Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Nutrición Hospitalaria
versión On-line ISSN 1699-5198versión impresa ISSN 0212-1611
Nutr. Hosp. vol.33 no.3 Madrid may./jun. 2016
https://dx.doi.org/10.20960/nh.282
TRABAJO ORIGINAL / Otros
Fish consumption and risk of breast cancer: meta-analysis of 27 observational studies
Consumo de pescado y riesgo de cáncer de mama: metaanálisis de 27 estudios observacionales
Wu Zhihui, Yan Weihua, Wu Zupei and Huang Jinlin
The 2nd Surgical Department. Shunde Hospital of Traditional Chinese Medicine. Foshan, China
ABSTRACT
Objectives: The association between fish consumption and the risk of breast cancer has not been established yet. Results from epidemiological studies are inconsistent. We conducted a meta-analysis to examine the association between fish consumption and the risk of breast cancer.
Methods: We identified eligible studies in Medline and EMBASE up to February 2015 and the reference lists of original studies and review articles on this topic. Summary relative risks with their 95% confidence intervals were calculated with a random-effects model.
Results: We identified 27 studies eligible for analysis. The summary relative risk of breast cancer for the highest consumption of fish compared with the lowest was 0.96 (95% CI = 0.87-1.07), with evidence of heterogeneity (Q = 69.09, p < 0.001, I2 = 68.0%). Four studies investigated lean fish consumption and revealed that there was a small increase in the risk of breast cancer (summary RR = 1.09, 95% CI = 1.00-1.19). As only four studies were included in the subgroup analysis, results must be interpreted with caution.
Conclusions: The overall current literature on fish consumption and the risk of breast cancer suggested no association. Further well-designed prospective studies are needed to explore fish consumption in relation to breast cancer risk.
Key words: Breast cancer. Fish consumption. Systematic review. Meta-analysis. Relative risks.
RESUMEN
Objetivos: hasta el momento, no se ha establecido una asociación entre el consumo de pescado y el riesgo de padecer cáncer de mama. Los resultados derivados de estudios epidemiológicos son inconsistentes. En este caso, llevamos a cabo un metaanálisis para examinar la relación entre el consumo de pescado y el riesgo de cáncer de mama.
Métodos: identificamos diversos estudios aptos en Medline y EMBASE hasta febrero de 2015, así como las referencias bibliográficas de estudios originales y artículos revisados sobre este tema. Se calculó el resumen de riesgo relativo con un intervalo de confianza del 95% mediante un modelo de efectos aleatorios.
Resultados: se identificaron 27 estudios aptos para análisis. El riesgo relativo de cáncer de mama asociado al mayor consumo de pescado respecto al menor fue de 0,96 (95% CI = 0,87-1,07), con manifiesta heterogeneidad (Q = 69,09, p < 0,001, I2 = 68,0%). Cuatro estudios investigaron el consumo de pescado magro y revelaron un pequeño incremento en cuanto al riesgo de cáncer de mama (resumen RR = 1,09, 95% CI = 1,00-1,19). Dado que solo fueron incluidos cuatro estudios en el análisis del subgrupo, los resultados han de ser interpretados con cautela.
Conclusiones: En general, la literatura actual sobre consumo de pescado y riesgo de padecer cáncer de mama sugiere que no existe asociación entre ambos. Es necesario llevar a cabo otros estudios prospectivos con un diseño adecuado para explorar dicha relación.
Palabras clave: Cáncer de mama. Consumo de pescado. Revisión sistemática. Metaanálisis. Riesgo relativo.
Introduction
Breast cancer is the most common malignancy afflicting women and is one of the leading causes of cancer mortality (1). A recent analysis by the National Institutes of Health has shown that the national medical cost of breast cancer care is estimated to be $16.50 billion in 2010 and will be $20.5 billion in 2020, accounting for the largest part of all cancer costs (2). Primary prevention of breast cancer is, therefore, very important.
The potential role that lifestyle plays as a cause of breast cancer remains an active area of research. Cigarette smoking is potentially linked to breast cancer (3). Several studies have suggested that physical activity, alcohol consumption (4), and low level of vitamin B6 intake (5) are associated with increased risk of breast cancer. A report published in 2008 by the World Cancer Research Fund and the American Institute for Cancer Research on the relationship between diet and cancer suggested that the consumption of certain types of food may be directly associated to the development of breast cancer (6).
Experimental evidence indicated that n-3 polyunsaturated fatty acids, which occur at high level in fish, have protective effects against some common cancers. The underlying mechanisms included suppression of neoplastic transformation, cell growth inhibition and enhanced apoptosis, and antiangiogenicity (7,8). However, epidemiological studies assessing the relation between breast cancer and fish consumption are inconclusive. Some studies showed a decrease in risk (9,10), and some found no association (11,12), while others revealed an increased risk associated to high consumption of fish (13,14). We therefore performed a systematic review and meta-analysis to assess the association between fish consumption and breast cancer risk.
Materials and methods
DATA SOURCES AND SEARCHES
Two authors independently performed a literature search using Medline and EMBASE database up to February 1st 2015. We searched the studies with the following text words and/or Medical Subject Heading (MeSH) terms: "diet" or "fish", "breast", "cancer" or "tumor" or "neoplasm" or "carcinoma". Furthermore, we reviewed the reference lists of retrieved articles to search for more studies.
STUDY SELECTION
To be included in our meta-analysis, the following criteria had to be met. First, the study had to have a case-control or cohort study design. Second, the exposure of interest had to be fish consumption. Third, the number of breast cancer cases and controls had to be reported. Fourth, the relative risks (RRs) or odds ratios (ORs) with their corresponding 95% confidence interval (CI) for the highest versus non/lowest level of fish consumption had to be reported. Two authors evaluated all the studies retrieved from the databases independently. Any discrepancies between the two reviewers were solved by joint reevaluation of the manuscript. In case of multiple publications from the same study, the most relevant was selected, using the other publications to clarify methodology or characteristics of the population.
DATA EXTRACTION AND QUALITY ASSESSMENT
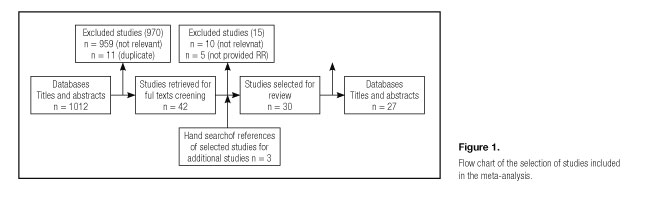
Three authors evaluated independently all of the studies retrieved according to the pre-specified selection criteria. Any discrepancies between reviewers were addressed by a joint reevaluation of the original article. We identified 30 potentially relevant articles concerning fish consumption and breast cancer risk (9,38). After duplicated studies were excluded, the remaining publications in the meta-analysis of fish consumption and breast cancer included 27 articles: eight cohort studies and 18 case-control studies. We used a standardized protocol and reporting form to collect the following data from each publication: reference (first author, year of publication), geographic location, study design, source of control (population-based or hospital-based), menstrual conditions, sample size, fish consumption level, effect estimates with 95% CI, and covariates adjusted in the statistical analysis. The quality of each study was assessed independently by two reviewers using the Newcastle-Ottawa Scale (NOS). The NOS consists of three parameters of quality: selection, comparability, and outcome (cohort studies) or exposure (case-control studies). The NOS assigns a maximum of four points for selection, a maximum of two points for comparability, and a maximum of three points for exposure or outcome. Any discrepancies between reviewers were addressed by a joint reevaluation of the original article.
STATISTICAL ANALYSIS
Study-specific RRs with corresponding 95% CI for the highest versus non/lowest fish consumption levels were extracted. We ignored the distinction between the various estimates of RR (i.e., OR, rate ratio, hazard ratio) and all measures were interpreted as RR for simplicity. When several estimates were available, we used the one that was adjusted for most variables. Q and Higgins I2 statistics were used to examine heterogeneity not only among studies but also between the subgroups included in this meta-analysis (39,40). For the Q statistics, p < 0.10 indicated statistically significant heterogeneity (39). We defined statistical significance as p < 0.10 rather than the conventional level of 0.05 due to the low power of this test (41). I2 values lie between 0% (no observed heterogeneity) and 100% (maximal heterogeneity); thus, an I2 value greater than 50% may be considered to represent substantial heterogeneity (40). Risk estimates were calculated using a random-effects model, incorporating both within- and between-study variability (39). Funnel plots and statistical tests for funnel plot asymmetry were performed to test evidence of publication bias. Meta-analyses were carried out using Review Manager version 5.0 software (Copenhagen: The Nordic Cochrane Centre; The Cochrane Collaboration, 2008). A two-tailed p-value less than 0.05 was considered to be significant.
Result
STADY CHARACTERISTICS
Twenty-seven articles that met our inclusion criteria in this meta-analysis were published between 1986 and 2009. There were 8 cohort studies and 19 case-control studies. One study provided RR from both population-based and hospital-based controls (33). Four studies presented results for pre- and postmenopausal women separately (30-33). So, we could use the four studies for subgroup analysis of menstrual conditions (Fig. 1). The main characteristics of the included studies were summarized in table I.
META-ANALYSIS
As the results of four studies were presented separately for pre- and postmenopausal women without a pooled analysis with total women (30-33), 23 studies were selected for analysis. Significant heterogeneity was found in the results across the 23 studies (Q = 69.09, p < 0.001, I2 = 68.0%). The summary RR for the 23 studies showed that high fish consumption was not associated with a reduction in breast cancer risk (summary RR = 0.96, 95% CI = 0.87-1.07) (Fig. 2).
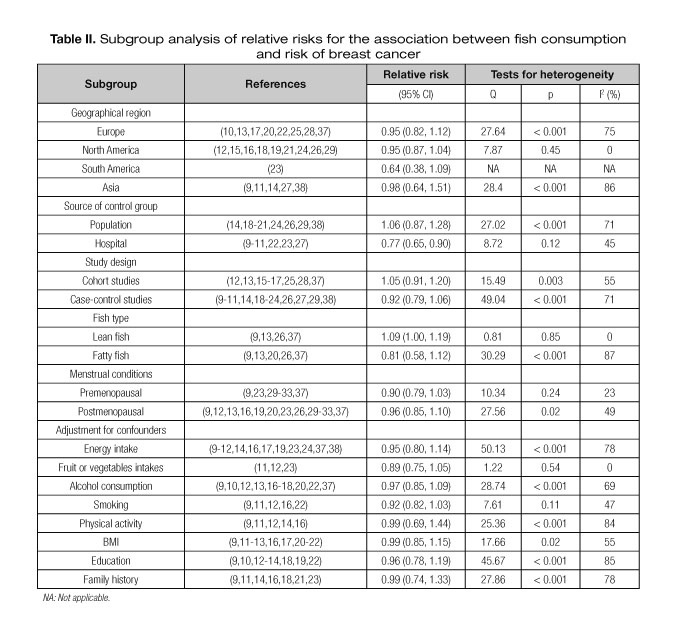
Significant heterogeneity was found among the 15 case-control studies (Q = 49.04, p < 0.001, I2 = 71.0%) and the 8 cohort studies (Q = 15.49, p = 0.03, I2 = 55.0%). Similar to the results from all studies combined, there was no significant association between fish consumption and breast cancer risk either in the case-control (summary RR = 0.92, 95% CI = 0.79-1.06) or cohort (summary RR = 1.05, 95% CI = 0.91-1.20) studies (Table II). The difference between study design strata was not significant (Q = 1.68, p = 0.19, I2 = 40.5%).
In case-control studies, the design could be further divided into hospital-based and population-based studies. By stratified analysis of two subgroups, significant heterogeneity was found within population-based studies (Q = 49.04, p < 0.001, I2 = 71.0%), but not among the hospital-based case-control studies (Q = 8.72, p = 0.12, I2 = 43.0%). The summary RR among hospital-based case-control studies showed a significant reduction in the risk of breast cancer with high fish consumption (summary RR = 0.77, 95% CI = 0.65-0.90) (Table II).
In stratified analysis by geographical locations (Europe, North America, South America, and Asia), we found no association between fish consumption and breast cancer risk for any of the geographical regions (Europe, summary RR = 0.95, 95% CI = 0.82-1.12; North America, summary RR = 0.95, 95% CI = 0.87-1.04; South America, summary RR = 0.64, 95% CI = 0.38-1.09; Asia, summary RR = 0.98, 95% CI = 0.64-1.51) (Table II).
We also limited the meta-analysis to studies that controlled for total energy input, fruit or vegetables intake. Regarding the 12 studies that controlled for energy input, significant heterogeneity was found among them (Q = 50.13, p < 0.001, I2 = 78.0%), and no significant association was found between fish consumption and breast cancer (summary RR = 0.95, 95% CI = 0.8-1.14). Among the four studies that controlled for fruit or vegetables intake, no significant heterogeneity was found (Q = 1.12, p < 0.54, I2 = 0%), and no significant association between fish consumption and breast cancer was observed (summary RR = 0.89, 95% CI = 0.75-1.05) (Table II).
Alcohol consumption, smoking, physical activity, education, family history of breast cancer and body mass index (BMI) are important confounders for breast cancer risk. When we limited the meta-analysis to studies that controlled for these potential risk factors, we also found no association between fish consumption and breast cancer risk (Table II).
Four studies provided available data for subgroup analysis about lean fish. No significant heterogeneity was found among the four studies (Q = 0.81, p = 0.85, I2 = 0%), and there was a small increase effect of lean fish consumption on breast cancer (summary RR = 1.09, 95% CI = 1.00-1.19). Five studies provided available data for subgroup analysis about fatty fish. Among the five studies, significant heterogeneity was observed (Q = 30.29, p < 0.001, I2 = 87%), and the association between fish consumption and breast cancer was not statistically significant (summary RR = 0.81, 95% CI = 0.58-1.12) (Table II).
Four studies provided RR with corresponding CI for subgroup analysis about menstrual conditions. No significant heterogeneity was found (Q = 10.34, p = 0.24, I2 = 23%) among the eight studies, and we also found no association between fish consumption and breast cancer risk in premenopausal women (summary RR = 0.90, 95% CI = 0.79-1.03). Among the four studies providing RR about postmenopausal women, significant heterogeneity was observed (Q = 27.56, p < 0.02, I2 = 49%), and the association between fish consumption and breast cancer was not statistically significant in postmenopausal women (summary RR = 0.96, 95% CI = 0.85-1.10).
PUBLICATION BIAS
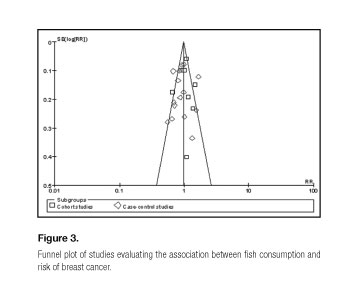
The shape of the funnel plots for studies on the association between fish consumption and breast cancer risk seemed symmetrical, indicating no publication bias (Fig. 3).
Discussion
Fish consumption has long been thought to play a role in the development of breast cancer, though evidence from the present studies is inconclusive. This present study summarized the evidence to date regarding the association between fish consumption and breast cancer risk. Overall, the summary RR for all of the studies suggested no significant association between fish consumption and breast cancer risk. There was significant heterogeneity among the studies. Although the pooled analysis from the hospital-based case-control studies suggested a small reduction in risk, the results from the population-based case-control and cohort studies were null.
The association between fish consumption and breast cancer is biologically plausible. Consumption of fish provides unsaturated essential fatty acids, certain vitamins and minerals. In a mouse model system, researchers have found that supplementing the diet of tumor-bearing mice or rats with n-3 fatty acids or inorganic selenium can slow the growth of various types of cancers (42,43). Larsson highlighted current knowledge on the potential mechanisms of the n-3 fatty acids' anti-carcinogenic actions. These included suppression of arachidonic acid-derived eicosanoid biosynthesis; influence on transcription factor activity, gene expression, and signal transduction; alteration of estrogen metabolism; increased or decreased production of free radicals and reactive oxygen species; and effect on insulin sensitivity and membrane fluidity (44).
There may be reasons for the discrepancies observed between the studies included. First, the protective effect of fish consumption on breast cancer risk may be counterbalanced by the negative effect of contaminants. Among contaminants found in fish are mercury (45), polychlorinated biphenyls (46), organochlorine residues, and other chemicals. These chemicals have high toxicity and carcinogenic potency, and a few epidemiological studies suggested that pesticides and some of these chemicals may be related to breast cancer risk (47,48).
Second, existing reports suggested n-6 fatty acids as pro-oncogenic and n-3 fatty acids as anti-oncogenic factors. N-6 fatty acids from fish oils induced growth of human breast cancer cells (49), and postmenopausal breast cancer was positively associated with high intakes of n-6 fatty acids (50). Fresh water fish contain lower levels of n-3 fatty acids but higher levels of n-6 fatty acids than marine fish. Most of the studies included in our meta-analysis, however, did not specify what type of fish was consumed. Third, variation in cooking methods across study populations on these studies may have contributed to the inconsistent findings. Heterocyclic amines (HA) and polycyclic aromatic hydrocarbons (PAH) formed during cooking fish at high temperatures may be one of the reasons. Four, self-reported dietary intake (especially via food frequency questionnaire) is notoriously poor and plagued by problems of random error and systematic error associated with participant characteristics.
We tried to carry out an analysis stratified by adjustment for confounding factors, smoking, alcohol consumption, physical activity, BMI, etc. However, we also found no association between fish consumption and breast cancer risk. Data from individual studies suggested that the association between fish consumption and the risk of breast cancer was stronger in premenopausal women than in postmenopausal women (51,52). It is also plausible that diet has a stronger impact on breast cancer risk during early adult life than later in life. However, the results from our analysis demonstrated no relationship between fish consumption and breast cancer risk both in pre- and postmenopausal women. Most of the contaminants accumulate in the fat; therefore, contaminants are more likely present in fatty fish. Because these agents accumulate in fat tissue, one would expect the elevated risk to be more pronounced with higher consumptions of fatty fish compared with lean fish, which was not the case in our study. Contrarily, there was a small increase effect of lean fish consumption on breast cancer, but not of fatty fish consumption. An explanation of this discrepancy could be that lean fish contain fewer contaminants, but the level of n-3 fatty acids on lean fish was lower than that on fatty fish. Only four studies were included in the subgroup analysis, thus results must be interpreted with caution.
Our meta-analysis has several strengths:
1. Studies were included after a comprehensive and systematic search of the literature by using an extensive search strategy.
2. The majority of the studies included evaluated multiple confounders including age, smoking, alcohol consumption, BMI, etc.
3. With available evidence and enlarged number of studies to date, we have enhanced statistical power to detect any associations between fish consumption and breast cancer risk.
Our meta-analysis has limitations that affect interpretation of the true results. First, 15 of 21 studies in this meta-analysis used a case-control design, which was more susceptible to recall and selection biases than a cohort design. On the other hand, cohort studies may be affected by detection bias. Second, there is substantial heterogeneity across studies. Heterogeneity was likely due to the variation in exposure definitions, exposure ranges, fish consumption assessment methods, and population characteristics between studies. Methods and units for measuring fish consumption varied across studies. Third, unmeasured or uncontrolled confounding inherited from original studies is a concern in this meta-analysis. Most risk estimates were derived from multivariable models, but individual studies did not adjust for potential confounding factors in a consistent way. Four, we included only those studies that were published in English. This is mainly because it is difficult for the authors to interpret all the data that are available in different languages.
In summary, from the present meta-analysis we still cannot draw the conclusion that fish consumption has preventive effects on breast cancer. Given the small number of cohort studies included in this meta-analysis, further prospective cohort studies with larger sample size, well-controlled for confounding factors, and more accurate assessment of fish consumption are needed to affirm the effect of fish on breast cancer.
Acknowledgments
ZP.W conceived and designed the study. ZH.W and WH.Y performed a literature search and identified eligible studies. ZH.W, WH.Y and JL.H extracted data from retrieved studies. ZH.W carried out statistical analysis and interpreted results. The authors do not have any possible conflicts of interest. All drafts of the reports were written by ZH.W. All authors read and approved the final paper.
References
1. Hortobagyi GN, De la Garza SJ, Pritchard K, Amadori D, Haidinger R, Hudis CA, et al. The global breast cancer burden: Variations in epidemiology and survival. Clin Breast Cancer 2005;6:391-401. [ Links ]
2. Mariotto AB, Yabroff KR, Shao Y, Feuer EJ, Brown ML. Projections of the cost of cancer care in the United States: 2010-2020. J Natl Cancer Inst 2011;103:117-28. [ Links ]
3. Lynch HT, Lynch JF. Cigarette smoking and breast cancer risk. Womens Health (Lond Engl) 2011;7:413-6. [ Links ]
4. Poli A, Marangoni F, Visioli F. Alcohol consumption and breast cancer risk. JAMA 2012;307:666. [ Links ]
5. Zhang SM, Willett WC, Selhub J, Hunter DJ, Giovannucci EL, Holmes MD, et al. Plasma folate, vitamin B6, vitamin B12, homocysteine, and risk of breast cancer. J Natl Cancer Inst 2003;95:373-80. [ Links ]
6. Wiseman M. The second world cancer research fund/American Institute for Cancer Research expert report. Food, nutrition, physical activity, and the prevention of cancer: A global perspective. Proc Nutr Soc 2008;67:253-6. [ Links ]
7. Fernández E, Chatenoud L, La Vecchia C, Negri E, Franceschi S. Fish consumption and cancer risk. Am J Clin Nutr 1999;70:85-90. [ Links ]
8. Rose DP, Connolly JM. Omega-3 fatty acids as cancer chemopreventive agents. Pharmacol Ther 1999;83:217-44. [ Links ]
9. Kim J, Lim SY, Shin A, Sung MK, Ro J, Kang HS, et al. Fatty fish and fish omega-3 fatty acid intakes decrease the breast cancer risk: A case-control study. Bmc Cancer 2009;9:216. [ Links ]
10. Franceschi S, Favero A. The role of energy and fat in cancers of the breast and colon-rectum in a southern European population. Ann Oncol 1999;10(Suppl. 6):61-3. [ Links ]
11. Zhang CX, Ho SC, Chen YM, Lin FY, Fu JH, Cheng SZ. Meat and egg consumption and risk of breast cancer among Chinese women. Cancer Causes Control 2009;20:1845-53. [ Links ]
12. Folsom AR, Demissie Z. Fish intake, marine omega-3 fatty acids, and mortality in a cohort of postmenopausal women. Am J Epidemiol 2004;160:1005-10. [ Links ]
13. Stripp C, Overvad K, Christensen J, Thomsen BL, Olsen A, Moller S, et al. Fish intake is positively associated with breast cancer incidence rate. J Nutr 2003;133:3664-9. [ Links ]
14. Dai Q, Shu XO, Jin F, Gao YT, Ruan ZX, Zheng W. Consumption of animal foods, cooking methods, and risk of breast cancer. Cancer Epidemiol Biomarkers Prev 2002;11:801-8. [ Links ]
15. Stampfer MJ, Willett WC, Colditz GA, Speizer FE. Intake of cholesterol, fish and specific types of fat in relation to risk of breast cancer. Amer Oil Chemists Society; 1987. p. 248-52. [ Links ]
16. Fung TT, Hu FB, Holmes MD, Rosner BA, Hunter DJ, Colditz GA, et al. Dietary patterns and the risk of postmenopausal breast cancer. Int J Cancer 2005;116:116-21. [ Links ]
17. Lund E, Engeset D, Alsaker E, Skeie G, Hjartaker A, Lundebye AK, et al. Cancer risk and salmon intake. Science 2004;305:477-8. [ Links ]
18. McElroy JA, Kanarek MS, Trentham-Dietz A, Robert SA, Hampton JM, Newcomb PA, et al. Potential exposure to PCBS, DDT, and PBDES from sport-caught fish consumption in relation to breast cancer risk in Wisconsin. Environ Health Perspect 2004;112:156-62. [ Links ]
19. Shannon J, Cook LS, Stanford JL. Dietary intake and risk of postmenopausal breast cancer (United States). Cancer Causes Control 2003;14:19-27. [ Links ]
20. Terry P, Rohan TE, Wolk A, Maehle-Schmidt M, Magnusson C. Fish consumption and breast cancer risk. Nutr Cancer 2002;44:1-6. [ Links ]
21. Gertig DM, Hankinson SE, Hough H, Spiegelman D, Colditz GA, Willett WC, et al. N-acetyl transferase 2 genotypes, meat intake and breast cancer risk. Int J Cancer 1999;80:13-7. [ Links ]
22. Fernandez E, Chatenoud L, La Vecchia C, Negri E, Franceschi S. Fish consumption and cancer risk. Am J Clin Nutr 1999;70:85-90. [ Links ]
23. De Stefani E, Ronco A, Mendilaharsu M, Guidobono M, Deneo-Pellegrini H. Meat intake, heterocyclic amines, and risk of breast cancer: A case-control study in Uruguay. Cancer Epidemiol Biomarkers Prev 1997;6:573-81. [ Links ]
24. Toniolo P, Riboli E, Shore RE, Pasternack BS. Consumption of meat, animal products, protein, and fat and risk of breast cancer: A prospective cohort study in New York. Epidemiology 1994;5:391-7. [ Links ]
25. Lund E, Bonaa KH. Reduced breast cancer mortality among fishermen's wives in Norway. Cancer Causes Control 1993;4:283-7. [ Links ]
26. Goodman MT, Nomura AM, Wilkens LR, Hankin J. The association of diet, obesity, and breast cancer in Hawaii. Cancer Epidemiol Biomarkers Prev 1992;1:269-75. [ Links ]
27. Kato I, Miura S, Kasumi F, Iwase T, Tashiro H, Fujita Y, et al. A case-control study of breast cancer among Japanese women: With special reference to family history and reproductive and dietary factors. Breast Cancer Res Treat 1992;24:51-9. [ Links ]
28. Vatten LJ, Solvoll K, Loken EB. Frequency of meat and fish intake and risk of breast cancer in a prospective study of 14,500 Norwegian women. Int J Cancer 1990;46:12-5. [ Links ]
29. Hislop TG, Coldman AJ, Elwood JM, Brauer G, Kan L. Childhood and recent eating patterns and risk of breast cancer. Cancer Detect Prev 1986;9:47-58. [ Links ]
30. Ambrosone CB, Freudenheim JL, Sinha R, Graham S, Marshall JR, Vena JE, et al. Breast cancer risk, meat consumption and N-acetyltransferase (NAT2) genetic polymorphisms. Int J Cancer 1998;75:825-30. [ Links ]
31. Hirose K, Tajima K, Hamajima N, Inoue M, Takezaki T, Kuroishi T, et al. A large-scale, hospital-based case-control study of risk factors of breast cancer according to menopausal status. Jpn J Cancer Res 1995;86:146-54. [ Links ]
32. Lee HP, Gourley L, Duffy SW, Esteve J, Lee J, Day NE. Risk factors for breast cancer by age and menopausal status: A case-control study in Singapore. Cancer Causes Control 1992;3:313-22. [ Links ]
33. Mannisto S, Pietinen P, Virtanen M, Kataja V, Uusitupa M. Diet and the risk of breast cancer in a case-control study: Does the threat of disease have an influence on recall bias? J Clin Epidemiol 1999;52:429-39. [ Links ]
34. Lee HP, Gourley L, Duffy SW, Esteve J, Lee J, Day NE. Dietary effects on breast-cancer risk in Singapore. Lancet 1991;337:1197-200. [ Links ]
35. Favero A, Parpinel M, Franceschi S. Diet and risk of breast cancer: Major findings from an Italian case-control study. Biomed Pharmacother 1998;52:109-15. [ Links ]
36. Franceschi S, Favero A, La Vecchia C, Negri E, Dal Maso L, Salvini S, et al. Influence of food groups and food diversity on breast cancer risk in Italy. Int J Cancer 1995;63:785-9. [ Links ]
37. Engeset D, Alsaker E, Lund E, Welch A, Khaw KT, Clavel-Chapelon F, et al. Fish consumption and breast cancer risk. The European prospective investigation into cancer and nutrition (epic). Int J Cancer 2006;119:175-82. [ Links ]
38. Shannon J, Ray R, Wu C, Nelson Z, Gao DL, Li W, et al. Food and botanical groupings and risk of breast cancer: A case-control study in Shanghai, China. Cancer Epidemiol Biomarkers Prev 2005;14:81-90. [ Links ]
39. DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials 1986;7:177-88. [ Links ]
40. Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ 2003;327:557-60. [ Links ]
41. Hedges LV, Pigott TD. The power of statistical tests in meta-analysis. Psychol Methods 2001;6:203-17. [ Links ]
42. Reddy BS, Sugie S, Maruyama H, Marra P. Effect of dietary excess of inorganic selenium during initiation and postinitiation phases of colon carcinogenesis in F344 rats. Cancer Res 1988;48:1777-80. [ Links ]
43. Rose DP, Connolly JM. Effects of dietary omega-3 fatty acids on human breast cancer growth and metastases in nude mice. J Natl Cancer Inst 1993;85:1743-7. [ Links ]
44. Larsson SC, Kumlin M, Ingelman-Sundberg M, Wolk A. Dietary long-chain n-3 fatty acids for the prevention of cancer: A review of potential mechanisms. Am J Clin Nutr 2004;79:935-45. [ Links ]
45. Sanzo JM, Dorronsoro M, Amiano P, Amurrio A, Aguinagalde FX, Azpiri MA. Estimation and validation of mercury intake associated with fish consumption in an epic cohort of Spain. Public Health Nutr 2001;4:981-8. [ Links ]
46. Sjodin A, Hagmar L, Klasson-Wehler E, Bjork J, Bergman A. Influence of the consumption of fatty Baltic sea fish on plasma levels of halogenated environmental contaminants in Latvian and Swedish men. Environ Health Perspect 2000;108:1035-41. [ Links ]
47. Laden F, Hunter DJ. Environmental risk factors and female breast cancer. Annu Rev Public Health 1998;19:101-23. [ Links ]
48. Rojas E, Herrera LA, Poirier LA, Ostrosky-Wegman P. Are metals dietary carcinogens? Mutat Res 1999;443:157-81. [ Links ]
49. Hammamieh R, Chakraborty N, Miller SA, Waddy E, Barmada M, Das R, et al. Differential effects of omega-3 and omega-6 fatty acids on gene expression in breast cancer cells. Breast Cancer Res Treat 2007;101:7-16. [ Links ]
50. Wirfalt E, Mattisson I, Gullberg B, Johansson U, Olsson H, Berglund G. Postmenopausal breast cancer is associated with high intakes of omega-6 fatty acids (Sweden). Cancer Causes Control 2002;13:883-93. [ Links ]
51. Shin MH, Holmes MD, Hankinson SE, Wu K, Colditz GA, Willett WC. Intake of dairy products, calcium, and vitamin D and risk of breast cancer. J Natl Cancer Inst 2002;94:1301-11. [ Links ]
52. Lin J, Manson JE, Lee IM, Cook NR, Buring JE, Zhang SM. Intakes of calcium and vitamin D and breast cancer risk in women. Arch Intern Med 2007;167:1050-9. [ Links ]
![]() Correspondence:
Correspondence:
Wu Zupei.
The 2nd Surgical Department.
Shunde Hospital of Traditional Chinese Medicine.
Jinsha Road, Daliang Block, Shunde District.
528300 Foshan, China
e-mail: wuzhihuifs@163.com
Received: 26/01/2016
Accepted: 07/03/2016














.jpg)
.jpg)
.jpg)