ, Eldridge AL, Beiseigel J, Thomasc W, Slavin JL. Greater satiety response with resistant starch and corn bran in human subjects. Nutr Res 2009; 29 (2): 100-5. [ Links ]
36. Gilhooly CH, Das SK, Golden JK, McCrory MA, Dallal GE, Saltzman E, et al. Food cravings and energy regulation: the characteristics of craved foods and their relationship with eating behaviors and weight change during 6 months of dietary energy restriction. Int J Obes 2007;31 (12): 1849-58. [ Links ]
37. Rocha JLM, Anunciação CP, Tostes MGV, Valdés ST, Carraro JCC, Alves NEG, Bressan J. Human ration does not alter weight and body composition, but improves the lipid profile of overweight woman. Nutr Hosp 2012; 27 (5): 1460-8. [ Links ]
38. Baladia E, Basulto J, Manera M, Martínez R, Calbet, D. Efecto del consumo de té verde o extractos de té verde en el peso y en la composición corporal; revisión sistemática y metaanálisis. Nutr Hosp 2014; 29 (3): 479-90. [ Links ]
39. Ek KL, Brand-Miller J, Copeland L. Glycemic effect of potatoes. Food Chemistry 2011; 128 (1): 1-11. [ Links ]
40. Wolever TMS. Dietary carbohydrates and insulin action in humans. Br J Nutr 2000; 83 (1): 97-102. [ Links ]
41. Ramos DP, Leonel M, Leonel S. Amido Resistente em Farinhas de Banana Verde. Alim Nutr 2002; 20 (3): 479-83. [ Links ]
42. Thakorlal J, Perera CO, Smith B, Englberger L, Lorens A. Resistant Starch in Micronesian Banana Cultivars Offers Health Benefits. Pacific Health Dialog 2010; 16 (1): 49-60. [ Links ]
![]() Correspondence:
Correspondence:
Sandra Tavares da Silva.
Departamento de Nutrição e Saúde.
Universidade Federal de Viçosa.
Av. PH Rolfs, s/n. Centro.
36570000 Viçosa. Brasil.
E-mail: sandratavs@hotmail.com
Recibido: 7-II-2014.
1.a Revisión: 11-II-2014.
Aceptado: 12-III-2014.














