Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Nutrición Hospitalaria
versión On-line ISSN 1699-5198versión impresa ISSN 0212-1611
Nutr. Hosp. vol.29 no.5 Madrid may. 2014
https://dx.doi.org/10.3305/nh.2014.29.5.6654
ORIGINAL / Síndrome metabólico
Hypocaloric diet associated with the consumption of jam enriched with microencapsulated fish oil decreases insulin resistance
La dieta hipocalórica junto con mermelada enriquecida con aceite de pescado microencapsulado disminuye la resistencia a la insulina
Anna Paula Soares de Oliveira Carvalho1, Sofia Kimi Uehara2, José Firmino Nogueria Netto3 and Glorimar Rosa4
1 Master's degreee candidate of the Graduate Program in Medicine/Cardiology. Federal University of Rio de Janeiro. Medical School.
2 Doctor in Nutritional Sciences at the Graduate Program in Nutrition. Josué de Castro Institute of Nutrition. Federal University of Rio de Janeiro. Brazil.
3 Professor of Department of Physiological Sciences. Biology Institute. State University of Rio de Janeiro. Rio de Janeiro. RJ. Brazil.
4Associate Professor of Clinical Nutrition. Department of Nutrition and Dietetic. Josué de Castro Institute of Nutrition. Federal University of Rio de Janeiro. Brazil.
We are especially thankful to FAPERJ (Research Funding Agency of the State of Rio de Janeiro); CAPES (Government agency linked to the Brazilian Ministry of Education in charge of promoting high standards for post-graduate courses in Brazil); and CNPq (National Council for Scientific and Technological Development) for their financial support to carry out this study.
ABSTRACT
Background: The metabolic syndrome is related to the increase in cardiovascular diseases. Polyunsaturated fatty acids from fish oil help in reducing cardiovascular risk factors and are natural bindings of PPARy2.
Objective: To evaluate the impact of hypocaloric diet associated with microencapsulated fish oil supplementation in women with metabolic syndrome.
Methods: We conducted a randomized, single-blind and placebo-controlled clinical trial with adult women who presented metabolic syndrome (n = 30) for 90 days. The volunteers were divided into two groups: placebo group (n = 15) and microencapsulated fish oil group (n = 15) (3 g/day of microencapsulated fish oil containing 0.41 g/day of eico-sapentaenoic acid and decosahexaneoic acid). Anthropometric, body composition, clinical and laboratory parameters were assessed before and after the intervention. Paired t-test was used for comparisons within groups and Student's t-test for comparison between groups. We considered p < 0.05 as significant values.
Results: The comparison between groups revealed a significant reduction of blood glucose, insulinemia and the homeostasis model assessment in the microencapsulated fish oil group after 90 days, as opposed to the placebo group. We also observed reduction of the systolic arterial pressure in the microencapsulated fish oil group.
Conclusion: A hypocaloric diet associated with the consumption of microencapsulated fish oil was effective in reducing blood glucose, insulinemia and insulin resistance in women with MS.
Key words: Metabolic syndrome. Fish oil. Insulin resistance. Hypocaloric diet. Microencapsulation.
RESUMEN
Antecedentes: El síndrome metabólico se relaciona con un incremento de las enfermedades cardiovasculares. Los ácidos grasos poliinsaturados del aceite de pescado ayudan a reducir los factores de riesgo cardiovascular y son ligandos naturales del PPARy2.
Objetivo: Evaluar el impacto de la dieta hipocalórica asociada con suplementación de aceite de pescado micro-encapsulado en mujeres con síndrome metabólico.
Métodos: Realizamos un ensayo clínico de distribución aleatoria, simple ciego y controlado con placebo en mujeres adultas con síndrome metabólico (n = 30) durante 90 días. Se dividió a las voluntarias en dos grupos: el grupo placebo (n = 15) y el grupo con aceite de pescado microencapsulado (n = 15) (3 g/día de aceite de pescado microencapsulado que contienen 0,41 g/día de ácido eicosapentaenoico y de ácido decosahexaneoico). Se evaluaron parámetros antropométricos, clínicos y de laboratorio y la composición corporal antes y después de la intervención. Se emplearon la prueba t pareada para las comparaciones dentro de los grupos y la prueba t de Student para la comparación entre grupos. Consideramos valores significativos de p < 0,05.
Resultados: La comparación entre grupos reveló una reducción significativa de la glucosa sanguínea, la insulinemia y la evaluación del modelo homeostático en el grupo de aceite de pescado microencapsulado tras 90 días, en comparación con el grupo placebo. También observamos una reducción de la presión arterial sistólica en el grupo con aceite de pescado microencapsulado.
Conclusión: La dieta hipocalórica asociada con el consumo de aceite de pescado microencapsulado fue eficaz en la reducción de la glucosa sanguínea, la insulinemia y la resistencia a la insulina en mujeres con SM.
Palabras clave: Síndrome metabólico. Aceite de pescado, Resistencia a la insulina. Dieta hipocalórica. Microencapsulación.
Introduction
Metabolic syndrome (MS) is characterized by a set of cardiovascular risk factors1 associated with central obesity and insulin resistance (IR) responsible for increased cardiovascular mortality2. The frequency of MS has been greater in women in Brazil3 and in countries like China4 and Spain5.
N-3 polyunsaturated fatty acids (PUFA) -derived from fish oil (FO), eicosapentaenoic acid (EPA) and decosahexaneoic acid (DHA) -act by increasing insulin sensitivity6. N-3 PUFA in FO is susceptible to lipid oxidation7. This characteristic leads to changes in its physical and chemical structure and compromises its sensory characteristics, such as odor and taste8. Microencapsulation increases the stability of PUFA,9 in addition to improving its palatability and avoiding the discontinuity of treatments.
Microencapsulation is a procedure that consists in coating of active materials (core) by an encapsulating matrix (water-soluble agents or insoluble in water) with the production of microcapsules whose size can vary from a few nanometers to several micrometers10. It is a procedure that has been successfully used by cosmetics, pharmaceutical and food industries10. Microencapsulation can be used in the enrichment of food with FO.
In Brazil, there is no food enriched with microencapsulated fish oil(ME FO). Studies of individuals with MS treated with food fortified with ME FO are nonexistent. Available studies on foods enriched with ME OP are scarce and have been conducted with healthy individuals11,12. This fact justifies our study and led to the elaboration of light strawberry jam enriched with ME FO. This study attempted to assess the impact of a hypocaloric diet associated with the consumption of light jam enriched with ME FO on insulin resistance in women with metabolic syndrome.
Patients and Methods
Study group
Adult women (aged 30 to 45 years) diagnosed with MS according to the International Diabetes Federation13 were recruited through advertisements in newspapers. Exclusion criteria were: allergy to strawberry; pregnancy; lactation; using fatty acid supplements; submission to bariatric surgery; and nutritional or pharmacological treatment for the reduction of body weight during the last three months before the trial.
The volunteers were informed about the research and signed an Informed Consent Form, in accordance with resolution 196/96 of the National Health Council. The study protocol was approved by the Committee of Ethics on Research of the Clementino Fraga Filho University Hospital, Federal University of Rio de Janeiro (181/10 Protocol).
Study design
We conducted a randomized single-blind and placebo-controlled clinical trial. The volunteers (n = 30) were distributed into two groups that received jam enriched with ME FO fortnightly (3 g/day, totaling 0.4 g/day of EPA and DHA) or placebo (jam not enriched) for 90 days. We performed blood collection and assessment of clinical, anthropometric, body composition, biochemical and insulin resistance parameters at the beginning (T0) and the end (T90) of the study.
Nutritional treatment
A hypocaloric diet was prescribed by predicting a body mass reduction of 2 kg/month with an estimated total energy value15,16. The macronutrient composition was in accordance with the recommendations of the Ministry of Health that suggests a distribution of 50 to 60% of the total energy value (TEV) of carbohydrates, 15% of TEV od proteins or 0.8% -1g/weight current/day and 25 to 30% in the form of lipids, in which saturated fatty acids correspond to less than 10% of TEV, polyunsaturated fatty acids up to 10% and monoinsaturated fatty acids up to 20%17. We assessed the adhesion to the eating plan through food records evaluated with the Food Processor software (version 12)18. During the 90 days of testing, fortnightly consultations, delivery of jams and nutritional follow-ups of the volunteers were conducted.
Microencapsulation, elaboration and enrichment of light strawberry jam with ME FO
We used the complex coacervation method for microencapsulation of FO19, through which we obtained a paste. Microencapsulation was performed at Mikron Microcapsules Laboratory (São Paulo, Brazil). The percentage of ME FO in the paste was 30%. For the preparation of jam, strawberries purchased on the market, sugar (reduced by 30%), sucralose, pectin and citric acid were used. The ME FO paste and the jam were mixed (ratio 2:1) manually and packed in food packaging kept under refrigeration until delivery. The placebo was strawberry jam without FO enrichment.
For the calculation of the amount to be provided to volunteers per consultation, we considered 16 days (one extra day as safety margin). The women were guided to consume the jam enriched (30 g/day, totaling 3 g/day of ME FO) at breakfast and keep it stored under refrigeration. The participants were instructed to not washing the pots and returning them fortnightly in order to assess jam leftovers.
Clinical, anthropometric and body composition assessments
Blood pressure (BP) was measured by auscultatory method with the use of aneroid sphygmomanometer and stethoscope20. Body mass (kg), height (m)21 and waist circumference22 were measured and the body mass index (BMI) calculated14. Fat and fat-free masses were assessed by the method of electric bioimpedance (BIA 450 bioimpedance analyzer-Biodynamics Corporation, USA). The volunteers were previously guided to the assessment of body composition23.
Blood collection and biochemical assessment
After a 12-hour night fasting, the volunteers were subjected to blood collection using tubes without anticoagulant (Vacutainer tubes,Becton, Dickinson & Company, USA). Aliquots of serum were obtained by centrifugation (4,000 rpm for 15 minutes) and stored at-20 oC until the analysis.
Serum concentrations of triglycerides, total cholesterol and HDL cholesterol were determined by enzymatic-colorimetric method (commercial kit, Bio-Systems), with the reading performed in an A15Automated Analyzer (BioSystems). Serum concentrations of LDL cholesterol were calculated using Friedwald's formula -[LDL - cholesterol = Total Cholesterol - (HDL-Cholesterol + triglyceridemia/5)], only valid for individuals with triglyceridemia < 400 mg/dL24.
Insulinemia was assessed by the radioimmunoassay method (commercial ImmuChem™ Coated Tube kit of MP Biomedicals®, Insulin, CT) using the 2470 WIZARD Automatic Gamma Counter (Perkin-Elmer Inc.). Insulin resistance was estimated by Homeostasis Model Assessment: Insulin Resistance (HOMA-IR)25.
Statistical analysis
The results were expressed as mean ± standard error. We used the Student's f-test and the paired t-test. We also used the Statistical Package for the Social Sciences (SPSS), version 17 andp value < 0.05 was considered significant.
Results
Women with MS selected for the study showed grade 1 obesity (32.6 ± 1.8 kg/m2)14 and their general characteristics can be observed in table I. Adhesion to jam consumption was assessed by observation of leftovers contained in the packs returned at fortnightly consultations. There were no reports of gastrointestinal discomforts.
The average consumption of macronutrients in the groups revealed to be in accordance with the prescriptions; however, the average energy intake was below the prescription (table I). There were no significant differences within and between groups (ME FO and placebo).
Both groups showed significant reduction of body mass, BMI and waist circumference (table II). Placebo group showed a significant reduction in body fat. ME FO group found no difference in body composition. The variation of these data did not differ significantly between the groups. ME FO group had a significant reduction in systolic arterial pressure, as opposed to the placebo group (table II).
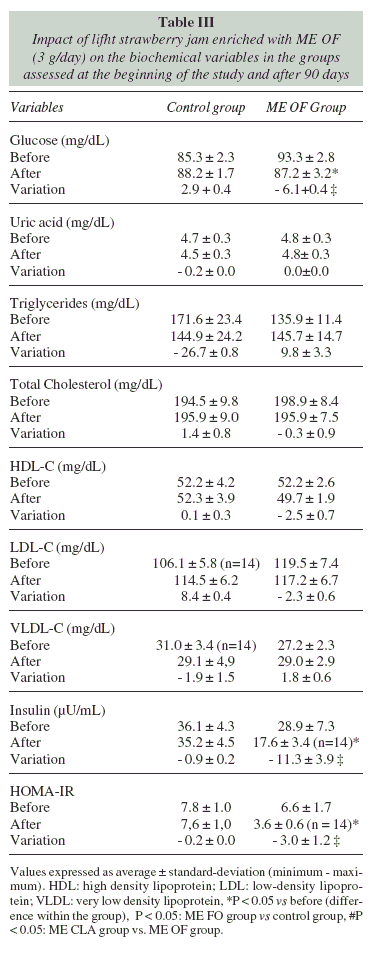
Blood glucose concentrations, insulinemia and HOMA-IR showed significant reduction in the ME FO group (fig. 1); whereas in the placebo group there was no significant reduction of these parameters (table III).
We did not observe significant differences on the lipid profile between the groups at the end of the study. This shows that the hypocaloric diet was also effective in the improvement of these data (table III).
Discussion
This study found that consumption of jam enriched with 3 g/day of ME FO, containing 0.41 g/day of EPA and DHA, resulted in the reduction of blood glucose, insulinemia and HOMA-IR after a 90-day intervention, as opposed to the hypocaloric diet.
Giacco et al.26 supplemented 3.6 g/day of FO (2.1 g of EPA and 1.5 g of DHA) for 90 days and did not observe any effect of FO on reducing blood glucose. The same result was observed by Brandy et al.27 who used 4 g of FO (2.5 g of EPA and DHA). On the other hand, 3 g/day of FO (180 mg of EPA and 120 mg of DHA) were used in a study carried out by Simon et al.28 and they observed increased blood glucose after 90 days.
These different results in blood glucose can occur due to the profile of subjects studied and the amount of EPA and DHA supplemented. Giacco et al.26 assessed overweight individuals, whereas the volunteers in our study suffered from grade 1 obesity.
The effect of FO in blood glucose can be attributed to the activation of PPARy2 by EPA and DHA ligands29. PPARy2 operates in the metabolism of glucose by increasing the expression of genes related to glucose transporter 4 (GLUT4) in the skeletal muscle and has high affinity for insulin, facilitating the absorption of glucose by the cells, thus aiding in reduction in blood glucose30.
Similar to our research, a study carried out by Ramelet al.31 with grade 1 obese patients undergoing a hypocaloric diet associated with 3 g of FO (1.3 g of EPA and DHA) supplementation also found insulinemia reduction after eight weeks. The same result was observed in a study carried out by Krebs et al.32 who supplemented 1 g of FO (1.3 g of EPA and 2.9 g of DHA). On the other hand, Waite et al.33 did not observe change in insulinemia after 60 days.
A study by Simon et al.28 resulted in an increase in HOMA-IR after 90 days of treatment with 3g/day of FO (180 mg of EPA and 120 mg of DHA). Such results are contrary to those found in the literature and our study, probably due to the amount of EPA and DHA supplemented. We can observe that the higher the dose (> 3 g dose) of EPA and DHA supplemented the more deleterious effect on glycidicmetabolism34.
N-3 PUFA can improve IR by several mechanisms, such as reducing the production of pro-inflammatory eicosanoids by inhibiting the phospholipase A2 enzymes35 and increasing the expression of the gene that encodes the adiponectin36, which promotes the oxidation of fatty acids and increases insulin sensitivity in muscles and liver37. This pathway is mediated by receptors of adiponectin leading to activation of protein kinase activated by adenosine monophosphate (AMPK), which is an important modulator of lipid metabolism and glucose. It increases the oxidation of lipids, reducing the concentration of free fatty acids in the plasma and thus improves the absorption of glucose in muscles by increasing their sensitivity to insulin38. In addition, PPARy directly modulates the signaling pathway of insulin transduction in adipose tissue by increasing the expression of intracellular proteins that stimulate the transport of glucose39.
PPARy2 activation by FO has been associated with the reduction of serum concentrations of fatty acids whose increase is related to the IR. This is another mechanism that could explain the reduction of IR by using FO40.
According to data observed in this study, supplementation with ME FO seems to promote greater SAP and DAP reduction than a hypocaloric diet. These results are in accordance with those observed in the literature41. It was demonstrated that adiponectin helps in reducing arterial pressure due to its direct action on the vascular endothelium, promoting endothelium-dependent vasodilatation41. We can observe that the activation of PPARy2 by n-3 PUFA present in the FO has direct effects on the regulation of the vascular structure and protective effect on the endothelium by inhibiting the production of endothelin 1 (ET-1)42. ET-1 is an important vasoconstrictor peptide secreted by endothelial cells in response to insulin. This way, high concentrations of ET-1 are observed in individuals with IR43.
Conclusion
A hypocaloric diet associated with the consumption of jam enriched with ME FO reduced blood glucose, insulinemia and HOMA-IR in women with MS.
References
1. Guzder RN, Gatling W, Mullee MA, Byrne CD. Impact of metabolic syndrome criteria on cardiovascular disease risk in people with newly diagnosed type 2 diabetes. Diabetologia 2006; 49: 49-55. [ Links ]
2. Gang H, Qiao Q, Tuomilehto J, Balkau B, Borch-Johnsen K, Pyorala K. Prevalence of the metabolic syndrome and its relation to all-cause and cardiovascular mortality in nondiabetic European men in women. Arch Intern Med 2004; 164: 1066-76. [ Links ]
3. Oliveira AC, Leite AB, Lima ARV, Vasconcelos SLV. Prevalência de síndrome metabólica em hipertensos de municipio da Zona da Mata Alagoana. Rer Bras Cardiol. 2010; 2395: 270-6. [ Links ]
4. Chien KL, Hsu HC, Sung FC, Su TC, Chen MF, Lee YT. Metabolic syndrome as a risk factor for coronary heart disease and stroke: an 11-year prospective cohort in Taiwan community. Atherosclerosis 2007; 194: 214 -21. [ Links ]
5. Jover A, Corbella E, Munoz A, Millán J, Pintó X, Mangas A, et al. Prevalence of metabolic syndrome and its components in patients with acute coronary syndrome. Rev Esp Cardiol 2011; 64 (7): 579-86. [ Links ]
6. Yamazaki RK, Brito GA, Coelho I, Pequitto DC, Yamaguchi AA, Borghetti G, Schiessel DL, Kryczyk M, Machado J, Rocha RE, Aikawa J. Low fish oil intake improves insulin sensitivity, lipid profile and muscle metaboliMS on insulin resistant MSG-obese rats. Lipids Health Dis. 2011; 28: 10-66. [ Links ]
7. Ruff N. Distribution of α-tocopherol in fillets of turbot (Scophthalmusmaximus) and Atlantic halibut (Hippoglossushippoglossus), following dietary α-tocopheryl acetate supplementation. Aquaculture Nutrition 2004; 10:75-81. [ Links ]
8. Silva C, Ribeiro A, Ferreira D, Veiga F. Administração oral de peptídeos e proteínas: II. Aplicação de métodos de microencapsulação. Rev Bra Ciênc Farm 2003; 39 (1): 1-20. [ Links ]
9. Matioli G, Rodriguez-Maya DB. Microencapsulação do licopeno com ciclodextrinas. Ciênc Tecnol Aliment Campinas 2003; 23: 102-5. [ Links ]
10. Favaro-Trindade CS, Pinho SC, Rocha GA. Revisão: Microencapsulação de ingredientes alimenticios. Braz J Food Technol 2008; 11 (2): 103-12. [ Links ]
11. Higgins S, Carroll YL, O'Brien NM, Morrissey PA. Use of microencapsulated fish oil as a means of increasing n-3 polyunsaturated fatty acid intake. J Hum Nutr Diet 1999; 12: 265-71. [ Links ]
12. Wallace JMW, McCabe AJ, Robson PJ, Keogh MK, Murray CA, Kelly P M, et al. Bioavailability of n-3 polyunsaturated fatty acids (PUFA) in foods enriched with microencapsulated fish oil. Ann Nutr Metab 2000; 44: 157-62. [ Links ]
13. Zimmet P, Magliano D, Matsuzawa Y, Alberti G, Shaw J. The metabolic syndorme: a global public health problem and a newdefinition. J Atheroscler Thromb 2005; 12: 295-300. [ Links ]
14. WHO. World Health Organization. (1998). Obesity: prevention and managing the global epidemic: report of a WHO consultation on obesity. Geneva. [ Links ]
15. Wishnofsky M. Caloric equivalents of gained or lost weight. JAMA 1960; 173 (1): 8. [ Links ]
16. Institute of Medicine. Energy (IOM). In: Dietary Reference Intakes - Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids. Chapter 5. Washington, DC: The National Academic Press, 2002, Part 1,5.1 - 5. [ Links ]114.
17. I Brazilian Guideline for Diagnosis and Treatment of Metabolic Syndrome. Arquivos Brasileiros de Cardiologia- Volume 84, Supplement I, April 2005. [ Links ]
18. Esha Research. (software). Version 12. Salem, USA. (25 November 2010). Retrieved from: <http://www.esha.com/food-prosql> [ Links ].
19. Jouzel B, Pennarun AL, Prost C, Renard D, Poncelet D, Demaimay M. Encapsulation of a lipid precursor, the eicosapentaenoic acid, to study the development of the Crassostreagigas oyster flavours. J Microencapsulation 2003; 20 (1): 35-46. [ Links ]
20. VI Brazilian Guidelines for Hypertension. Rev Bras Hipertens 2010; 17 (1): 1-64. [ Links ]
21. Gibson RS. Principles of nutritional assesMSent. 2nded. New York, Oxford University Press, [ Links ] 2005.
22. Latin-American Consensus on Obesity Report. (1999). Ministry of Health. [ Links ]
23. Heyward VH, Stolarczyk KM. Fundamentos da composição corporal. In: Heyward VH, Stolarczyk KM. Avaliação da composição corporal aplicada. São Paulo: Manole; 2000. p. [ Links ] 2-22.
24. Friedwald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem 1972; 18: 499-502. [ Links ]
25. Matthews DR, Hosker JP, Rudenski BA, Naylor DF, Treacher DF, Turner RC. Homeostasis model assesMSent: insulin resistance and β-cell function from fasting plaMSa glucose and insulin concentrations in man. Diabetologia 1985; 28: 412-9. [ Links ]
26. Giacco R, Cuomo V, Vessby B, Uusitupa M, Hermansen K, Meyer BJ, et al. Fish oil, insulin sensitivity, insulin secretion and glucose tolerance in healthy people: Is there any effect of fish oil supplementation in relation to the type of background diet and habitual dietary intake of n-6 and n-3 fatty acids? Nutrition, Metabolism & Cardiovascular Diseases. 2007; 17: 572-80. [ Links ]
27. Brady LM, Lovegrove SS, Lesauvage SVM, Gower BA, Minihane AM, Williams CM, et al. Increased n-6 polyunsaturated fatty acids do not attenuate the effects of long-chain n-3 polyunsaturated fatty acids on insulin sensitivity or triacylglycerol reduction in Indian Asians. Am J ClinNutr 2004; 79 (6): 983- 91. [ Links ]
28. Simão ANC, Godeny P, Lozovoy MAB, Dichi JB, Dichi I. Efeito dos ácidos graxos n-3 no perfl glicêmico e lipidico, no estresse oxidativo e na capacidade antioxidante total de pacientes com sindrome metabòlica. Arq Bras Endocrinol Metab 2010; 54 (5): 463-9. [ Links ]
29. Jump DB Dietary polyunsaturated fatty acids and regulation of 9 gene transcription. Curr Opin Lipidol 2002; 13 (2): 155-64. [ Links ]
30. Almeida APF, Moura L, Chaves FR, Romaldini JH. Dislipidemias e diabetes mellitus: fisiopatologia e tratamento. Rev Ciênc Méd Campinas 2007; 16 (4- 6): 267-277. [ Links ]
31. Ramel A, Martinez A, Kiely M. Beneficial effects of long-chain n-3 fatty acids included in an energy-restricted diet on insulin resistance in overweight and obese European young adults. Diabetologia. 2008; 51: 1261-8. [ Links ]
32. Krebs JD, Browning LM, McLean NK. Additive benefits of long-chain n-3 polyunsaturated fatty acids and weight-loss in the management of cardiovascular disease risk in overweight hyper-insulinaemic women. Int J Obes 2006; 30: 1535-44. [ Links ]
33. Waite N, Lodge J, Hart K. The impact of fish-oil supplements on insulin sensitivity. J Hum Nutr Diet 2008; 21:402-3. [ Links ]
34. Parra D, Bandarra NM, Kiely M, Thorsdottir I, Martinez JA. Impact of fish intake on oxidative stress when included into a moderate energy-restricted program to treat obesity. Eur J Nutr 2007; 46 (8): 460-7. [ Links ]
35. Perez-Matute P, Perez-Echarri N, Martinez JA, Marti A, Moreno-Aliaga MJ. Eicosapentaenoic acid actions on adiposity and insulin resistance in control and high-fat-fed rats: role of apoptosis, adiponectin and tumour necrosis factor-alpha. Br J Nutr 2007; 97: 389-98. [ Links ]
36. Pighin D, Karabatas L, Rossi A, Chicco A, Basabe JC, Lombardo YB. Fish oil affects pancreatic fat storage, pyruvate dehydrogenase complex activity and insulin secretion in rats fed a sucrose-rich diet. J Nutr 2003; 133 (12): 4095-101. [ Links ]
37. Combs TP, Berg AH, Obici S, Scherer PE, Rossetti L. Endogenous glucose production is inhibited by the adipose-derived protein Acrp30. J Clin Invest 2008; 108: 1875-81. [ Links ]
38. Yamauchi T, Kamon J, Minokoshi Y, Ito Y, Waki H, Uchida S, et al. Adiponectin stimulates glucose utilization and fatty-acid oxidation by activating AMP-activated protein kinase. Nat Med 2008; 8: 1288-95. [ Links ]
39. Yamauchi T, Kamon J, Ito Y, Tsuchida A, Yokomizo T, Kita S, et al. Cloning of adiponectin receptors that mediate antidiabe-ticmetabolic effects. Nature. 2003; 423: 762-9. [ Links ]
40. Miura K, Stamler J, Nakagawa H, Elliott P, Ueshima H, Chan Q, et al. Relationship of dietary linoleic acid to blood pressure. The International Study of Macro-Micronutrients and Blood Pressure Study. Hypertension 2008; 52 (2): 408-14. [ Links ]
41. Ohashi K, Kihara S, Ouchi N, Kumada M, Fujita K, Hiuge A, et al. Adiponectin replenishment ameliorates obesity-related hypertension. Hypertension 2006; 47: 1108-16. [ Links ]
42. Beyer AM, Baumbach GL, Halabi CM, Modrick ML, Lynch CM, Gerhold TD, et al. Interference With PPAR Signaling Causes Cerebral Vascular Dysfunction, Hypertrophy, and Remodeling. Hypertension 2008; 51: 867-71. [ Links ]
43. Bahia L, Aguiar LGK, Villela NR, Bottino D, Bouskela E. O Endotèlio na Sindrome Metabòlica. Arq Bras Endocrinol Metab 2006;50:291-303. [ Links ]
![]() Correspondence:
Correspondence:
Glorimar Rosa.
Instituto de Nutrição Josué de Castro.
Departamento de Nutrição e Dietética.
Av. Carlos Chagas Filho, 373, Bloco J, 2.o andar, sala 25.
Ilha da Cidade Universitária.
CEP: 21941-590 Rio de Janeiro - RJ - Brasil.
E-mail: glorimar@nutricao.ufrj.br
Recibido: 6-VI-2013.
1.a Revisión: 18-VII-2013.
2.a Revisión: 20-I-2014.
Aceptado: 18-II-2014.