BACKGROUND
Glutamine (GLN) is the most abundant free amino acid in the body and is commonly known as a nonessential amino acid, because of the ability of most cells to produce it. It has many essential metabolic functions in the organism, such as the transportation of nitrogen, and is the most important substrate for renal ammoniagenesis 1 2 3. GLN is also a beneficial substrate for metabolically stressed patients, especially during critical illness, where patients experience general nutritional depletion and augmented infectious complications, which are correlated with low plasma GLN concentrations 4 5 6 7 8. GLN has an important influence on the inflammatory response, oxidative stress, apoptosis modulation, and the integrity of the gut barrier 9 10 11 through the attenuation of multiple inflammatory pathways. It decreases nuclear factor kappa-light-chain-enhancer of activated B cells (NF-B) expression, protein kinases, and inhibits the increase in nitric oxide synthase expression 12.
Moreover, GLN has beneficial immune functions related to cardiac protection after ischemia/reperfusion (I/R) in cardiopulmonary bypass (CPB); it increases myocardial adenosine triphosphate-adenosine diphosphate (ATP-ADP) substrate, prevents intracellular lactate accumulation, and enhances the accumulation of myocardial glutathione (GSH), a major stress substrate for the stressed myocardium, post-I/R injury 13. Patients undergoing CPB are at increased risk of having abnormal inflammation in their body after surgery. Such inflammation can contribute to slower recovery from surgery, an increased risk of infection, an increased risk of damage to organs other than the heart, and a more complicated course 14.
Numerous experimental and clinical trials have demonstrated the cardioprotective effects of GLN, including dose-dependent enhanced myocardial functional recovery following acute normothermic ischemia in rats 15. GLN has also been shown to reduce infarct size to approximately 39% in a rabbit model following ischemia/reperfusion injury 16. GLN treatment also increased load tolerance in patients with ischemic heart disease (IHD) 13,17.
Enteral GLN is suggested to contribute to de novo synthesis of Arginine (ARG). ARG is an important regulator of protein synthesis and proteolysis, and is the sole precursor for nitric oxide generation, a signaling agent with a crucial role in immunity, inflammation, and organ perfusion. GLN supplementation is suggested to be a more physiologic way of correcting ARG concentrations and subsequently achieving both GLN and ARG benefits. GLN, by restoring ARG availability should improve tissue oxygenation and myocardial protection 18.
Despite various experimental data showing the cardioprotective effects of GLN, there is a lack of clinical trials with patients undergoing CPB. The finding that lower levels of cardiac injury markers are observed in patients treated with oral GLN prior to CPB can have major implications for these patients. The purpose of this study was to determine whether an oral supplementation of 0.5 g/kg GLN prior to heart surgery contributes to a reduction in cardiac injury markers, postoperative complications, and mortality in Mexican patients.
METHODS
PATIENTS
A randomized clinical trial was performed in 28 Mexican patients with a confirmed diagnosis of IHD who underwent CPB with extracorporeal circulation. The study was conducted between January 2014 and September 2015 in the Specialty Hospital of the Western National Medical Center, Mexican Institute of Social Security.
The present protocol included men and non-pregnant women aged 40-70 years with a confirmed diagnosis of IHD that required coronary revascularization under CPB. All surgical procedures were performed under the same anesthetic technique and the same group of cardiovascular surgeons.
Exclusion criteria included preexisting kidney or liver dysfunction, demonstrated with a creatinine level above 1.6 mg/dl and total bilirubin level above 2 mg/dl. Also, any comorbid condition, such as drug or alcohol abuse, human immunodeficiency virus infection, hepatitis B or C or suspicious of any active infection defined as presence of fever above 37.8 °C, leukocytosis with or without a positive culture. Also patients with known allergies to the components of GLN or maltodextrin as well as signs of ongoing ischemia (defined by a persistent elevation of troponin-I [TROP-I] and creatine phosphokinase-Mb [CPK-Mb] levels and ingestion of high-protein diet or a diet with any supplemental GLN) were not considered candidates for this study.
TREATMENT
Following study enrollment, patients were randomly assigned (blinded envelopes were opened sequentially by a blinded study pharmacist) to receive oral GLN supplement (study group) or maltodextrin as an isocaloric complex carbohydrate as control group.
All investigators and clinical caregivers were blinded to the study intervention. All patients in the GLN group received an oral GLN supplement (Glutapack-10(tm) VICTUS, Miami, FL, USA). The total GLN/maltodextrin dose given to patients was standardized to 0.5 g/kg/day for the 3 days prior to CPB and one final dose of 0.25 g/kg/day of GLN/maltodextrin on the morning of surgery, 4 hours prior to initiation of anesthesia. Compliance with ingestion of the study drug was assessed via daily reminder calls from the study investigator and required empty package returns.
PATIENT SAMPLE COLLECTION AND ANALYSIS
Blood was collected at baseline (one hour prior to surgery) and one hour after surgery, and then at 12 and 24 hours postoperatively. After collection, blood was processed for the analysis of the cardiac injury markers TROP-I, creatine phosphokinase (CPK), and CPK-MB, and analyzed using Meso Scale technology (Meso Scale Discovery, Gaithersburg, MD, USA). These markers constituted the primary outcome variables.
CLINICAL DATA COLLECTION
All essential demographic information was obtained of all patients. Preoperative cardiac evaluation included cardiac ejection fraction measured closest to the surgical procedure, and during surgery total pump time and aortic clamping time were included in the analysis.
Infectious complications were defined as follow: Systemic inflammatory response syndrome was defined as a systemic response to a variety of severe clinical insults manifested by at least 2 of the following conditions: a) temperature > 38 °C or < 36 °C; b) heart rate faster than 90 beats/min; c) respiratory rate faster than 20 breaths/min or an arterial partial pressure of carbon dioxide below 4.3 kPa; and d) white blood cell count larger than 12,000 cells/mm3, less than 4,000 cells/mm3, or more than 10% of immature forms. Sepsis was defined as the systemic response to infection and septic shock was defined as sepsis with hypotension despite adequate fluid resuscitation and the presence of perfusion abnormalities, including but not limited to lactic acidosis, oliguria, or an acute alteration in mental status. Multiple organ dysfunction syndrome was defined as the presence of altered organ function in an acutely ill patient such that homeostasis could not be maintained without intervention.
Other Infectious morbidity included pneumonia which was defined as a chest radiographic examination showing new or progressive infiltrate, consolidation, and cavitation (interpreted by a radiologist blinded to a patient's treatment assignment), and at least 2 of the following: a) temperature above 38.5 °C or below 35°C; b) a white blood cell count larger than 10 x 103/L or less than 3 x 103/L; and c) isolation of pathogens from the sputum, bronchial aspirates or bronchial brushing. Bacteremia was diagnosed when a pathogen was isolated from the blood with a temperature above 38.5 °C or below 35 °C or a white blood cell count larger than 10 x 103/L or less than 3 x 103/L, and it was not related to infection at another site. Urinary tract infection was defined as the isolation of at least 105 colonies/mL of a pathogen from the urine.
Catheter-related sepsis was diagnosed if the patient had local signs of infection at the entry site, a temperature > 38.5 °C or < 35 °C, a white blood cell count > 10 x 103/L or < 3 x 103/L that resolved after catheter removal with no other infection site, the semi quantitative culture of the catheter tip showing more than 15 colony-forming units/mL, or isolation of a pathogen from blood cultures.
Inotropic support was defined as a requirement for infusion of an inotrope or vasopressor (dopamine, adrenaline, dobutamine, noradrenaline) equivalent to dopamine dosages (> 5 μg/kg per minute) or their combination for at least 8 hours after surgery.
Secondary efficacy parameters included durations of hospital stay, intensive care unit (ICU) stay, ventilatory support, and the incidence of mortality.
SAMPLE SIZE
The sample size was predetermined. We consider the results obtained by Sufit et al. 12, in a pilot study, which evaluated markers of myocardial damage in patients undergoing cardiac surgery and who received or not received oral glutamine preoperatively. They showed a drop of more than 50% in the level of troponin I, 24 hours after the study compared to the control. Using a formula for mean differences, with a confidence level () of 0.05 and a power () of 0.10, a total of 14 patients per group were established as sufficient.
STATISTICAL ANALYSIS
Descriptive analyses for quantitative variables included the mean, standard deviation, and standard error of the mean (SEM) for values related to cardiac injury markers. Raw numbers and percentages were used for qualitative variables. Inferential statistical analyses included the parametric Student's t test for independent samples and the 2 test and/or Fisher's exact test. Relative risk (RR) and 95% confidence intervals were also calculated. A p value < 0.05 was considered significant. Office Excel 2007 (Microsoft Corp., Redmond, WA, USA) and SPSS version 20 for Windows (IBM Corp., Armonk, NY, USA) were used for data processing and statistical analysis, respectively.
ETHICAL CONSIDERATIONS
The study was conducted according to the principles of the Declaration of Helsinki of 1989 and the Mexican Health Guidelines for Human Research. The study protocol was approved by the Mexican Local Committee for Ethics and Research (2014-1301-76) and was registered at www.clinicaltrials.gov with the identifier number NCT02491931. All patients included gave their signed informed consent to participate in this study.
RESULTS
During the period of study, 317 patients underwent cardiac surgery. Ninety-eight patients required valve replacement, 69 patients were treated without CPB (off-pump coronary revascularization), 55 required valve replacement and coronary revascularization, 29 patients presented with hepatitis B or C infection, kidney or liver insufficiency, or a history of a high-protein diet, and 21 were treated for tumors or congenital conditions. Forty-five patients were suitable for inclusion in the study, but 17 patients did not accept the invitation to participate. The remaining 28 patients agreed to participate. They were randomized into two groups: the GLN group (n = 14) and the control (CONT) group (n = 14). All patients were diagnosed with ischemic heart disease and underwent coronary revascularization with extracorporeal circulation. The gender distribution was equal in both groups, with 3 women
(21.4%) and 11 men (78.6%) in each group. The mean ages were 62.5 ± 7.4 and 63.2 ± 8.0 years for the GLN and CONT groups, respectively, with no statistical difference (p = 0.82). The results are illustrated in table I.
Anthropometric data revealed normal weight in 7 patients in the GLN group and overweight or obesity in the remaining 7. In contrast, the CONT group consisted of 5 patients of normal weight and 9 overweight or obese patients. However, the difference was not statistically significant (p = 0.73). The body mass index (BMI) was similar between groups, 25.7 ± 3.1 and 24.4 ± 2.3 for GLN and CONT groups, respectively (p = 0.23). Among the comorbid conditions, smoking, alcohol consumption, and dyslipidemia incidences were also similar between groups, as shown in table I. Preexisting type 2 diabetes mellitus was observed in a large proportion of our patients; however, the most common comorbid condition was arterial hypertension, with an incidence of 78.5% of the total sample (n = 22). The preoperative evaluation of cardiac function revealed a mean left ventricular ejection fraction of 52.5 ± 13.3 and 48.0 ± 28.3 L/min in the GLN and CONT groups, respectively (p = 0.60). The total pump time during surgery was 111.0 ± 40 min in the GLN group and 127.8 ± 38.1 min in the CONT group (p = 0.27). The total duration of aortic clamping was similar between groups.
CARDIAC INJURY MARKERS
Troponin-I serum levels at 24 hours postoperatively were lower in the GLN group (1.9 ± 0.24) than in the CONT group (3.02 ± 0.25). This difference was statically significant (p = 0.004). At 12 hours postoperatively, their levels were also lower in comparison to the CONT group (2.07 ± 0.21 vs. 2.7 ± 0.26, p = 0.06); troponin-I levels were slightly lower after surgery, as shown in figure 1A.
Similarly, CPK levels were also found to be lower in the GLN group than in the CONT group at 12 hours postoperatively. The difference was statically significant (p = 0.01), as shown in figure 1B.
Regarding to CPK-Mb, serum levels were also lower in the GLN group than in the CONT group at 1, 12, and 24 hours postoperatively with significant differences, as shown in figure 1C.
POSTOPERATIVE COURSE
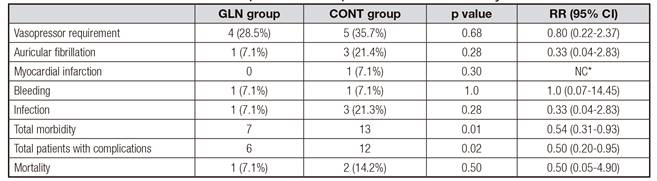
The complications in both groups are described in table II. There were no differences in individual complications between groups. These included postoperative vasopressor requirement, postoperative arrhythmias (atrial fibrillation), myocardial infarction, postoperative bleeding requiring surgical reintervention for hemostasis, and infections, which included two episodes of pneumonia, one of which also developed purulent mediastinitis (CONT group), and a urinary tract infection in one patient (GLN group). The overall morbidity was significantly less frequent in the GLN group (p = 0.01, RR = 0.54, 95% CI 0.31-0.93) and the number of patients with complications was also lower in the GLN group (p = 0.02, RR = 0.50, 95% CI 0.20-0.95%).
Mortality was observed in 3 patients. Of these, multiple organ failure was observed in two cases (pneumonia and pneumonia plus purulent mediastinitis) in the CONT group (14.2%) and pulmonary embolism was observed in one case in the GLN group (7.1%); no significant difference was observed between the groups (p = 0.54). The mean length of hospital stay in the UCI was 4 ± 1.9 days for the GLN group and 4.7 ± 2.4 days for the CONT group, with no significant difference (p = 0.24).
DISCUSSION
Preoperative oral supplementation of GLN at a standardized dose of 0.5 g/kg/day in our study group showed a significant reduction in postoperative myocardial damage. Lower TROP-I, CPK-Mb, and CPK levels were observed in patients receiving GLN, and the total morbidity and total number of patients with any complications was also lower in this group.
Elevated TROP-I levels are common in patients who undergo CPB. A close correlation between TROP-I concentration and mortality during postoperative follow-up has been recognized 19. Numerous mechanisms for the cardioprotective effects of GLN have been described in multiple studies 20.
GLN is also an indirect precursor of GSH, and is involved in antioxidant protection by increasing the ratio of reduced to oxidized GSH. The correlation between the perioperative use of GLN and plasma GSH concentrations in patients after CPB has been previously shown 21. Moreover, in cases of I/R injury, GLN increases the myocardial adenosine triphosphate- adenosine diphosphate ratio and prevents intracellular lactate accumulation 9.
The supplementation of GLN has contributed new and valuable information to allow clinicians to understand the mechanism associated with the reduction in myocardial damage after heart surgery. Lomivorotov et al. in 2011, demonstrated a significant reduction in TROP-I levels after CPB with coronary artery bypass grafting at 24 hours postoperatively (p = 0.035) in patients who received intravenous GLN (Dipeptiven, 0.4 g/kg per day; Fresenius Kabi, Bad Homburg, Germany); the median stroke index was also higher in the study group (p = 0.023), and there was decreased median systemic vascular resistance (p = 0.001). There were no significant differences in the postoperative complications or mortality between groups (13). Under that same line of investigation, in 2012, Sufit and colleagues also observed that GLN conferred cardiac protection when supplemented orally (37.5 g/per day) during the 3 days prior to surgery in patients who underwent CPB without artery bypass grafting. TROP-I, CPK, and CPK-Mb levels were significantly lower at 24, 48, and 72 hours postoperatively (p < 0.03, p < 0.05, and p < 0.04), respectively, in the study group. This reduction in myocardial injury was associated with a reduction in clinical complications (p = 0.03). This pilot feasibility trial assessed the safety, tolerability, and patient compliance of preoperative supplementation of oral GLN therapy in patients undergoing CPB. Given the small sample size (n = 10), the potential for clinical efficacy must be confirmed in a larger, definitive multicenter trial of GLN therapy prior to cardiac surgery 22.
In comparison with previous studies of oral supplementation of GLN, we observed a stronger effect in the GLN group. Comorbidities were also observed in a large number of patients, at both study groups, as shown in table I. The determination of cardiac injury markers are indicative of myocardial damage and worse prognosis, that's why it was so important in our study to measure all the markers to relate our results, that also support those previously reported regarding the reduction of cardiac injury markers such as TROP-I, CPK-Mb, and CPK, suggesting a beneficial clinical effect in patients undergoing open heart surgery for multi-vessel ischemic disease, reducing myocardial I/R injury during CPB in Mexican patients with this exclusive condition 21. A convenient and practical administration regimen of preoperative GLN at safe doses (0.5 g/kg per day and cumulative) resulted in a robust benefit that is a favorable benefit-risk. This action resulted in a decrease in all cardiac injury CPB patients. There were no significant differences in clamp time or pump time between groups, and times were consistent with published normal and safe values. In our cohort, the comorbid conditions presented before surgery may have influenced postoperative morbidity but nevertheless they also had clear and robust benefit and was safe and well tolerated included in this patient population with multiple clinical conditions and different morbidities, glutamine was effective. Diabetes is a risk factor for cardiovascular disease and is associated with an elevated risk of coronary heart disease, hence the impaction glutamine on cardiac biochemistry and function may reflect in cardioprotection beyond purely ischemic events, but also in conditions of metabolic dysregulation that impact cardiac and cardiovascular function. More than 50% of our patients suffered diabetes mellitus in addition to another comorbid condition. Recently, Mansour et al. 23 demonstrated that 6 weeks of oral GLN supplementation (30 g/d) was safe and significantly reduced some cardiovascular risk factors such as fasting plasma glucose, HbA1c levels, blood pressure, and waist circumference in patients with type 2 diabetes. In addition, the oral GLN supplementation reduced the effect of a high-fat diet on the incidence of obesity, which is an important risk factor in the development of myocardial infarction.
In our study, the number of postsurgical complications, such as vasopressor requirement, atrial fibrillation, and myocardial infarction, was less frequent in the GLN group. The clinical population of this study had a lot of comorbidities and cardiovascular risk factors. The fact that GLN was consistently effective (and safe at the regimen dose used) in this clinical population, opens a new possibilities for more detailed and larger studies on GLN and cardioprotection, impact on cardiac and vascular function, and complications. Glutamine depletion occurs in critically injured patients, and may contribute to the high rate of infection. In our study we had 3 patients complicated with infection in the control (CONT) group and 1 in the GLN group. Although our sample was relatively small, a fourteen control clinical trials meta-analysis done by Wang et al., showed that the risk of infection was reduced in patients who received GLT parenteral nutrition compared with normal parenteral nutrition (p = 0.02) 24, other possible directions for further research of oral supplementation of glutamine in patients with multiple co-morbidities and different disease conditions and underlying pathophysiologies, like is a fact known that the myocardial concentration of glutamine changes during ischemia and reperfusion due to alterations in metabolic and ionic processes.
Because myocardial damage is directly related to the degree of cell damage by ischemia, deeper and wider intervention in more patients will clarify the cardioprotective effects of GLN in cardiac surgery. More complicated cases could be interesting, and may help to strengthen our results.
This form of oral supplementation may be a safe and practical way for the patient to lower the concentrations of TROP-I during the postoperative period without the need for medical assistance. Because both oral and intravenous administrations were shown to reduce the equally dependent perioperative variables in previous studies, it is recommended that a study of both interventions is conducted with the same type of patients during the pre/postoperative period.
The present study has some limitations that must be addressed. We were unable to study all of the mechanisms involved in the cardioprotective effects of GLN. Thus, it may be interesting to evaluate whether a correlation exists between GLN oral supplementation and myocardial HSP expression, as well as proinflammatory interleukins IL-1, IL-6, TNF-, and myoglobin, which have been found to contribute to cardiac injury 25. Another limitation was that the cohort included patients with a low ejection fraction (< 50%) and with comorbidities such as diabetes mellitus (64.2%) and hypertension (78.5%), which may explain the relatively high morbidity in both groups of patients, although it can be considered a limitation of the study, the observation of a beneficial effect of oral glutamine (at a safe regimen) in the clinical patient population of the present study (that is, with heterogeneous disease condition and comorbidities) also suggest that glutamine has effect in these complex population which then may benefit from preoperative oral supplementation of glutamine, this needs to be followed in larger clinical studies.
In summary, the majority of these findings may be related to GLN supplementation, suggesting that it has the ability to enhance cell survival during I/R, attenuate the systemic inflammatory response, oxidative stress, apoptosis modulation, and the integrity of cardiac cells by increasing myocardial ATP/ADP substrate, and enhance myocardial GSH 26.
CONCLUSIONS
This study demonstrated that oral supplementation of GLN prior to cardiac surgery confers a protective effect on the heart by decreasing the biochemical injury markers and clinical complications. These data indicate that a larger number of trials with GLN supplementation, whether orally or intravenously administered to patients undergoing cardiac surgery, are needed to confirm a clinical benefit. Future GLN dose-response studies are warranted in these areas.