Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Nutrición Hospitalaria
versión On-line ISSN 1699-5198versión impresa ISSN 0212-1611
Nutr. Hosp. vol.33 no.4 Madrid jul./ago. 2016
https://dx.doi.org/10.20960/nh.402
REVISIÓN
Effects of a nutritional intervention in a fast-track program for a colorectal cancer surgery: systematic review
Efectos de la intervención nutricional en los programas fast-track en cirugía de cáncer colorrectal: revisión sistemática
Carmina Wanden-Berghe1,2, Javier Sanz-Valero2, Antonio Arroyo-Sebastián3, Kamila Cheikh-Moussa1,2 and Pedro Moya-Forcen3
1Hospital General Universitario de Alicante. ISABIAL-FISABIO. Alicante, Spain.
2Universidad Miguel Hernández de Elche. ISABIAL-FISABIO. Alicante, Spain.
3Hospital General Universitario de Elche. Alicante, Spain
ABSTRACT
Background and aim: Preoperative nutritional status (NS) has consequences on postoperative (POSTOP) recovery. Our aim was to systematically review the nutritional interventions (NI) in fast-track protocols for colorectal cancer surgery and assess morbidity-mortality and patient's recovery.
Method: Systematic review of scientific literature after consulting bibliographic databases: Medline, The Cochrane Library, Scopus, Embase, Web of Science, Institute for Scientific Information, Latin American and Caribbean Health Sciences Literature, The Cumulative Index to Nursing and Allied Health Literature. MeSH Descriptors: "colorectal surgery", "fast-track", "perioperative care", "nutrition therapy" and "enhanced recovery programme". Filters: "humans", adult (19+ years) and "clinical trial". Variables POSTOP outcomes: bowel recovery (BR), hospital stay (HS), complications and death.
Results: Selected studies, 27, had good or excellent methodological quality. From 25 to 597 patients were included. Aged between 16-94 years, men were predominant in 66.6%. NS was evaluated in 13 studies; 7 by body mass index while one by subjective global assessment. One presented POSTOP data. Fast-track groups had solids, liquids or supplements (SS) in prior 2-8 hours. SS were high in carbohydrates, immune-nutrients and non-residue. Free liquids, solids and SS intake was allowed in POSTOP. Half traditional groups fasted between 3-12 hours and resumed POSTOP food intake progressively.
Conclusions: Fast-track groups had early BR (p < 0.01). Traditional groups had more infections episodes, deaths and a longer HS. Great variability between NI but had a common item; early intake. Although was seen patient's recovery. Future studies with detailed NI characteristics are need. Nutritional status must be assessed for a higher acknowledgement of NI impact.
Key words: Fast-track. Colorectal surgery. Perioperative care. Nutrition therapy.
RESUMEN
Introducción y objetivo: el estado nutricional (NS) preoperatorio tiene consecuencias sobre la recuperación postoperatoria (POSTOP). El objetivo fue revisar sistemáticamente las intervenciones nutricionales (NI) en los protocolos de fast-track en la cirugía de cáncer colorrectal y evaluar la morbilidad-mortalidad y la recuperación del paciente.
Método: revisión sistemática de la literatura científica previa consulta a las bases de datos bibliográficas: Medline, Cochrane Library, Scopus, Embase, Web of Science, Latin American and Caribbean Health Sciences Literature (LILACS), The Cumulative Index to Nursing and Allied Health Literature (CINAHL). Descriptores MeSH: "colorectal surgery", "fast-track", "perioperative care", "nutrition therapy" and "enhanced recovery programme". Filtros: "humans", "adult (19+ years)" and "clinical trial". Variables resultados después de la operación: recuperación del intestino (BR), estancia hospitalaria (HS), complicaciones y la muerte.
Resultados: los 27 estudios seleccionados tenían buena o excelente calidad metodológica. Incluían desde 25 a 597 pacientes, con edades comprendidas entre 16-94 años; los hombres fueron predominantes en el 66,6%. El estado nutricional se evaluó en 13 estudios; 7 por el índice de masa corporal, mientras que uno lo fue por la evaluación subjetiva general. Uno de ellos presentó datos después de la operación. Los grupos fast-track ingirieron, líquidos o suplementos (SS) en 2-8 horas antes. SS contenían altas cantidades de hidratos de carbono, inmunonutrientes y sin-residuos. En POSTOP se administraron líquidos, sólidos y SS. Los grupos tradicionales estuvieron en ayunas entre 3-12 horas y se reanudó la ingesta de alimentos progresivamente.
Conclusiones: los grupos fast-track presentaron BR temprana (p < 0,01), los tradicionales tuvieron más infecciones, muertes y un HS más larga. Se observó gran variabilidad en las NI, pero había un punto común: ingesta temprana. A pesar de que se observó una recuperación del paciente, se necesitan futuros estudios con características de la NI más detalladas. Se debe evaluar el NS para poder reconocer el estado nutricional para un mayor reconocimiento del impacto NI.
Palabras clave: Fast-track. Cirugía colorrectal. Atención perioperativa. Terapia nutricional.
Introduction
In the last few years the patients who underwent a surgery for colorectal cancer had experimented important beneficial effects derived of the advances in the fields of anesthesia, minimally invasive surgery and perioperative care and used as a whole. The Multimodal Rehabilitation (MMR) or fast-track (FT) surgery has been an initiative coordinated to combine educating patient before surgery, stress reduction by new anesthetics, analgesic and pharmacologic techniques, minimal invasive surgery and the revision of the fundamental postoperative care principles (use of tubes, drains, catheters, monitoring devices, early oral nutrition, mobilization, etc.) in order to define an active perioperative multimodal rehabilitation program. This concept is based on the combination of different unimodal interventions evidence-based for the creation of multimodal care "packages" that allow the achievement of a synergic or additive effect to enhance patient's recovery (1).
A meta-analysis published in October of 2013 which included 13 randomized trials with 1,910 patients showed a decreased hospital stay and postoperative complications after applying a MMR protocol versus classic protocols in the surgical treatment of colorectal cancer (2).
On the other hand, patients who need a surgery for digestive cancer present a high risk of malnutrition. There are several factors that deteriorate more their nutritional status like surgery aggression and its resultant increasing of energy expenditure, also the perioperative fasting periods. Preoperative malnutrition has important consequences on postoperative results explained by the relation between weight losses and morbid-mortality.
Malnutrition is associated with body composition changes, progressive tissue exhaustion and malfunction of organs such as cardiopulmonary, renal and digestive systems. This results in a decrease of immunity that causes in these patients development of wound infection complications or sepsis of an intra-abdominal source. In the immediate postoperative period low muscle strength have a higher risk of cardiorespiratory complications as mobility recovery deceleration that prolong the rehabilitation of the patient. Also they present abnormalities of their inflammatory response and failure of the wound healing process with the consequent anastomotic dehiscence risk and later infection complications. Even a well-nourished patient may suffer the adverse consequences derived of an inadequate nutritional support. For this reason, an adequate perioperative nutritional intervention has a favorable impact on morbidity and mortality outcomes (3,4).
Objetive
The aim of this present revision is to analyze whether the combined application of a perioperative nutritional intervention with a fast-track program in the patient undergoing a colorectal cancer surgery improves the mortality and morbidity outcomes, hospital stay and enhance patient's recovery. And to assess if a perioperative nutrition should be an item to follow in a fast-track program.
Methods
DESIGN
A critical analysis of papers recovered for a bibliographic review using a systematic technique. The systematic review was conducted in accordance with PRISMA guidelines (http://www.prisma-statement.org).
SOURCE OF DATA COLLECTION
All data were obtained by a direct consultation via Internet of the scientific literature contained in the following databases:
- Medlars Online International Literature (Medline), via PubMed.
- Scopus.
- Embase.
- Web of Science, Institute for Scientific Information (ISI).
- The Cochrane Library.
- Latin American and Caribbean Health Sciences Literature (LILACS).
- The Cumulative Index to Nursing and Allied Health Literature (CINAHL).
- International Pharmaceutical Abstracts (IPA).
INFORMATION SEARCH
Articles published from the start of indexation of each of the primary source were studied regardless of the country, institution or researcher and language in which it was published.
The search equation developed for its use in Medline database and The Cochrane Library was: ("Colorectal Surgery"[Mesh] OR "Colorectal Surgery"[Title/Abstract] OR "fast-track"[Title/Abstract]) AND ("Perioperative Care"[Mesh] OR "Perioperative Care"[Title/Abstract] OR "Enhanced recovery programme"[Title/Abstract]) AND ("Nutrition Therapy"[Mesh] OR "Nutrition Therapy"[Title/Abstract]).
The following filters were used: "humans", adult (19+ years) y clinical trial". Subheadings were not used and it wasn't necessary the application of tags.
Subsequently this search strategy was adapted for the -bibliographic databases mentioned above.
The search was performed from the first available date until 1st of May of 2014 (last update date) according to the characteristics of each database.
ARTICLES SELECTION
The selection of articles was done according to the following criteria:
- Inclusion criteria: all papers had to be clinical trials published in peer-reviewed journals with available complete text. The selected articles were those that focused on patients undergoing colorectal surgery using the fast-track method which is described and compared for its nutritional intervention.
- Exclusion criteria: were works that didn't focus on the target population (adults) and didn't report the directly derived effects of the nutritional intervention.
Additionally a second search was performed consulting the reference list of the identified works in order to reduce possible publication bias and to identify undetected studies in the electronic search.
Two authors assessed the relevance and adequacy of the studies independently (Ch-M y S-V). To consider valid the process of selection it was established that the assessment of the concordance between both authors (Kappa index) must be higher than 0.6 (good or very good strength of concordance).
Whenever this condition is met, any discrepancies would be resolved by consulting the coordinator of the review (WB) and subsequently by consensus among all authors. To reinforce the articles quality assessment the Critical Appraisal Skills Programme Spain (CASPe) was used among its 11 items applying the first three of them for screening and the questionnaire for the quantification of the critical analysis of scientific studies: clinical trials (CACEC-EC) which with its initial screening part reject the papers that don't reach the score of 6 points and has the following quality assessment values: low (0-6), good (7-14) and excellent (15-20).
To extract the data all the papers were grouped by the variables that define the postoperative outcomes (bowel recovery signs, hospital stay, postoperative complications, readmissions and deaths). The most important data of each work were summarized in a table (authors, publication year, design, nutritional intervention type, target population and principal results).
Results
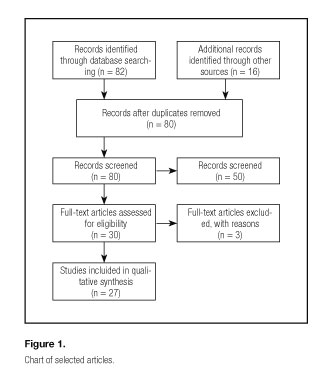
A total of 82 papers were localized in the following databases: Medline 20, Cochrane Library 19, Embase 42 and Scopus 1. Eighteen of them were redundant. After applying the inclusion and exclusion criteria 53 of them were rejected (Fig. 1). With the remaining 11 articles a total of other 16 papers were identified in the bibliographic list of the reviewed studies. Finally 27 articles were included (5-31).
The concordance between the authors was of 80% (Kappa index) which needed the intervention of a third author to reach consensus about the inclusion of two articles which finally were rejected.
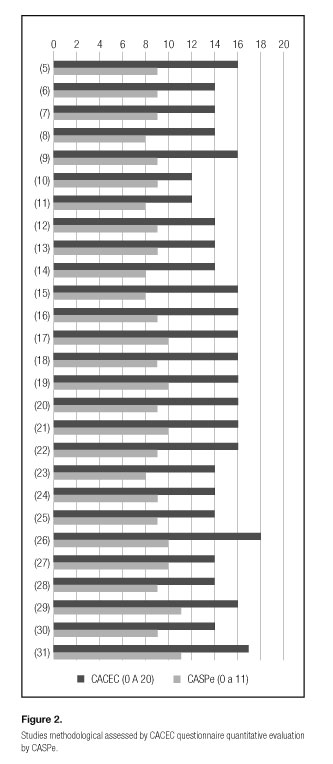
The quality of the articles was assessed with the CACEC-EC questionnaire, 14 (51.9%) of them presented good quality and 13 (48.1%) papers an excellent quality (Fig. 2). Also a qualitative assessment was performed by the CASPe for which 20 (74.1%) studies met between 9 (81.8%) and 10 (90.9%) of the evaluated items while 2 (7.4%) articles (29,31) included the eleven items (100,0%) (Fig. 2). Fifteen (55.5%) of 27 selected articles were published in the last 5 years: 2009-2013 (17-31).
The studies sample size oscillated between 597 (29) and 25 persons (9). The mean age of the included patients was between 16 and 94 years (5,8), except one of them (16) that had a small group of children of 10 years, whom were not considered in this revision and only adults were included in the review. Men were more predominant within 18 (66.6%) of the studies (10-14,17-20,22,23,25-31) and there was no mention about sex proportion in 2 (7.4%) works (6,8). Most of the studies proposed 2 groups, the intervention group (fast-track, G-FT) and the control group (traditional, G-T), only in 2 works 4 groups were designed (22,28) (Table I). The number of treated patients was available in all works, some of them had the same number in each study group (6,7,13,15,17,20). The postoperative follow-up varied by time and used method ranging from a minimum of 3 postoperative days (POD) and a maximum of 29 POD (8,11,12,15-17,19,22,27), only on author didn't mention the total follow-up period (9). The most of the authors monitored their patients even though others didn't mention It (5-7,13,14,23,25,28,29). Sometimes the follow-up was performed twice a day (9,12) while in another occasions a telephonic follow-up was used (10,18,21,24,27,30), those that reported the duration of follow-up period extended it until the 7th POD or until patient hospital discharge (31). In another work it remained for three months by making a visit to the hospital (27).
PATIENT'S NUTRITIONAL ASSESSMENT
The assessment of the nutritional status was performed in 13 (48.1%) studies (8,9,11-14,18,21,22,26,28-30) generally in the preoperative stage, body mass index (BMI) was used in 7 (53.8%) occasions (8,9,11,12,14,18,26) data was reported mostly by medians and interquartile range being the G-FT groups between 17 and 38.8 kg/m2 and the G-T groups between 17 and 56.8 kg/m2. Only one author (11.1%) used the subjective global assessment (SGA) (21) of patients nutritional status with which he found a higher number of moderately malnourished in G-FT (47.0%) versus a (7%) in G-T, in both groups was observed (7.0%) of severe malnourished. When BMI was used 18 (62.0%) of the patients presented overweight (BMI > 25 kg/m2) versus to 3 (7.0%) in G-T.
In the postoperative stage the nutritional data were collected in one paper (8) in which serum albumin was assessed, a decrease of its plasma levels was observed in both groups. However weight loss wasn't observed despite the fact that a decrease of fat body mass was found for both groups, it was only significant for G-T with 329 g (6.0%). Also a significant muscle body mass loss was observed in the G-T with 429 g (7.0%) but for G-FT it was insignificant loss of 158 g (2.0%). While other authors make no reference about the classification of their patient's nutritional status.
SURGICAL TECHNIQUE
A colorectal surgery was performed in all studies, technique wasn't specified in 7 of them (6,11,12,15,24,29,31), others described an open surgery (7,9,14,16-20,25,30), a laparoscopic procedure (5,10,13,26,27) and both techniques were used in another occasions (21,22,28).
NUTRITIONAL INTERVENTION
Traditional group
In the preoperative stage between 3 and 12 hours before surgery was given nil by mouth (6,8,9,12,15,16,18-22,28,30) the rest of author didn't reported its duration.
Intravenous fluids (8,11,15,16,18,22,28,29) were administrated before (8,11,15,18,28), during (15,16,22,29) and after surgery (11,16,18,28,29). It consisted in saline solution and Ringer's lactate (8,16,18,22,28) which remained until the 3rd POD (11,28) or until liquids were tolerated (16,29). Few works reported the amount of administrated liquids, 2 ml/kg/hours (18) o 3,000 ml/day (8).
In the postoperative phase liquid intake was permit in different times; immediately after surgery (12,14,19,22,31), from the 1st POD (7,11,23,28), from the second POD (10,13,18,20), until patients tolerance (16), resolution of ileus or the passage of first flatulence (5,6,15,17,21,24-27,29), or depending on the attending surgeon (8,9) (Table I).
Solids intake was allowed directly after surgery in 2 studies (12,19), others did it from the 1st POD (22,28,31) or after the second and fourth POD (11,14,18). While in the rest of studies it was limited until passage of flatulence (10,15,21,23), oral liquid tolerance (5-7,16,17,24-26) or under surgeon criteria (8,9,13).
Only in three studies was mentioned the type of diet used, mashed (11) and soft (14,16). Nutritional drinks weren't the most frequent election, only one author decided to use a supplement high in protein and calorie before giving solids (30).
Fast-track group
The first intake was allowed between the last 2 and 8 hours before surgery despite Lobato et al. whom established fasting during 12 hours (21). The liquids were administrated were unspecified (15,16,23), reported as clear liquids (6,18) or nutritional drinks (9,12,13,19,20,22,27-30). The supplements were rich in carbohydrates (9,12,19,20,22,28,29,30), plus pre/probiotics (9,12), with immune-nutrients (13) (arginine, omega 3, nucleotides) and in another occasion a supplement without residues (27).
An intravenous administration was used for fluids supply in the preoperative period (2-4 hours) (8,15,18,28), during surgery (15,16,18,22,24,27,29) and in the first 2 postoperative hours (11,15,16,22,23,28,29). The fluids used were saline solution (8,16) and Ringer's lactate (18,22,28). Different liquids amounts were used (Table I).
The first oral intake was established in 19 (70.4%) studies between the first 2 and 12 postoperative hours. Using unspecified liquids, clear type (7,9-12,15,16,18,19,23,25,31) or supplements rich in carbohydrates (22,28,30) or high in protein (8). Others allowed intake from the 1st POD giving a complete liquid diet (5,6,17,21,24), semi-soft diet (20) and in one occasion supplements rich in immune-nutrients (13).
Two authors began at the same time liquids and solids intake (8,19); the first one with a semi-soft diet (19) and the another one didn't reported its type (8).
The advance to a complete diet was done with different times and types of diet, a great variability between authors was observed (Table I). Most of the patients who started the intake of liquids (unknown type) progressed from the 1st POD with a conventional diet (5-7,11,15,17,18,20,21,24,25,31). In one study simultaneously a protein supplement was given (18) and in another work was a carbohydrate drink (31) while others introduced it gradually; as a semi-soft, soft, semi-solid and solid diet (22,23,26-28) and in another studies solids were allowed in order of patients tolerance (9,10,12,16).
PATIENT RECOVERY
Traditional group
The tolerance of food was observed between the 1st POD (Med 1 ,0-1, p < 0.01) (11) and the 5th POD (Med 5, 2-19, p < 0.001) (5,23) (Table II).
The recovery of bowels function was considered as the passage of flatus, peristalsis sounds and/or the passage of first stool. The authors presented their results as means and medians as shown in table II.
Fast-track group
Tolerance of food wasn't reported in all studies. Raue et al. (11) and another 2 authors (26,27) report that it happened the same day of surgery (Med 0, 0-0, p < 0.01) (11) while the latest one began from the 2nd POD (x 2.6 ± 0.1, p < 0.001) (5).
The onset of the three signs of bowels recovery occurred from the 1st POD, first flatulence (Med 1, 1-3, p < 0.001) (26), the passage of first stool (Med 1, 0-2, p < 0.001) (8) y and first peristalsis sounds (x¯ 1.3 ± 0.8, p < 0.001) (19).
SURGICAL COMPLICATIONS
Traditional group
Only four studies presented results about wound infection (21,23,28,31). In the G-T groups there was a total of 107 cases and Serclová et al. observed the highest incidence among 52 patients, 17 (32.7%) had this complication (19). Only in one studies no wound infections were reported (7). From eight (3.7%) patients who had abscesses, 5 (62.5%) of them were from Yang et al. (30) work, only one (12.5%) case was registered in the others studies (5,11,18).
Wound's dehiscence occurred in 59 (11.1%) cases registered in 6 studies (7,14,17,22,25,26), the highest incidence was observed in García-Botello et al. work with 50 (86.2%) of their patients (26).
Anastomotic leakage happened in a total of 46 (4.3%) patients in 12 works (5,6,14,15,17,18,20,22,24-26,29), the highest incidence was for Da Fonseca et al. with 4 (15.3%) of 26 patients (24). Hartsell et al. didn't register this complication (6).
Twelve works registered intestinal ileus in 52 (5.6%) patients (5,8-10,12,14,18,22,24,26,27,29) García-Botello et al. registered the highest incidence in 11 (18,9%) of their patients (26).
Vomits were reported in 14 studies for a total of 106 (13.8%) patients (5-7,12,13,15,17-20,22,23,26,30), the highest incidence was registered by Hartsell et al. in 14 (48.2%) of their patients (6).
Fast-track group
A total of 83 (5.7%) cases of wound infection were registered between the G-FT groups, Vlug et al. recorded the largest proportion of it in 11 (18.3%) patients (22). No episodes were registered in 3 studies (21,23,28) (Table III). A total of 4 (0.3%) cases of abscesses were obtained from all works (5,10,15,30).
Wound dehiscence happened in 62 (4.2%) cases, García-Botello et al. (26) presented the highest incidence with 51 (83.6%) patients.
Ten studies reported the presence of anastomotic leakage (4,15,17,18,20,22,24-26,29), Vulg et al. (22) registered the highest incidence (7.7%).
The presence of Intestinal ileus was detected in a total of 50 (3.4%) cases; García-Botello et al. reported the highest incidence in 11 (19.6%) of their patients (26). Three authors (6,24,27) didn't observe ileus in their patients.
Vomits happened in 123 (20.0%) patients from 5 studies (5-7,10,12,13,15,17-20,23,24,26,30), Harstell et al. Registered the highest incidence with a total of 16 (55.1%) patients (6).
Sharma et al. (31) didn't mention about complications.
HOSPITAL STAY, READMISSIONS AND DEATHS
Traditional group
All authors considered the hospital stay of patient, la shortest one for G-T was a Med 4 (p < 0.001 versus their FT group) and the longest one was a Med 12 (IQR 5-21), (p < 0.001 versus their FT group) (Table II).
Readmissions of patients happened in 11 studies (8,10-12,15-18,20,22,26) a total of 37 (6.1%) of them were registered. No data was presented in 12 (44.4%) works (5-7,9,13,14,21,25,27-29,31).
A total of 17 (3.3%) patients that deceased were recorded in 10 studies (6,7,9,12,14,15,22-24,26). Vulg et al. registered the higher number of them (22).
Fast-track group
In G-FT the hospital stay ranged between 2 and 9 days, the shorter was of Med 2 (IQR 1-3), p = 0.001 (16) and the largest stay was of Med 9 (IQR 4-81), p = 0.979 (14).
The readmissions for this group were registered in 11 (40.7%) studies (8,10-12,15-18,22,24,26) with a total of 38 (6.6%) patients (Table III). In 9 (33.3%) works (5,9,14,19-21,23,28,29) no readmissions were required.
There was a total of 15 (1.0%) deaths (12,14,22-24,26). The greatest number of deceased patients was found in Vulg et al. (22) work (Table III).
Discussion
This work shows that the aim of this revision is a current theme because 14 of the included articles (17-30) were published in the last 5 years (2009-2013). It is relevant to state that the nutritional intervention wasn't a primary aim of most of the studies. A high or excellent methodological quality was found for the included studies.
A great heterogeneity was found in the studies almost for all variables; sample size, sex, age also for outcome variables.
It is important to highlight that in most of the works patient's nutritional status wasn't assessed before surgery neither the recommended screening methods and tools were used, expect in the case of Lobato Dias Consoli (21) who used BMI and SGA and Basse et al. (8) who determined the serous albumin and used 36 DXA (dual X ray of absorptiometry) for the assessment of body composition for fat and muscle mass on femur.
Regarding to the characteristics that differs the nutritional intervention of the fast-track method from the traditional protocol, in the G-FT it was characterized by allowing liquids intake between the previous 2 and 8 hours before surgery predominantly by the use of carbohydrate nutritional supplements (CH) (9,12,19,20,22,28-30) where in 2 occasions pre/probiotics (9,12) were previously administrated. In another studies, supplements rich in immune-nutrients (13) or residue-free (27) were used. Existing literature about fast-track protocols declare that the intake of liquids and CH in the previous 2 and 4 hours is considered the most important item. Although in the G-T fasting was established between 3-12 hours before surgery.
In both groups (G-FT and G-T) fluids were administrated during the surgery by intravenous via, some authors maintained it in G-FT for the two postoperative hours (15,16,22,29) while for the G-T two authors continued administrating it until patient tolerated oral intake (16,29).
A free liquids intake was established between 2-12 postoperative hours for most of the G-FT but the amount was controlled in case nutritional supplements were used; 3-4 units/day of drinks high in CH (22,28,30), proteins (8) and immune-nutrients (13).
In most of the studies, the intake of a complete diet was achieved from the 1st POD for the G-FT, referred as normal diet, while two authors (8,19) introduced 2 hours after surgery. Also authors refer in their studies that drink rich in proteins (18) or carbohydrates (31) supplemented the diet. In the majority of traditional groups protocols liquids intake started with the appearance of intestinal signs [presence (10,15,21,23) of flatulencies or intestinal ileus resolution (5,6,17,24-27,29)] and when liquids were tolerated the intake of solids was allowed (5-7,16,17,24-26).
An earlier food tolerance was observed in G-FT as a tendency between the studies before it happened for the G-T, the work of Raue et al. is a paradigm for this issue by recording it with a median of 0 (0-0); p < 0.01 (11) after administrating a hospital diet with 1.5 L/day of liquids.
Intestinal function and food tolerance were used as parameters to assess patient's recovery. Bowel recovery was recorded by using four signs referred in our results (ileus resolution, presence of peristaltic sounds, gases expulsion, and first stool) indicators variability was observed between the studies for determining criteria. In Spanjersberg et al. (33) study the first passage of stool was observed in the G-FT before the G-T as an intestinal recovery sign recorded with a means difference of 1.12 days. Also Zhuang et al. (2) founded that the passage of stool was sooner in the G-Ft at the 3rd (1-5) day versus the G-T recorded at the 5th (0-23) day. Both authors support with their results the data found in this study.
The postoperative associated complication were classified in six types and their wasn't an unified criteria for its registration neither was referred a concert definition of them keeping doubts about the difference between wound infection and abscesses when both of them were referred as surgical infections. Regarding to the overall incidence of complications it was greater for the traditional groups, registered in 430 (28.9%) of 1,484 patients while it occurred in 378 (25.7%) of 1469 patients of the G-FT.
The most frequent complication in the G-FT was vomiting as in G-T although for this last group wound infection had the same frequency. A higher incidence of vomits was found in patients of the G-FT that had from the first postoperative hours until the 2 POD a complete liquid diet (n = 100) (5-7,17,20,23,24,26). In the case of patients in G-T a greater vomits incidence was registered for patients that started liquids intake after the presence of flatulencies and continued on drinking until the 2nd or 3rd POD (n = 67) (6,7,13,20,24,26). These results go along with Zhuang et al. outcomes found for the relation between the frequency of vomits and food early intake in G-FT compared to the G-T (RR 1.08; 95% CI 0.77-1.52; p = 0.65). Patients of G-T that received in the postoperative period a normal diet (12) as those that were allowed to start food intake after ileus resolution (17) or presence of flatulencies (26) and continued on consuming only liquids for the 2nd or 3rd POD presented the greatest incidence of wound infection (Table III).
It is important to highlight that no presence of wound infection was registered for patients of the G-FT that consumed pre/probiotics 7 days prior surgery and were administrated carbohydrate supplements at evening before the intervention (12).
Hospital stay was collected by all the studies and was considered a principal outcome. A shorter stay was presented in the G-FT versus the G-T as it is shown in Zhuang et al. meta-analysis founding a means difference of 2.4 days; p < 0.00001 (2).
The second variable considered by the authors was the number of readmissions recorded in 12 studies (8,10-12,15-18,20,22,24,26) but the reason wasn't specified. A similar number of readmissions was registered in both groups, 38 were in G-FT and 37 in G-T, this result is similar to Zhuang et al. (2,32) that found no significant difference in the number of readmissions between groups (RR 0.9; 95% CI 0.6-1.5; p = 0.88) (2,32).
It is important to remark that not all the studies considered the number of deaths as an outcome and it surprised that one author didn't present the number of deceased patients but indicated that the result hasn't reached a statistical significance (27). A slight difference was found for the number of deaths, 15 (1.0%) in G-FT and 17 (1.1%) in G-T but didn't reach significance. But Spanjersberg et al. (33) didn't found significant difference between groups, 1 in G-FT versus 3 for G-T (p = 0.8).
LIMITATIONS
In the present review serious difficulties were found for data extraction and information synthesis due to the presented heterogeneity in study's methodology, variables, also the applied statistics and available data made difficult the possibility to perform a deeper analysis of the studies.
Conclusions
The application of fast-track protocols is a current topic that is being addressed by a good methodological quality. Underlining the great heterogeneity found in the protocols for the nutritional intervention as well as for the monitoring process.
Generally in most of the protocols there was a lack of a nutritional assessment before the intervention neither it was applied for a postoperative assessment of patients status. It is important that in further studies this issue be considerate in order to assess the influence of nutritional status on outcome variables.
The lack of a common and homogenous nutritional intervention pattern shows a enormous variability between protocols. Although the early oral intake was a common denominator but interventions are far from being clear to able compression between them. Despite this, an earlier food tolerance and intestinal recovery were seen for G-FT than for the G-T.
It is not possible to conclude that the nutritional intervention in G-FT versus G-T protocol had a decreasing effect on complications and the hospital stay, it would be interesting that the impact of the nutritional intervention could be assessed in future designs.
References
1. Kehlet H. Fast-track surgery: the facts and the challenges. Cir Esp 2006; 80(4):187-8. [ Links ]
2. Zhuang CL, Ye XZ, Zhang XD, Chen B, Yu Z. Enhanced recovery after surgery programs versus traditional care for colorectal surgery: a meta-analysis of randomized controlled trials. Dis Colon Rectum 2013;56(5):667-78. [ Links ]
3. Rham D. A Guide to Perioperative Nutrition. Aesthetic Surg J 2004;24(4):385-90. [ Links ]
4. Huhmann MB, August DA. Perioperative nutrition support in cancer patients. Nutr Clin Pract 2012;27(5):586-92. [ Links ]
5. Reissman P, Teoh TA, Cohen SM, Weiss EG, Nogueras JJ, Wexner SD. Is Early Oral Feeding Safe After Elective Colorectal Surgery? A Prospective Randomized Trial. Ann Surg 1995;222(1):73-7. [ Links ]
6. Hartsell PA, Frazee RC, Harrison JB, Smith RW. Early Postoperative Feeding After Elective Colorectal Surgery. Arch Surg 1997;132(5):518-21. [ Links ]
7. Stewart BT, Woods RJ, Collopy BT, Fink RJ, Mackay JR, Keck JO. Early feeding after elective open colorectal resections: a prospective randomized trial. Aust NZJ Surg 1998;68(2):125-8. [ Links ]
8. Basse L, Raskov HH, Hjort Jakobsen D, et al. Accelerated postoperative recovery programme after colonic resection improves physical performance, pulmonary function and body composition. Br J Surg 2002;89(4):446-53. [ Links ]
9. Anderson AD, McNaught CE, MacFie J, Tring I, Barker P, Mitchell CJ. Randomized clinical trial of multimodal optimization and standard perioperative surgical care. Br J Surg 2003;90(12):1497-504. [ Links ]
10. Delaney CP, Zutshi M, Senagore AJ, Remzi FH, Hammel J, Fazio VW. Prospective, randomized, controlled trial between a pathway of controlled rehabilitation with early ambulation and diet and traditional postoperative care after laparotomy and intestinal resection. Dis Colon Rectum 2003;46(7):851-9. [ Links ]
11. Raue W, Haase O, Junghans T, Scharfenberg M, Müller JM, Schwenk W. 'Fast-track' multimodal rehabilitation program improves outcome after laparoscopic sigmoidectomy: a controlled prospective evaluation. Surg Endosc 2004;18(10):1463-8. [ Links ]
12. Gatt M, Anderson AD, Reddy BS, Hayward-Sampson P, Tring IC, MacFie J. Randomized clinical trial of multimodal optimization of surgical care in patients undergoing major colonic resection. Br J Surg 2005;92(11):1354-62. [ Links ]
13. Finco C, Magnanini P, Sarzo G, Vecchiato M, Luongo B, Savastano, et al. Prospective randomized study on perioperative enteral immunonutrition in laparoscopic colorectal surgery. Surg Endosc 2007;21(7):1175-9. [ Links ]
14. Han-Geurts IJ, Hop WC, Kok NF, Lim A, Brouwer KJ, Jeekel J. Randomized clinical trial of the impact of early enteral feeding on postoperative ileus and recovery. Br J Surg 2007;94(5):555-61. [ Links ]
15. Khoo CK, Vickery CJ, Forsyth N, Vinall NS; Eyre-Brook IA. A prospective randomized controlled trial of multimodal perioperative management protocol in patients undergoing elective colorectal resection for cancer. Ann Surg 2007;245:867-72. [ Links ]
16. Kuzma J. Randomized clinical trial to compare the length of hospital stay and morbidity for early feeding with opioid-sparing analgesia versus traditional care after open appendectomy. Clin Nutr 2008;27(5):694-9. [ Links ]
17. El Nakeeb A, Fikry A, El Metwally T, Fouda E, Youssef M, Ghazy H, et al. Early oral feeding in patients undergoing elective colonic anastomosis. Int J Surg 2009;7(3):206-9. [ Links ]
18. Muller S, Zalunardo MP, Hubner M, Clavien PA, Demartines N; Zurich Fast Track Study Group. A fast-track program reduces complications and length of hospital stay after open colonic surgery. Gastroenterology 2009;136(3):842-7. [ Links ]
19. Serclová Z, Dytrych P, Marvan J, Nová K, Hankeová Z, Ryska O, et al. Fast-track in open intestinal surgery: prospective randomized study (Clinical Trials Gov Identifier no. NCT00123456). Clin Nutr 2009;28(6):618-24. [ Links ]
20. Ionescu D, Iancu C, Ion D, Al-Hajjar N, Margarit S, Mocan L, et al. Implementing fast-track protocol for colorectal surgery: a prospective randomized clinical trial. World J Surg 2009;33(11):2433-8. [ Links ]
21. Lobato Dias Consoli M, Maciel Fonseca L, Gomes da Silva R, Toulson Davisson Correia MI. Early postoperative oral feeding impacts positively in patients undergoing colonic resection: results of a pilot study. Nutr Hosp 2010;25(5):806-9. [ Links ]
22. Vlug MS, Wind J, Hollmann MW, et al.; LAFA study group. Laparoscopy in combination with fast track multimodal management is the best perioperative strategy in patients undergoing colonic surgery: a randomized clinical trial (LAFA-study). Ann Surg 2011;254(6):868-75. [ Links ]
23. Morończyk DA, Krasnodębski IW. Implementation of the fast track surgery in patients undergoing the colonic resection: own experience. Pol Przegl Chir 2011;83(9):482-7. [ Links ]
24. Da Fonseca LM, Profeta da Luz MM, Lacerda-Filho A, Correia MI, Gomes da Silva R. A simplified rehabilitation program for patients undergoing elective colonic surgery--randomized controlled clinical trial. Int J Colorectal Dis 2011;26(5):609-16. [ Links ]
25. Dag A, Colak T, Turkmenoglu O, Gundogdu R, Aydin S. A randomized controlled trial evaluating early versus traditional oral feeding after colorectal surgery. Clinics (Sao Paulo) 2011;66(12):2001-5. [ Links ]
26. García-Botello S, Cánovas de Lucas R, Tornero C, et al. Implementation of a perioperative multimodal rehabilitation protocol in elective colorectal surgery. A prospective randomized controlled study. Cir Esp 2011;89(3):159-66. [ Links ]
27. Wang Q, Suo J, Jiang J, Wang C, Zhao YQ, Cao X. Effectiveness of fast-track rehabilitation vs conventional care in laparoscopic colorectal resection for elderly patients: a randomized trial. Colorectal Dis 2012;14(8):1009-13. [ Links ]
28. Van Bree SH, Vlug MS, Bemelman WA, et al. Faster recovery of gastrointestinal transit after laparoscopy and fast-track care in patients undergoing colonic surgery. Gastroenterology 2011;141(3):872-80. [ Links ]
29. Ren L, Zhu D, Wei Y, Pan X, Liang L, Xu J, et al. Enhanced Recovery After Surgery (ERAS) program attenuates stress and accelerates recovery in patients after radical resection for colorectal cancer: a prospective randomized controlled trial. World J Surg 2012;36(2):407-14. [ Links ]
30. Yang D, He W, Zhang S, Chen H, Zhang C, He Y. Fast-track surgery improves postoperative clinical recovery and immunity after elective surgery for colorectal carcinoma: randomized controlled clinical trial. World J Surg 2012;36(8):1874-80. [ Links ]
31. Sharma M, Wahed S, O'Dair G, Gemmell L, Hainsworth P, Horgan AF. A randomized controlled trial comparing a standard postoperative diet with low-volume high-calorie oral supplements following colorectal surgery. Colorectal Dis 2013;15(7):885-91. [ Links ]
32. Zhuang CL, Ye XZ, Zhang CJ, Dong QT, Chen BC, Yu Z. Early versus traditional postoperative oral feeding in patients undergoing elective colorectal surgery: a meta-analysis of randomized clinical trials. Dig Surg 2013;30(3): 225-32. [ Links ]
33. Spanjersberg WR, Reurings J, Keus F, Van Laarhoven CJ. Fast track surgery versus conventional recovery strategies for colorectal surgery. Cochrane Database Syst Rev 2 2011;(2):CD007635. [ Links ]
![]() Correspondence:
Correspondence:
Carmina Wanden-Berghe.
Hospital General Universitario de Alicante - ISABIA-FISABIO.
C/ Pintor Baeza, s/n. 03010 Alicante, Spain
e-mail: carminaw@telefonica.net
Received: 16/11/2015
Accepted: 01/12/2015