INTRODUCTION
Prevalence rates of nutritional risk and malnutrition are high among cancer patients 1) (2. Malnutrition prevalence at the time of diagnosis has been estimated to range from 15% to 40%, with this value increasing to 80% with cancer progression. Hospital malnutrition compromises surgical treatment outcomes and leads to more infectious complications, increased length of hospital stay, and mortality 1) (3) (4.
Cancer patients are at high nutritional risk. When malnourished and unable to meet their nutritional needs through the oral route alone, cancer patients are candidates for the early introduction of high-quality effective nutritional therapy. Enteral nutrition therapy (ENT) is considered to be the best route of nutrition when the gastrointestinal (GI) tract is structurally and functionally intact 5. Nutrients provided by the digestive system help to maintain the architecture of intestinal microbiota and to modulate the intestinal immune system. Thus, compared to parenteral nutritional therapy, ENT is associated with a lower incidence of infectious complications in surgical patients 6. However, benefits of ENT for cancer patients are only achieved if ENT is administered properly and efficiently.
The Task Force of Clinical Nutrition at the International Life Sciences Institute of Brazil (ILSI - Brazil) proposed indicators to assess the quality of ENT provided by hospitals. These quality indicators in nutritional therapy (QINTs) have been used for the practical assessment of the quality of ENT provided by various health services 7) (8) (9) (10) (11. Despite the availability of QINTs, however, only a few published studies have monitored the quality of using exclusive ENT 12, particularly in cancer patients 13. In this context, the present study aimed to analyze the adequacy and quality of ENT used in patients with cancer diagnoses and undergoing treatment at specialized public hospitals in São Paulo City, Brazil, by applying selected QINTs.
MATERIAL AND METHODS
ETHICAL CONSIDERATIONS
This research was previously approved by the Research Ethics Committee of the corresponding institutions (NP 315/12 and CEP125/13). The protocol was performed in accordance with the principles of the Declaration of Helsinki (1975).
PATIENTS STUDIED
This prospective, observational, descriptive study included adult patients admitted to the Instituto do Câncer do Estado de São Paulo (ICESP), a tertiary public-referral hospital with expertise in cancer management in the city of São Paulo, Brazil. Cancer patients admitted to wards were aged 19 years or older, with exclusive ENT for at least 72 h. All patients were under treatment for cancer complications and/or chemotherapy and radiation therapy and provided their informed consent for inclusion in the study. Exclusion criteria were as follows: age less than 19 years; ENT use for less than 72 h; prescription for an oral diet, parenteral nutrition therapy, or parenteral and enteral nutrition; surgical treatment; palliative care; colostomy and/or ileostomy; and admission to the intensive care unit (ICU). All patients who did not fit the exclusion criteria were included in the study.
NUTRITIONAL STATUS ASSESSMENT
Data related to patient demographics, clinical information, nutritional status, and ENT characteristics were collected by consulting the TASI(c) electronic medical records collected during the period from June to November 2013. The Nutritional Risk Screening (NRS) tool was used for nutritional risk assessment 14. In the presence of nutritional risk, Subjective Global Assessment (SGA) 15 was applied to evaluate nutritional status. NRS and SGA were carried out by nutritionists in different hospital wards. Caloric and protein needs were estimated on an individualize basis, according to the patient's initial clinical condition and nutritional status, and in compliance with the institutional protocol, which includes specific guidelines for cancer nutrition therapy 16.
ENTERAL NUTRITION THERAPY
In all patients, ENT was administered into the stomach. Correct positioning of the enteral tube was confirmed by X-ray before introduction of ENT. After medical and diet prescriptions were established, enteral formulas (closed system) were administered by infusion pumps (Lifemed(r) Model LF 2001 Brazil), according to the institutional protocol (intermittently in six steps per day, during the period from 8:00 to 23:00 hours). We used three types of enteral formulas available at the institution: polymeric hypercaloric, normal protein with and without fiber, and oligomeric.
After data collection, percentages of caloric and protein adequacy were calculated as the ratio between the amount of calories and protein administered effectively and the amount of calories and protein prescribed each day. Then, the mean percentage of adequacy and the cumulative calorie and protein deficits for each patient were calculated. Outcome data for each patient were collected from electronic medical records.
SELECTED INDICATORS OF ENTERAL NUTRITION
We applied six QINTs, which were related to the frequency of nutritional assessment, calorie/protein requirements, ENT administration, fasting and digestive motility complications caused by ENT (Table I). With regard to bowel habits, the patient was considered to have diarrhea when there were more than three episodes of watery stools per day. The patient was considered to have constipation in the absence of evacuation for three consecutive days 7) (9) (11) (16) (17.
STATISTICAL ANALYSIS
Statistical analyses were performed using STATA(r) software. We used the Kolmogorov-Smirnov test (p > 0.05) to verify sample normality. For parametric variables, we used mean and standard deviation (SD) values. For nonparametric values, median values and interquartile ranges (IQRs, p25-p75) were used. To compare qualitative variables, we used the chi-square test (χ2). To compare quantitative variables, we used ANOVA and Student's t-test. For parametric variables, the Kruskal-Wallis test was used. For nonparametric variables, the Mann-Whitney test was used. A difference with p < 0.05 was considered statistically significant for all tests.
RESULTS
The sample comprised 211 patients (mean age: 59 ± 10 years, 67.3% men). Head and neck (HN) was the most common cancer diagnosis, accounting for 68.2% of cancer diagnoses. Other cancer diagnoses encountered are presented in table II. Primary reasons for admission to the hospital were problems related to cancer or its treatment (97.6% of cases). Two patients were hospitalized to finalize chemotherapy cycles. And two others were hospitalized to receive concurrent chemotherapy and radiotherapy. At the time of admission, 93.3% (n = 197) of patients were at nutritional risk, including 84.2% (n = 166) who had moderate and severe malnutrition (SGA B+C). Prevalence of malnutrition was higher in patients with HN and GI tract cancer. Table III presents details related to nutritional status for all patients studied, stratified according to cancer diagnosis.
Table II Most common primary cancer diagnoses in patients treated exclusively with enteral nutritional therapy

Table III Nutritional status in cancer patients with exclusive enteral nutritional therapy

NRS: Nutritional Risk Screening; SGA: subjective global assessment; SGA B+C: moderate and severe malnourishment.
Source: Serviço de Nutrição e Dietética.
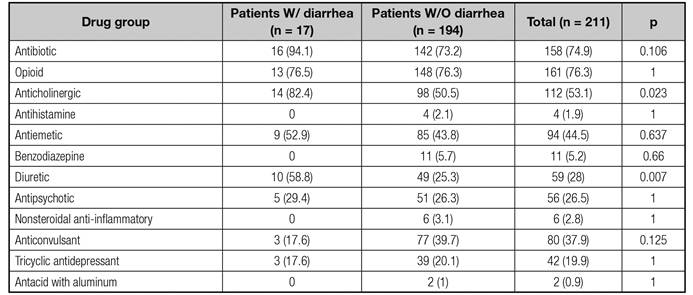
On average, patients used ENT exclusively for 9.7 ± 7 days. Daily amount of enteral diet prescribed was approximately 1 L (1,500 calories). However, the volume administered was lower than that prescribed; on average, this resulted in a reduced supply of calories and protein. The ratio of prescribed/infused enteral diet volume, calories and protein was 74.3% (Table IV). On average, the cumulative deficit for the entire sample was more than 3,000 kcal and 130 g protein. The cumulative energy and protein deficits for all patients (stratified for HN and GI cancer) are shown in figure 1.
Table IV Characteristics of enteral nutritional therapy in cancer patients

ENT: enteral nutrition therapy.
Source: Serviço de Nutrição e Dietética.
Source: Serviço de Nutrição e Dietética
Figure 1 Dispersion of cumulative caloric/protein deficit values in cancer patients treated exclusively with enteral nutritional therapy. HN: head and neck cancer; GI: gastrointestinal cancer.
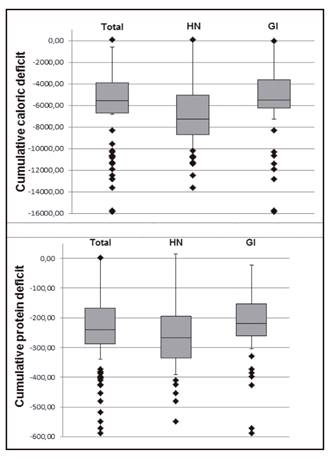
Among the six applied QINTs, three (50%) were in line with goals established by the institution: QINT I, which evaluated the frequency of SGA application in patients at nutritional risk; QITN II, which verified the fulfillment of energy and protein requirements; and QITN V, which investigated the frequency of episodes of diarrhea. Three QINTs presented disagreement with the proposed goals: QINT III, which evaluated the frequency of patients with ENT-infused volume of greater than 70%; QINT IV, which addressed the frequency of digestive fasting for more than 24 h in patients using ENT; and QINT VI, which evaluated the frequency of constipation in patients on ENT (Table V).
Table V Quality indicators in nutritional therapy in cancer patients treated by exclusive enteral nutritional therapy
SGA: subjective global assessment; ENT: enteral nutrition therapy.
Source: Serviço de Nutrição e Dietética.
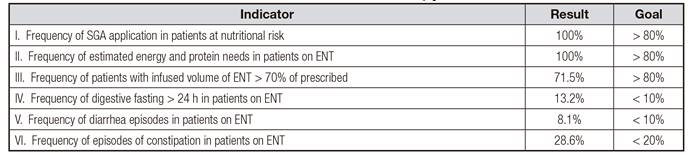
Most frequently prescribed drugs for patients with constipation or diarrhea were antibiotics (74.9%), opioids (76.3%), and anticholinergic drugs (53.1%). No relationship was found between prescription of these drugs and the presence of constipation. Use of anticholinergic drugs (p = 0.023) or diuretics (p = 0.007) was associated with diarrhea (Table VI). No relationship between the presence of constipation or diarrhea and chemotherapy was identified; however, patients who did not undergo chemotherapy tended to have more constipation (p = 0.059).
DISCUSSION
Among cancer patients, malnutrition is associated with a high risk of infection and hospital admission, lower rate of survival, and ecreased quality of life 18. In our study, we found nutritional risk in 93.3% of cancer patients who were treated exclusively with ENT. In this group, 84.2% of patients had some degree of malnutrition (SGA B+C). These data were not in accordance with a report by Silander et al. 19, in which values of malnutrition varied from 26% to 66% among a group of 119 cancer patients.
In our study, HN and GI tract cancers were most prevalent; among this subgroup, greater than 80% of patients had malnutrition. Depending on tumor location and disease progression, these patients can suffer from anorexia and dysphagia, factors that contribute to malnutrition, which is exacerbated by a delayed cancer diagnosis and difficulty of accessing public treatment associated with low economic social status, which was prevalent amount our patient population 1-3,5,16,20.
Due to the difficulty of oral feeding in malnourished patients with HN and GI cancers, the use of ENT represents a useful alternative 3,16,18,20. However, use of ENT is not free of adverse effects, such as refeeding syndrome, high residual gastric volume, diarrhea, and constipation. In addition, successive periods of fasting for various tests or procedures result in reduced protein and calorie intakes 5,12,21. To attain the expected benefits of ENT, it must be administered properly and efficiently, in accordance with institutional guidelines 22.
To control the quality of ENT use, our hospital has applied the Brazilian QINTs 7,9,11,13 since 2008. In our study, nutritional status was assessed using the SGA (QINT I) in all patients at nutritional risk. Combined use of the NRS and SGA can predict negative clinical outcomes 23. We found 197 patients at nutritional risk and 166 with SGA B+C. Among this latter group, we calculated a mortality rate of 59%, according to the findings of Raslam et al. 23. These data reinforce the utility of systematic implementation of SGA in cancer patients at nutritional risk.
We estimated caloric and protein needs (QINT II) using "pocket formulas" 16 for all patients studied. Various tools can be used to estimate caloric needs in cancer patients, but indirect calorimetry remains the gold standard in terms of performance. In clinical practice, the use of predictive equations and "pocket formulas" for estimating patient energy expenditure predominates 5,12,13,16,24.
It is assumed that patients receiving ENT volumes close to 100% of the prescribed volume will progress with lower rates of infectious complications, shorter hospital stays, and with a tendency to a lower mortality rate 25. However, discrepancies between prescribed and infused volume had been reported as an important factor for hypoalimentation including high nutritional risk patients 25-27,32. In 3390 patients at high nutritional risk at ENT, it was found poor adequacy in protein and energy supply (57.6% and 61.2%) and 74% of them did not receive at least 80% of their nutritional goal 32. In cancer patients at high nutritional risk at ENT it was found 89.1% of adequacy of the volume prescribed 28. However, in our study, 28.5% of patients received less than 70% of the prescribed volume of ENT (QINT III). Our findings were similar to those observed for critically ill patients at nutritional risk 12. This indicator can be interpreted in conjunction with the QINT IV (frequency of digestive fasting > 24 h in patients on ENT). In our group, this calculation yielded a value of 13.2% with respect to the established goal. This value is in accordance with observations for 93 critically ill patients at nutritional risk treated exclusively with ENT more than 72 h 12.
The total volume of the prescribed enteral diet that was not administered can be attributed to GI intolerance, due to abdominal distension, diarrhea, and/or vomiting, as well as fasting pauses for exams and surgical procedures 21,27-30. There are other reasons for pausing ENT, such as the absence of or noncompliance with specific ENT protocols 22,27 and failures in the logistics of ENT delivery to wards by the nutrition service 29. Successive delays in the delivery of ENT to wards, in addition to prolonged fasting times, can exacerbate the calorie and protein deficits in cancer patients treated exclusively with ENT 29.
Refusal of patients to participate in the last hours of enteral feeding at night was one reason that some did not receive full enteral nutrition (QINTs III). The unsuitability of a prescribed volume, administered effectively, resulted in a cumulative deficit of 3,000 calories and 130 g protein, which can impair the development and survival of patients 31. Given the results of QINTs III and IV, we opted to modify the ENT administration from intermittent to continuous over a period of 14 h per day. This approach resulted in improved results on QINT III (not shown).
Several barriers have been reported to prevent the full offer of the prescribed energy and protein amount to ENT patients 21,28-30. The prolonged time to achieve the nutritional goal, for example, was the main reason to contribute for energy-protein deficit in cancer patients at high nutritional risk 28. Also, GI intolerances may delay the attainability of nutritional goal in ENT patients 33. Recently we have shown that the average time to reach the nutritional target was 61.2 h for ENT cancer patients 28. The timing to advance to full nutritional goal on ENT is still unclear, but, when tolerated, enteral feeding should be advanced to full goal after 48-72 h of start. However, with reduced GI tolerance (diarrhea, constipation and/or abdominal distension) feeding should be advanced with caution to achieve full goal by 5 to 7 days 5.
Frequency of diarrhea associated with ENT varies from 9% to 41% 24,25,34. In our study, we observed a lower frequency of episodes of diarrhea in patients on ENT (QINT V). Among clinical cancer patients, diarrhea can be considered as an adverse effect of antineoplastic treatment (mucositis, enteritis) and/or associated with intensive antibiotic therapy, including Clostridium difficile infection 35. It is known that diarrhea can be attributed to the use of certain drugs, and we found that patients treated with anticholinergic drugs (p = 0.023) and diuretics (p = 0.007) presented diarrhea during follow-up.
We observed episodes of constipation in 28.6% of patients with ENT (QINT VI), which was beyond the desired value. Machado et al. 36 showed constipation in 58.5% of patients treated exclusively with ENT, while Bittencourt et al. 34 found constipation in 70% of patients with and without cancer, especially among those who received formula without fiber. At our institution, according to protocol, all patients with no evidence of diarrhea or GI discomfort received standard polymeric formula containing a fiber mix (soluble and insoluble; average, 20 g/d) and water as required. Drugs such as opioids have been associated with constipation 35,37; however, in our study, there was no relationship between drugs prescribed and the presence of constipation, as observed in severely ill patients in the ICU with ENT 37. Patients who did not undergo chemotherapy at our institution tended to have more episodes of constipation (p = 0.059).
We believe that the application of the QINTs is important and useful in evaluating the quality of nutritional care 7-13,17,24,38,39 and should be performed according to the guidelines provided by the Joint Commission on Accreditation of Health Care Organization 40 and The Task Force of Clinical Nutrition at the ILSI - Brazil 7. Evaluating the quality of nutritional care allows nutritionists to recognize deviations from established goals, which, when corrected, can ensure patient access to the very best nutritional therapy. This approach facilitates the recovery/maintenance of nutritional status at low cost and the medium- and long-term improvement of quality of life 7,9,11,17,38,39.
Many QINTs are available for use, and it is challenging for health professionals to define the QINTs to be applied at each hospital. There is no general rule for selecting a QINT. These decisions should be made based on the needs and experience at each particular institution 17.
Nutrition programs aiming improvement of ENT can be done with success, as shown by increased caring out of admission nutrition screening, implementing oral intake, ENT and parenteral nutrition, or by reducing involuntary withdrawal of enteral feeding tubes, and diarrhea episodes rates among hospitalized patients 12,24,34,38,39. In Brazil, Waitzberg and Correia 39 recently published the main implemented strategies that resulted in quality improvement of nutrition therapy. The authors pointed out that a rigorous monitoring by Nutrition Support Team is paramount, in addition to the creation/execution of continuous education projects to all members of the multidisciplinary team and the periodical selection and application of QINT.
Our study was the first to assess quality control of ENT, through the implementation and monitoring of QINTs in cancer patients treated exclusively with ENT. However, the study had certain limations. The study was developed in only one assistance referral hospital for cancer patients, had a reduced number of cancer diagnoses, and included a small number of patients. For better results, it will be necessary to carry out more studies to assess cancer patients on ENT, parenteral nutrition, and nutritional oral supplementation, and to relate QINT application to questionnaires assessing quality of life. Carrying out further studies may also allow for a reasonable comparison among health institutions and guide future strategic actions to improve nutrition therapy.
CONCLUSION
Prevalence rates of nutritional risk and malnutrition are high among cancer patients treated exclusively with ENT. Application of ENT was moderately impaired by episodes of fasting and intestinal motility disorders. The QINTs implementation is important and useful to assess quality in the management of ENT among cancer patients at high nutritional risk.